Label: CYCLOGYL- cyclopentolate hydrochloride solution/ drops
-
NDC Code(s):
0065-0395-15,
0065-0396-02,
0065-0396-05,
0065-0396-15, view more0065-0397-02, 0065-0397-05, 0065-0397-15
- Packager: Alcon Laboratories, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated June 25, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Rx Only
DESCRIPTION: CYCLOGYL® (cyclopentolate hydrochloride ophthalmic solution, USP) is an anticholinergic prepared as a sterile, borate buffered, solution for topical ocular use. It is supplied in three strengths. The active ingredient is represented by the structural formula:
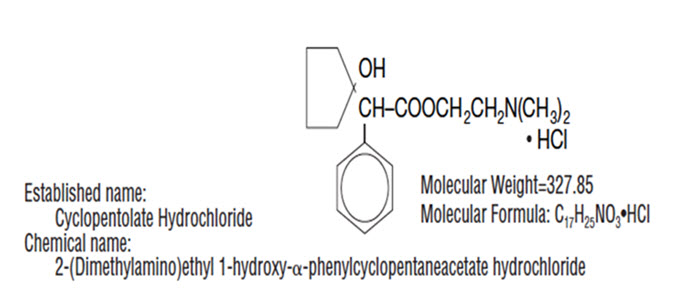
Each mL of CYCLOGYL® (cyclopentolate hydrochloride ophthalmic solution, USP) contains: Active: cyclopentolate hydrochloride 0.5%, 1% or 2%. Preservative: benzalkonium chloride 0.01%. Inactives: boric acid, edetate disodium, potassium chloride (except 2% strength), sodium carbonate and/or hydrochloric acid (to adjust pH), purified water. The pH range is between 3.0 and 5.5.
-
CLINICAL PHARMACOLOGY
CLINICAL PHARMACOLOGY: This anticholinergic preparation blocks the responses of the sphincter muscle of the iris and the accommodative muscle of the ciliary body to cholinergic stimulation, producing pupillary dilation (mydriasis) and paralysis of accommodation (cycloplegia). It acts rapidly, but has a shorter duration than atropine. Maximal cycloplegia occurs within 25 to 75 minutes after instillation. Complete recovery of accommodation usually takes 6 to 24 hours. Complete recovery from mydriasis in some individuals may require several days. Heavily pigmented irides may require more doses than lightly pigmented irides.
- INDICATIONS & USAGE
- CONTRAINDICATIONS
-
WARNINGS
WARNINGS: FOR TOPICAL OPHTHALMIC USE ONLY. NOT FOR INJECTION. This preparation may cause Central Nervous System (CNS) disturbances. This is especially true in younger age groups, but may occur at any age, especially with the stronger solutions. Infants are especially prone to CNS and cardiopulmonary side effects from cyclopentolate. To minimize absorption, use only 1 drop of 0.5% CYCLOGYL solution per eye, followed by pressure applied over the nasolacrimal sac for two to three minutes. Observe infants closely for at least 30 minutes following instillation.
Mydriatics may produce a transient elevation of intraocular pressure. Therefore, patients with untreated narrow angle glaucoma or anatomically narrow angles may be susceptible to angle closure following administration.
-
PRECAUTIONS:
General: The lacrimal sac should be compressed by digital pressure for two to three minutes after instillation to reduce excessive systemic absorption. Caution should be observed when considering use of this medication in the presence of Down's syndrome.
Information for Patients: Do not touch dropper tip to any surface, as this may contaminate the solution. A transient burning sensation may occur upon instillation. Patients should be advised not to drive or engage in other hazardous activities while pupils are dilated. Patients may experience sensitivity to light and should protect eyes in bright illumination during dilation. Parents should be warned not to get this preparation in their child's mouth and to wash their own hands and the child's hands following administration. Feeding intolerance may follow ophthalmic use of this product in infants. It is recommended that feeding be withheld for 4 hours after examination.
Drug Interactions: Cyclopentolate may interfere with the ocular anti-hypertensive action of carbachol, pilocarpine, or ophthalmic cholinesterase inhibitors.
Carcinogenesis, Mutagenesis, Impairment of Fertility: Studies in animals or humans have not been conducted to evaluate the carcinogenic potential of CYCLOGYL® (cyclopentolate hydrochloride ophthalmic solution, USP).
Pregnancy: Animal reproduction studies have not been conducted with cyclopentolate. It is also not known whether cyclopentolate can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Cyclopentolate should be administered to a pregnant woman only if clearly needed.
Nursing Mothers: It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when cyclopentolate hydrochloride is administered to a nursing woman.
Pediatric Use: Use of cyclopentolate has been associated with psychotic reactions and behavioral disturbances in pediatric patients. Increased susceptibility to cyclopentolate has been reported in infants, young children, and in children with spastic paralysis or brain damage. These disturbances include ataxia, incoherent speech, restlessness, hallucinations, hyperactivity, seizures, disorientation as to time and place, and failure to recognize people. Feeding intolerance and necrotizing enterocolitis (NEC) in preterm infants may follow ophthalmic use of this product. Cases of NEC have been reported in preterm infants following administration; however, causality has not been established. It is recommended that feeding be withheld for four (4) hours after examination. Observe infants closely for at least 30 minutes (see WARNINGS).
-
ADVERSE REACTIONS:
Ocular: Increased intraocular pressure, burning, photophobia, blurred vision, irritation, hyperemia, conjunctivitis, blepharoconjunctivitis, punctate keratitis, synechiae have been reported.
Non-ocular: Use of cyclopentolate has been associated with psychotic reactions and behavioral disturbances, usually in children, especially with 2% concentration. These disturbances include ataxia, incoherent speech, restlessness, hallucinations, hyperactivity, seizures, disorientation as to time and place, and failure to recognize people. This drug produces reactions similar to those of other anticholinergic drugs, but the central nervous system manifestations as noted above are more common. Other toxic manifestations of anticholinergic drugs are skin rash, abdominal distention in infants, unusual drowsiness, tachycardia, hyperpyrexia, vasodilation, urinary retention, diminished gastrointestinal motility and decreased secretion in salivary and sweat glands, pharynx, bronchi and nasal passages. Severe manifestations of toxicity include coma, medullary paralysis and death.
-
OVERDOSAGE
OVERDOSAGE: Excessive dosage may produce behavioral disturbances, tachycardia, hyperpyrexia, hypertension, elevated intraocular pressure, vasodilation, urinary retention, diminished gastrointestinal motility and decreased secretion in salivary and sweat glands, pharynx, bronchi and nasal passages. Patients exhibiting signs of overdosage should receive supportive care and monitoring.
-
DOSAGE & ADMINISTRATION
DOSAGE AND ADMINISTRATION: Adults: Instill one or two drops of 0.5%, 1% or 2% solution in the eye which may be repeated in five to ten minutes if necessary. Complete recovery usually occurs in 24 hours. Complete recovery from mydriasis in some individuals may require several days. Children: Instill one or two drops of 0.5%, 1% or 2% solution in the eye which may be repeated five to ten minutes later by a second application of 0.5% or 1% solution if necessary. Small Infants: A single instillation of one drop of 0.5% in the eye. To minimize absorption, apply pressure over the nasolacrimal sac for two to three minutes. Observe infant closely for at least 30 minutes following instillation. Individuals with heavily pigmented irides may require higher strengths.
-
HOW SUPPLIED
HOW SUPPLIED: In multiple-dose plastic DROP-TAINER® dispensers:
0.5% CYCLOGYL 1% CYCLOGYL 2% CYCLOGYL
15 mL NDC 0065-0395-15 2 mL NDC 0065-0396-02 2 mL NDC 0065-0397-02
5 mL NDC 0065-0396-05 5 mL NDC 0065-0397-05
15 mL NDC 0065-0396-15 15 mL NDC 0065-0397-15
Storage: Store at 8°C to 25°C (46°F to 77°F). After opening, Cyclogyl can be used until the expiration date on the bottle.
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL
NDC 0065-0395-15
Alcon
Cyclogyl® 0.5%
(cyclopentolate hydrochloride ophthalmic solution, USP)
15 mL Sterile
INGREDIENTS: Each mL contains: Active: cyclopentolate hydrochloride 0.5%. Preservative: benzalkonium chloride 0.01%. Inactives: boric acid, edetate disodium, potassium chloride, sodium carbonate and/or hydrochloric acid (to adjust pH), purified water. The pH range is between 3.0 and 5.5.
FOR TOPICAL OPHTHALMIC USE ONLY.
Rx Only
USUAL DOSAGE: Instill one or two drops in the eye which can be repeated in five to ten minutes. Read enclosed insert.
PRECAUTION: Do not touch dropper tip to any surface, as this may contaminate the solution.
STORAGE: Store at 8° to 25°C (46°-77°F). After opening Cyclogyl can be used until the expiration date on the bottle.
ALCON
ALCON LABORATORIES, INC.
Fort Worth, Texas 76134 USA
Product of Spain
300037981-0520
SN:
LOT:
EXP.:
GTIN: 00300650395151
NDC 0065-0395-15
Alcon
Cyclogyl® 0.5%
(cyclopentolate hydrochloride ophthalmic solution, USP)
Sterile 15 mL
Rx Only
INGREDIENTS: Each mL contains : Active: cyclopentolate hydrochloride 0.5%. Preservative: benzalkonium chloride 0.01%. Inactives: boric acid, edetate disodium, potassium chloride, sodium carbonate and/or hydrochloric acid (to adjust pH), purified water. The pH range is between 3.0 and 5.5.
PRECAUTION: Do not touch dropper tip to any surface, as this may contaminate the solution.
FOR TOPICAL OPHTHALMIC USE ONLY
USUAL DOSAGE: Instill one or two drops in the eye which can be repeated in five to ten minutes if necessary. Read enclosed insert.
STORAGE: Store at 8° to 25°C (46° to 77°F).
ALCON LABORATORIES, INC.
Fort Worth, Texas 76134 USA
Printed in USA
300037980-0520
LOT: EXP.:
NDC 0065-0396-05
Alcon
Cyclogyl® 1%
(cyclopentolate hydrochloride ophthalmic solution, USP)
5 mL Sterile
INGREDIENTS: Each mL contains : Active: cyclopentolate hydrochloride 1%. Preservative: benzalkonium chloride 0.01%. Inactives: boric acid, edetate disodium, potassium chloride, sodium carbonate and/or hydrochloric acid (to adjust pH), purified water. The pH range is between 3.0 and 5.5.
Rx Only
USUAL DOSAGE: Instill one or two drops in the eye which can be repeated in five to ten minutes if necessary. Read enclosed Insert.
PRECAUTION: Do not touch dropper tip to any surface, as this may contaminate the solution.
STORAGE: Store at 8° to 25°C (46° to 77°F). After opening, Cyclogyl can be used until the expiration date on the bottle.
FOR TOPICAL OPHTHALMIC USE ONLY.
Alcon
ALCON LABORATORIES, INC.
Fort Worth, Texas 76134 USA
Product of Spain
Printed in the USA
300037979 -0520
SN:
LOT:
EXP.:
GTIN: 00300650396059
NDC 0065-0396-05
Alcon
Cyclogyl® 1%
(cyclopentolate hydrochloride ophthalmic solution, USP)
Sterile 5 mL
Rx Only
INGREDIENTS: Each mL contains : Active: cyclopentolate hydrochloride 1%. Preservative: benzalkonium chloride 0.01%. Inactives: boric acid, edetate disodium, potassium chloride, sodium carbonate and/or hydrochloric acid (to adjust pH), purified water. The pH range is between 3.0 and 5.5.
PRECAUTION: Do not touch dropper tip to any surface, as this may contaminate the solution.
USUAL DOSAGE: Instill one or two drops in the eye which can be repeated in five to ten minutes if necessary. Read enclosed insert.
STORAGE: Store at 8° to 25°C (46° to 77°F).
ALCON LABORATORIES, INC.
Fort Worth, Texas 76134 USA Printed in USA
LOT EXP.:
300037978-0520
NDC 0065-0397-15
Alcon
Cyclogyl® 2%
(cyclopentolate hydrochloride ophthalmic solution, USP)
15 mL Sterile
Rx Only
USUAL DOSAGE: Instill one or two drops in the eye which can be repeated in five to ten minutes if necessary. Read enclosed insert.
PRECAUTION: Do not touch dropper tip to any surface, as this may contaminate the solution.
STORAGE: Store at 8° to 25°C (46° to 77°F). After opening, Cyclogyl can be used until the expiration date on the bottle.
INGREDIENTS: Each mL contains : Active: cyclopentolate hydrochloride 2%. Preservative: benzalkonium chloride 0.01%. Inactives: boric acid, edetate disodium, sodium carbonate and/or hydrochloric acid (to adjust pH), purified water. The pH range is between 3.0 and 5.5.
FOR TOPICAL OPHTHALMIC USE ONLY.
Alcon
ALCON LABORATORIES, INC.
Fort Worth, Texas 76134 USA
Product of Spain
Printed in USA
300037977-0520
SN:
LOT:
EXP.:
GTIN: 00300650397155
NDC 0065-0397-15
Alcon
Cyclogyl® 2%
(cyclopentolate hydrochloride ophthalmic solution, USP)
Sterile 15 mL
Rx Only INGREDIENTS: Each mL contains : Active: cyclopentolate hydrochloride 2%. Preservative: benzalkonium chloride 0.01%. Inactives: boric acid, edetate disodium, sodium carbonate and/or hydrochloric acid (to adjust pH), purified water. The pH range is between 3.0 and 5.5.
PRECAUTION: Do not touch dropper tip to any surface, as this may contaminate the solution.
USUAL DOSAGE: Instill one or two drops in the eye which can be repeated in five to ten minutes if necessary. Read enclosed Insert.
STORAGE: Store at 8° to 25°C (46° to 77°F).
ALCON LABORATORIES, INC.
Fort Worth, Texas 76134 USA
Printed in USA
LOT: EXP.:
300037975-0520
-
INGREDIENTS AND APPEARANCE
CYCLOGYL
cyclopentolate hydrochloride solution/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0065-0395 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CYCLOPENTOLATE HYDROCHLORIDE (UNII: 736I6971TE) (CYCLOPENTOLATE - UNII:I76F4SHP7J) CYCLOPENTOLATE HYDROCHLORIDE 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) BORIC ACID (UNII: R57ZHV85D4) EDETATE DISODIUM (UNII: 7FLD91C86K) SODIUM CARBONATE (UNII: 45P3261C7T) HYDROCHLORIC ACID (UNII: QTT17582CB) WATER (UNII: 059QF0KO0R) POTASSIUM CHLORIDE (UNII: 660YQ98I10) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0065-0395-15 1 in 1 CARTON 10/15/1975 1 15 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA084109 10/15/1975 CYCLOGYL
cyclopentolate hydrochloride solution/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0065-0396 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CYCLOPENTOLATE HYDROCHLORIDE (UNII: 736I6971TE) (CYCLOPENTOLATE - UNII:I76F4SHP7J) CYCLOPENTOLATE HYDROCHLORIDE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) BORIC ACID (UNII: R57ZHV85D4) EDETATE DISODIUM (UNII: 7FLD91C86K) SODIUM CARBONATE (UNII: 45P3261C7T) HYDROCHLORIC ACID (UNII: QTT17582CB) WATER (UNII: 059QF0KO0R) POTASSIUM CHLORIDE (UNII: 660YQ98I10) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0065-0396-02 1 in 1 CARTON 10/15/1975 1 2 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC:0065-0396-05 1 in 1 CARTON 10/15/1975 2 5 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 3 NDC:0065-0396-15 1 in 1 CARTON 10/15/1975 07/31/2019 3 15 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA084110 10/15/1975 CYCLOGYL
cyclopentolate hydrochloride solution/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0065-0397 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CYCLOPENTOLATE HYDROCHLORIDE (UNII: 736I6971TE) (CYCLOPENTOLATE - UNII:I76F4SHP7J) CYCLOPENTOLATE HYDROCHLORIDE 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) BORIC ACID (UNII: R57ZHV85D4) EDETATE DISODIUM (UNII: 7FLD91C86K) SODIUM CARBONATE (UNII: 45P3261C7T) HYDROCHLORIC ACID (UNII: QTT17582CB) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0065-0397-02 1 in 1 CARTON 10/15/1975 1 2 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC:0065-0397-05 1 in 1 CARTON 10/15/1975 2 5 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 3 NDC:0065-0397-15 1 in 1 CARTON 10/15/1975 3 15 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA084108 10/15/1975 Labeler - Alcon Laboratories, Inc. (008018525) Establishment Name Address ID/FEI Business Operations Alcon Research LLC 007672236 manufacture(0065-0395, 0065-0396, 0065-0397)







