Label: GZE ROSEMARY HAIR CARESET- rosemary oil kit
-
NDC Code(s):
83566-016-01,
83566-017-01,
83566-018-01,
83566-019-01, view more83566-020-01
- Packager: Guangzhou Yilong Cosmetics Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 28, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
GZE Rosemary Shampoo Bar:
BITTER ORANGE OIL
POTASSIUM SORBATE
ORYZA SATIVA WHOLE
COCAMIDOPROPYL BETAINE
EUCALYPTUS GLOBULUS LEAF
CITRIC ACID ACETATE
CYMBOPOGON SCHOENANTHUS OIL
LIME OIL
SODIUM BENZOATE
WATERGZE Rosemary Hair Oil:
COCODIMONIUM HYDROXYPROPYL HYDROLYZED KERATIN (1000 MW)
ASCORBIC ACID
RETINYL PROPIONATE
ASCORBYL PALMITATE
PANTHENOL
TOCOPHEROL
WATER
MENTHA PIPERITA LEAF
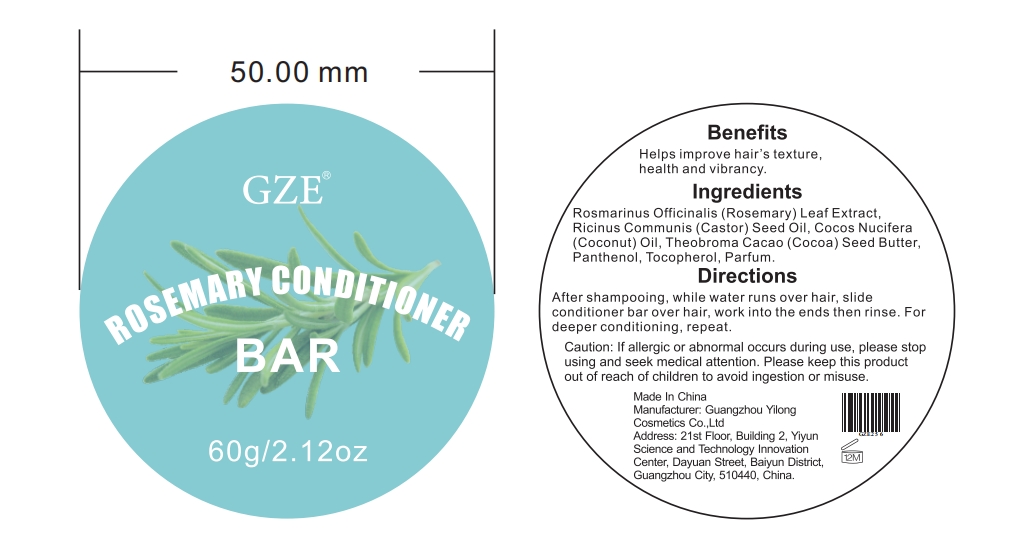
METHICONE (20 CST)GZE Rosemary Conditioner Bar:
COCONUT OIL
COCOABUTTER
PANTHENOL
TOCOPHEROL
PARFUMIDINEGZE Black Sesame Rosemary Keratin Hair Mask:
JOJOBA OIL
HAIR KERATIN AMINO ACIDS
PANTHENOL
ASCORBYL TOCOPHERYL MALEATE
ASCORBIC ACID
FRAGRANCE 13576
DIMETHICONE
HYDROLYZED JOJOBA ESTERS (ACID FORM)
ETHYLHEXYLGLYCERIN
CETEARYL STEARATE
WATER - PURPOSE
-
INDICATIONS & USAGE
Step1: Wet your hair with warm water.
Step2: Take an appropriate amount of product and whip it into foam,apply it evenly onyour hair, and rub it to clean your scalp.
Step3: Add 10-12 drops of rosemary essential oil to shampoo to enhance hair care effect.
Step4: Clean the foam with warm water.Wash again with hair conditioner soap.Let your hair dry and apply a hair mask.
- WARNINGS
- STOP USE
- DO NOT USE
- WHEN USING
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Step1: Wet your hair with warm water.
Step2: Take an appropriate amount of product and whip it into foam,apply it evenly onyour hair, and rub it to clean your scalp.
Step3: Add 10-12 drops of rosemary essential oil to shampoo to enhance hair care effect.
Step4: Clean the foam with warm water.Wash again with hair conditioner soap.Let your hair dry and apply a hair mask.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GZE ROSEMARY HAIR CARESET
rosemary oil kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83566-016 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83566-016-01 1 in 1 KIT; Type 1: Convenience Kit of Co-Package 06/28/2024 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE, DROPPER 30 mL in 330 Part 2 1 BAG 60 g in 330 Part 3 1 CAN 100 g in 330 Part 4 1 BAG 60 g in 330 Part 1 of 4 GZE ROSEMARY HAIR OIL
rosemary oil oilProduct Information Item Code (Source) NDC:83566-020 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ROSEMARY OIL (UNII: 8LGU7VM393) (ROSEMARY OIL - UNII:8LGU7VM393) ROSEMARY OIL 46 g in 100 mL Inactive Ingredients Ingredient Name Strength ASCORBYL PALMITATE (UNII: QN83US2B0N) CYCLOMETHICONE 4 (UNII: CZ227117JE) HYDROLYZED JOJOBA ESTERS (ACID FORM) (UNII: UDR641JW8W) ASCORBIC ACID (UNII: PQ6CK8PD0R) MENTHA PIPERITA LEAF (UNII: A389O33LX6) RETINYL RETINOATE (UNII: 5AT5X9J439) TOCOPHEROL (UNII: R0ZB2556P8) WATER (UNII: 059QF0KO0R) PANTHENOL (UNII: WV9CM0O67Z) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83566-020-01 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M032 Part 2 of 4 GZE ROSEMARY CONDITIONER BAR
rosemary oil soapProduct Information Item Code (Source) NDC:83566-018 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ROSEMARY OIL (UNII: 8LGU7VM393) (ROSEMARY OIL - UNII:8LGU7VM393) ROSEMARY OIL 46 g in 100 g Inactive Ingredients Ingredient Name Strength COCONUT OIL (UNII: Q9L0O73W7L) COCOA BUTTER (UNII: 512OYT1CRR) PANTHENOL (UNII: WV9CM0O67Z) PARFUMIDINE (UNII: A2Q2LHC6MA) TOCOPHEROL (UNII: R0ZB2556P8) RICINUS COMMUNIS SEED (UNII: 7EK4SFN1TX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83566-018-01 60 g in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) Part 3 of 4 GZE BLACK SESAME ROSEMARY KERATIN HAIRMASK
rosemary creamProduct Information Item Code (Source) NDC:83566-019 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ROSEMARY OIL (UNII: 8LGU7VM393) (ROSEMARY OIL - UNII:8LGU7VM393) ROSEMARY OIL 5 g in 100 g Inactive Ingredients Ingredient Name Strength ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) ASCORBYL TOCOPHERYL MALEATE (UNII: D2G6259XR5) FRAGRANCE 13576 (UNII: 5EM498GW35) ASCORBIC ACID (UNII: PQ6CK8PD0R) PANTHENOL (UNII: WV9CM0O67Z) CETEARYL BEHENATE (UNII: 7ARI9LTH0U) SESAME SEED (UNII: 7Y1255HVXR) CETYL DIMETHICONE 150 (UNII: 5L694Y0T22) HYDROLYZED JOJOBA ESTERS (ACID FORM) (UNII: UDR641JW8W) HAIR KERATIN AMINO ACIDS (UNII: G46579QK1M) SIMMONDSIA CHINENSIS SEED (UNII: D24K2Q1F6H) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83566-019-01 100 g in 1 CAN; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) Part 4 of 4 GZE ROSEMARY SHAMPOOBAR
rosemary oil soapProduct Information Item Code (Source) NDC:83566-017 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ROSEMARY OIL (UNII: 8LGU7VM393) (ROSEMARY OIL - UNII:8LGU7VM393) ROSEMARY OIL 5 g in 100 g Inactive Ingredients Ingredient Name Strength CITRUS AURANTIFOLIA SEED OIL (UNII: EW089HAI77) CYMBOPOGON SCHOENANTHUS OIL (UNII: XE7K568ILO) BITTER ORANGE OIL (UNII: 9TLV70SV6I) WATER (UNII: 059QF0KO0R) CITRIC ACID ACETATE (UNII: DSO12WL7AU) ORYZA SATIVA WHOLE (UNII: 84IVV0906Z) EUCALYPTUS GLOBULUS LEAF (UNII: S546YLW6E6) SODIUM BENZOATE (UNII: OJ245FE5EU) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83566-017-01 60 g in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M032 07/04/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M032 06/28/2024 Labeler - Guangzhou Yilong Cosmetics Co., Ltd (712647107) Establishment Name Address ID/FEI Business Operations Guangzhou Yilong Cosmetics Co., Ltd 712647107 manufacture(83566-016)