Label: LINCOMYCIN- lincomycin injection, solution
- NDC Code(s): 42571-374-75, 42571-374-88
- Packager: Micro Labs Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated July 2, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
BOXED WARNING
(What is this?)
WARNING
Clostridioides difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including lincomycin and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
Because lincomycin therapy has been associated with severe colitis which may end fatally, it should be reserved for serious infections where less toxic antimicrobial agents are inappropriate, as described in the INDICATIONS AND USAGE section. It should not be used in patients with nonbacterial infections such as most upper respiratory tract infections.
C. diffficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
-
DESCRIPTION
Lincomycin injection, USP is a sterile solution containing lincomycin hydrochloride which is the monohydrated salt of lincomycin, a lincosamide antibacterial produced by the growth of a member of the lincolnensis group of Streptomyces lincolnensis (Fam. Streptomycetaceae). The chemical name for lincomycin hydrochloride is Methyl 6,8-dideoxy-6-(1-methyl-trans-4-propyl-L2-pyrolidinecarboxamido)-1-thio-D-erythro-α-D-galacto-octopyranoside monohydrochloride monohydrate. The molecular formula of lincomycin hydrochloride is C 18H 34N 2O 6S.HCl.H 2O and the molecular weight is 461.01.
The structural formula is represented below:
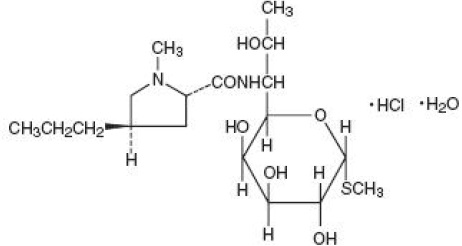
Lincomycin hydrochloride is a white or practically white, crystalline powder. Its solutions are acid and are dextrorotatory. Lincomycin hydrochloride is freely soluble in water; soluble in dimethylformamide and insoluble in acetone.
Lincomycin Injection, USP contains lincomycin hydrochloride in a sterile, clear, colorless to slightly yellow solution with benzyl alcohol used as a preservative 9.45 mg/mL, and water for injection.
Lincomycin injection, USP is a sterile solution for intramuscular and intravenous use. Lincomycin injection, USP is supplied in 2 mL and 10 mL multiple-dose vials containing 300 mg/mL of lincomycin (equivalent to 340 mg/mL of lincomycin hydrochloride, USP).
-
CLINICAL PHARMACOLOGY
Intramuscular administration of a single dose of 600 mg of lincomycin produces average peak serum concentrations of 11.6 mcg/mL at 60 minutes and maintains therapeutic concentrations for 17 to 20 hours for most susceptible gram-positive organisms. Urinary excretion after this dose ranges from 1.8 to 24.8 percent (mean: 17.3 percent).
A two hour intravenous infusion of 600 mg of lincomycin achieves average peak serum concentrations of 15.9 mcg/mL and maintains therapeutic concentrations for 14 hours for most susceptible gram-positive organisms. Urinary excretion ranges from 4.9 to 30.3 percent (mean: 13.8 percent).
The biological half-life after intramuscular or intravenous administration is 5.4 ± 1.0 hours. The serum half-life of lincomycin may be prolonged in patients with severe renal impairment compared to patients with normal renal function. In patients with hepatic impairment, serum half-life may be twofold longer than in patients with normal hepatic function. Hemodialysis and peritoneal dialysis are not effective in removing lincomycin from the serum.
Tissue distribution studies indicate that bile is an important route of excretion. Significant concentrations have been demonstrated in most body tissues. Although lincomycin appears to diffuse into cerebrospinal fluid (CSF), concentrations of lincomycin in the CSF appear inadequate for the treatment of meningitis.
Microbiology:
Mechanism of Action
Lincomycin inhibits bacterial protein synthesis by binding to the 23S RNA of the 50S subunit of the bacterial ribosome. Lincomycin is predominantly bacteriostatic in vitro.
Resistance
Cross resistance has been demonstrated between clindamycin and lincomycin. Resistance is most often due to methylation of specific nucleotides in the 23S RNA of the 50S ribosomal subunit, which can determine cross resistance to macrolides and streptogramins B (MLSB phenotype). Macrolide-resistant isolates of these organisms should be tested for inducible resistance to lincomycin/clindamycin using the D-zone test or other appropriate method.
Antimicrobial Activity
Lincomycin has been shown to be active against most strains of the following bacteria both in vitro and in clinical infections: (see INDICATIONS AND USAGE).
Staphylococcus aureus
Streptococcus pneumoniae
The following in vitro data are available, but their clinical significance is unknown.
Lincomycin has been shown to be active in vitro against the following microorganisms; however, the safety and efficacy of lincomycin in treating clinical infections due to these organisms have not been established in adequate and well controlled trials.
Gram-positive bacteria:
Corynebacterium diphtheriae
Streptococcus pyogenes
Viridans group streptococci
Anaerobic bacteria:
Clostridium tetani
Clostridium perfringens
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
-
INDICATIONS AND USAGE
Lincomycin injection is indicated in the treatment of serious infections due to susceptible strains of streptococci, pneumococci, and staphylococci. Its use should be reserved for penicillin-allergic patients or other patients for whom, in the judgment of the physician, a penicillin is inappropriate. Because of the risk of CDAD, as described in the BOXED WARNING, before selecting lincomycin the physician should consider the nature of the infection and the suitability of other alternatives.
Indicated surgical procedures should be performed in conjunction with antibacterial therapy.
Lincomycin injection may be administered concomitantly with other antimicrobial agents when indicated.
Lincomycin is not indicated in the treatment of minor bacterial infections or viral infections.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of lincomycin and other antibacterial drugs, lincomycin should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
- CONTRAINDICATIONS
-
WARNINGS
See BOXED WARNING.
Clostridioides difficile associated diarrhea
Clostridioides difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Lincomycin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against
C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Hypersensitivity
Severe hypersensitivity reactions, including anaphylactic reactions and severe cutaneous adverse reactions (SCAR) such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), acute generalized exanthematous pustulosis (AGEP), and erythema multiforme (EM) have been reported in patients receiving lincomycin therapy. If an anaphylactic reaction or severe skin reaction occurs, lincomycin should be discontinued and appropriate therapy should be initiated. (see ADVERSE REACTIONS)
Benzyl Alcohol Toxicity in Pediatric Patients (Gasping Syndrome)
Lincomycin injection contains benzyl alcohol as a preservative.
The preservative benzyl alcohol has been associated with serious adverse events, including the “gasping syndrome”, and death in pediatric patients. Although normal therapeutic doses of this product ordinarily deliver amounts of benzyl alcohol that are substantially lower than those reported in association with the “gasping syndrome”, the minimum amount of benzyl alcohol at which toxicity may occur is not known. The risk of benzyl alcohol toxicity depends on the quantity administered and the liver and kidneys’ capacity to detoxify the chemical. Premature and low-birth weight infants may be more likely to develop toxicity.
Inadequate for Use in Meningitis
Although lincomycin appears to diffuse into cerebrospinal fluid, concentrations of lincomycin in the CSF may be inadequate for the treatment of meningitis.
-
PRECAUTIONS
General
Review of experience to date suggests that a subgroup of older patients with associated severe illness may tolerate diarrhea less well. When lincomycin is indicated in these patients, they should be carefully monitored for change in bowel frequency.
Lincomycin should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
Lincomycin should be used with caution in patients with a history of asthma or significant allergies.
Certain infections may require incision and drainage or other indicated surgical procedures in addition to antibacterial therapy.
The use of lincomycin may result in overgrowth of nonsusceptible organisms, particularly yeasts. Should superinfections occur, appropriate measures should be taken as indicated by the clinical situation. When patients with pre-existing Candida infections require therapy with lincomycin, concomitant antifungal treatment should be given.
The serum half-life of lincomycin may be prolonged in patients with severe renal impairment compared to patients with normal renal function. In patients with hepatic impairment, serum half-life may be twofold longer than in patients with normal hepatic function.
Patients with severe renal impairment and/or hepatic impairment should be dosed with caution and serum lincomycin concentrations monitored during high-dose therapy. (see DOSAGE AND ADMINISTRATION)
Lincomycin MUST be diluted prior to intravenous infusion. For intravenous infusion, infuse over at least 60 minutes as directed in the DOSAGE AND ADMINISTRATION Section. Do NOT administer as an intravenous bolus. Severe cardiopulmonary reactions have occurred at greater than the recommended concentration and rate.
Prescribing lincomycin in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Information for Patients
Patients should be counseled that antibacterial drugs including lincomycin should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When lincomycin is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by lincomycin or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibacterial which usually ends when the antibacterial is discontinued. Sometimes after starting treatment with an antibacterial, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after taking the last dose of the antibacterial. If this occurs, patients should contact their physician as soon as possible.
Laboratory Tests
During prolonged therapy with lincomycin, periodic liver and kidney function tests and blood counts should be performed.
Drug Interactions
Lincomycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents; therefore, it should be used with caution in patients receiving such agents.
Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic potential of lincomycin has not been evaluated.
Lincomycin was not found to be mutagenic in the Ames Salmonella reversion assay or the V79 Chinese hamster lung cells at the HGPRT locus. It did not induce DNA strand breaks in V79 Chinese hamster lung cells as measured by alkaline elution or chromosomal abnormalities in cultured human lymphocytes. In vivo, lincomycin was negative in both the rat and mouse micronucleus assays and it did not induce sex-linked recessive lethal mutations in the offspring of male Drosophila. However, lincomycin did cause unscheduled DNA syntheses in freshly isolated rat hepatocytes.
Impairment of fertility was not observed in male or female rats given oral 300 mg/kg doses of lincomycin (0.36 times the highest recommended human dose based on mg/m 2).
Pregnancy
There are no adequate and well-controlled studies in pregnant women. Lincomycin Sterile Solution contains benzyl alcohol as a preservative. Benzyl alcohol can cross the placenta. See WARNINGS. Lincomycin should be used during pregnancy only if clearly needed.
Teratogenic Effects:
In a study with 60 pregnant women, cord serum concentrations were approximately 25% of the maternal serum concentrations, indicating that lincomycin crosses the placenta, and no substantial accumulation occurred in the amniotic fluid. Experience with 345 obstetrical patients receiving lincomycin revealed no ill effects related to pregnancy.
There was no evidence of teratogenicity when lincomycin was administered in diet to pregnant Sprague Dawley rats during the period of major organogenesis at doses up to 5000 mg/kg (approximately 6 times the maximum recommended human dose [MRHD], respectively, based on body surface area comparison).
Nonteratogenic Effects:
Reproduction studies performed in rats administered oral lincomycin in diet for 2 weeks prior to mating, throughout pregnancy and lactation, revealed no adverse effects on survival of offspring from birth to weaning at doses up to 1000 mg/kg (1.2 times the MRHD based on body surface area comparison) up to 2 generations.
Nursing Mothers
Lincomycin has been reported to appear in human milk in concentrations of 0.5 to 2.4 mcg/mL. Because of the potential for serious adverse reactions in nursing infants from lincomycin, a decision should be made whether to discontinue nursing, or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Lincomycin injection contains benzyl alcohol as a preservative. Benzyl alcohol has been associated with a fatal “Gasping Syndrome” in premature infants. See WARNINGS. Safety and effectiveness in pediatric patients below the age of one month have not been established. (see DOSAGE AND ADMINISTRATION)
-
ADVERSE REACTIONS
The following adverse reactions have been reported with the use of lincomycin.
Gastrointestinal disorders
Diarrhea, nausea, vomiting, glossitis, stomatitis, abdominal pain, abdominal discomfort†, anal pruritus
Skin and subcutaneous tissue disorders
Toxic epidermal necrolysis, Stevens-Johnson syndrome, acute generalized exanthematous pustulosis, dermatitis bullous, dermatitis exfoliative, erythema multiforme (see WARNINGS), rash, urticaria, pruritus
Infections and infestations
Vaginal infection, pseudomembranous colitis, Clostridioides difficile colitis (see WARNINGS)
Blood and lymphatic system disorders
Pancytopenia, agranulocytosis, aplastic anemia, leukopenia, neutropenia, thrombocytopenic purpura
Immune system disorders
Anaphylactic reaction (see WARNINGS), angioedema, serum sickness
Hepatobiliary disorders
Jaundice, liver function test abnormal, transaminases increased
Renal and urinary disorders
Renal impairment, oliguria, proteinuria, azotemia
Cardiac disorders
Cardio-respiratory arrest (see DOSAGE AND ADMINISTRATION)
Vascular disorders
Hypotension (see DOSAGE AND ADMINISTRATION), thrombophlebitis†
Ear and labyrinth disorders
Vertigo, tinnitus
Neurologic disorders
Headache, dizziness, somnolence
General disorders and administration site conditions
Injection site abscess sterile‡, injection site induration‡, injection site pain‡, injection site irritation‡
†Event has been reported with intravenous injection.
‡Reported with intramuscular injection.
To report SUSPECTED ADVERSE REACTIONS, contact Micro Labs USA, Inc. at 1-855-839-8195 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
- OVERDOSAGE
-
DOSAGE AND ADMINISTRATION
If significant diarrhea occurs during therapy, lincomycin should be discontinued. (see BOXED WARNING)
INTRAMUSCULAR - Adults: Serious infections—600 mg (2 mL) intramuscularly every 24 hours. More severe infections—600 mg (2 mL) intramuscularly every 12 hours or more often. Pediatric patients over 1 month of age: Serious infections—one intramuscular injection of 10 mg/kg (5 mg/lb) every 24 hours. More severe infections— one intramuscular injection of 10 mg/kg (5 mg/lb) every 12 hours or more often.
INTRAVENOUS - Adults: The intravenous dose will be determined by the severity of the infection. For serious infections doses of 600 mg of lincomycin (2 mL of lincomycin) to 1 gram are given every 8 to 12 hours. For more severe infections these doses may have to be increased. In life-threatening situations daily intravenous doses of as much as 8 grams have been given. Intravenous doses are given on the basis of 1 gram of lincomycin diluted in not less than 100 mL of appropriate solution (see PHYSICAL COMPATIBILITIES) and infused over a period of not less than one hour.
Dose
Vol. Diluent
Time
600 mg100 mL
1 hr1 gram
100 mL
1 hr
2 grams200 mL
2 hr
3 grams300 mL
3 hr
4 grams400 mL
4 hr
These doses may be repeated as often as required to the limit of the maximum recommended daily dose of 8 grams of lincomycin.
Pediatric patients over 1 month of age: 10 to 20 mg/kg/day (5 to 10 mg/lb/day) depending on the severity of the infection may be infused in divided doses as described above for adults.
NOTE: Severe cardiopulmonary reactions have occurred when lincomycin has been given at greater than the recommended concentration and rate (see PRECAUTIONS).
SUBCONJUNCTIVAL INJECTION - 0.25 mL (75 mg) injected subconjunctivally will result in ocular fluid concentrations of antibacterial (lasting for at least 5 hours) sufficient for most susceptible pathogens.
Patients with Renal Impairment
When therapy with lincomycin is required in individuals with severe renal impairment, an appropriate dose is 25 to 30% of that recommended for patients with normally functioning kidneys (see PRECAUTIONS).
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
-
HOW SUPPLIED
Lincomycin Injection, USP is a sterile, clear, colorless to slightly yellow solution, having a slight odor. Free from any visible particles available in the following strength and package sizes:
Unit of Sale Concentration NDC 42571-374-75
2 mL multiple-dose vial600 mg/2 mL
(300 mg/mL)NDC 42571-374-88
10 mL multiple-dose vial3,000 mg/10 mL
(300 mg/mL)
Each mL of Lincomycin Injection, USP contains 300 mg lincomycin (equivalent to 340 mg lincomycin hydrochloride, USP); also benzyl alcohol, 9.45 mg added as preservative.
Store at controlled room temperature 20° to 25°C (68° to 77°F) [see USP].
-
ANIMAL PHARMACOLOGY
In vivo experimental animal studies demonstrated the effectiveness of lincomycin preparations (lincomycin) in protecting animals infected with Streptococcus viridans, β- hemolytic Streptococcus, Staphylococcus aureus, Streptococcus pneumoniae and Leptospira pomona. It was ineffective in Klebsiella, Pasteurella, Pseudomonas, Salmonella and Shigella infections .
-
PHYSICAL COMPATIBILITIES
Physically compatible for 24 hours at room temperature unless otherwise indicated.
Infusion Solutions
5% Dextrose Injection
10% Dextrose Injection
5% Dextrose and 0.9% Sodium Chloride Injection 10% Dextrose and 0.9% Sodium Chloride Injection Ringer’s Injection
1/6 M Sodium Lactate Injection Travert 10%-Electrolyte No. 1
Dextran in Saline 6% w/v
Vitamins in Infusion Solutions
B-Complex
B-Complex with Ascorbic Acid
Antibacterial in Infusion Solutions
Penicillin G Sodium (Satisfactory for 4 hours)
Cephalothin
Tetracycline HCl
Cephaloridine
Colistimethate (Satisfactory for 4 hours)
Ampicillin
Methicillin
Chloramphenicol
Polymyxin B Sulfate
Physically Incompatible with:
Novobiocin
Kanamycin
IT SHOULD BE EMPHASIZED THAT THE COMPATIBLE AND INCOMPATIBLE DETERMINATIONS ARE PHYSICAL OBSERVATIONS ONLY, NOT CHEMICAL DETERMINATIONS. ADEQUATE CLINICAL EVALUATION OF THE SAFETY AND EFFICACY OF THESE COMBINATIONS HAS NOT BEEN PERFORMED.
Rx only
Manufactured by:
Micro Labs Limited
Bangalore-560099, INDIA.
Manufactured for:
Micro Labs USA, Inc.
Somerset, NJ 08873
Rev.07/2022
-
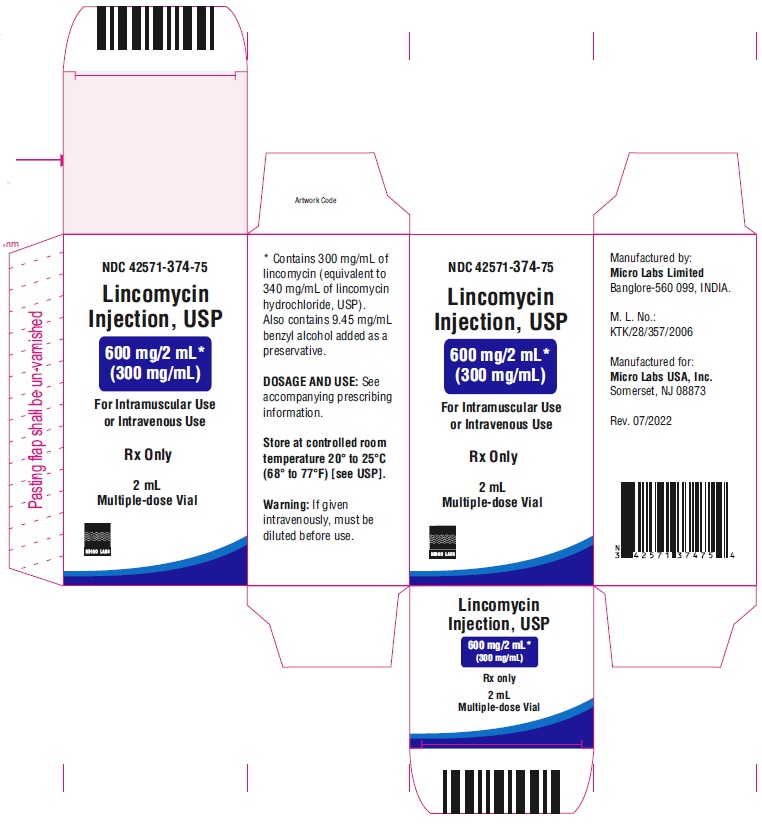
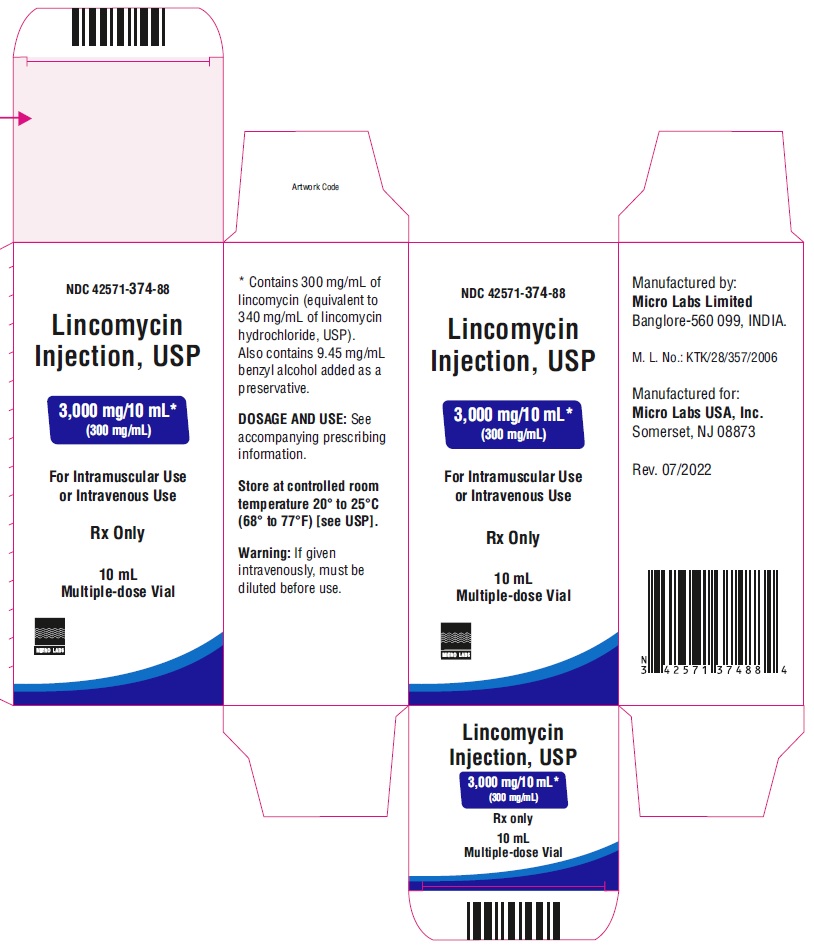
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC-42571-374-75
Lincomycin Injection, USP
600 mg/ 2 mL*
(300 mg/mL)
For Intramuscular Use or
Intravenous UseRx only
2 mL Multiple-dose Vial
Micro Labs Limited
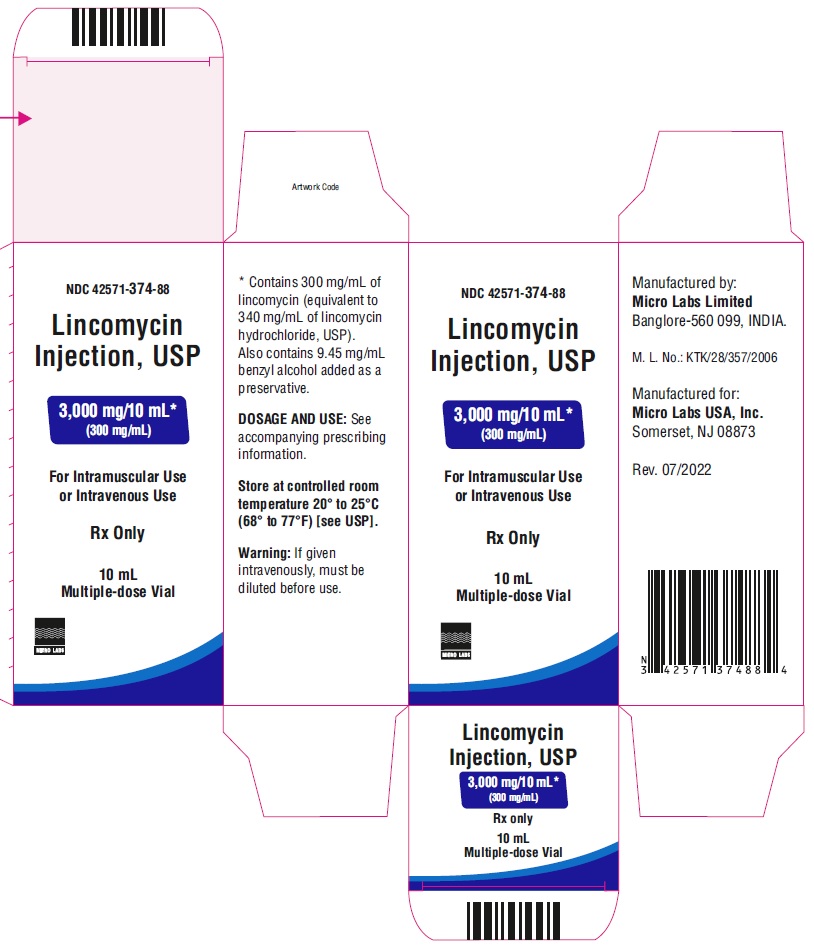
NDC-42571-374-88Lincomycin Injection, USP
3,000 mg/ 10 mL*
(300 mg/mL)
For Intramuscular Use or
Intravenous UseRx only
10 mL Multiple-dose Vial
Micro Labs Limited
-
INGREDIENTS AND APPEARANCE
LINCOMYCIN
lincomycin injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:42571-374 Route of Administration INTRAVENOUS, INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LINCOMYCIN HYDROCHLORIDE (UNII: M6T05Z2B68) (LINCOMYCIN - UNII:BOD072YW0F) LINCOMYCIN 300 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZYL ALCOHOL (UNII: LKG8494WBH) 9.45 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42571-374-75 1 in 1 CARTON 02/01/2022 1 2 mL in 1 VIAL; Type 0: Not a Combination Product 2 NDC:42571-374-88 1 in 1 CARTON 02/01/2022 2 10 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA215082 02/01/2022 Labeler - Micro Labs Limited (862174955) Establishment Name Address ID/FEI Business Operations Micro Labs Limited 677600482 analysis(42571-374) , label(42571-374) , manufacture(42571-374) , pack(42571-374)