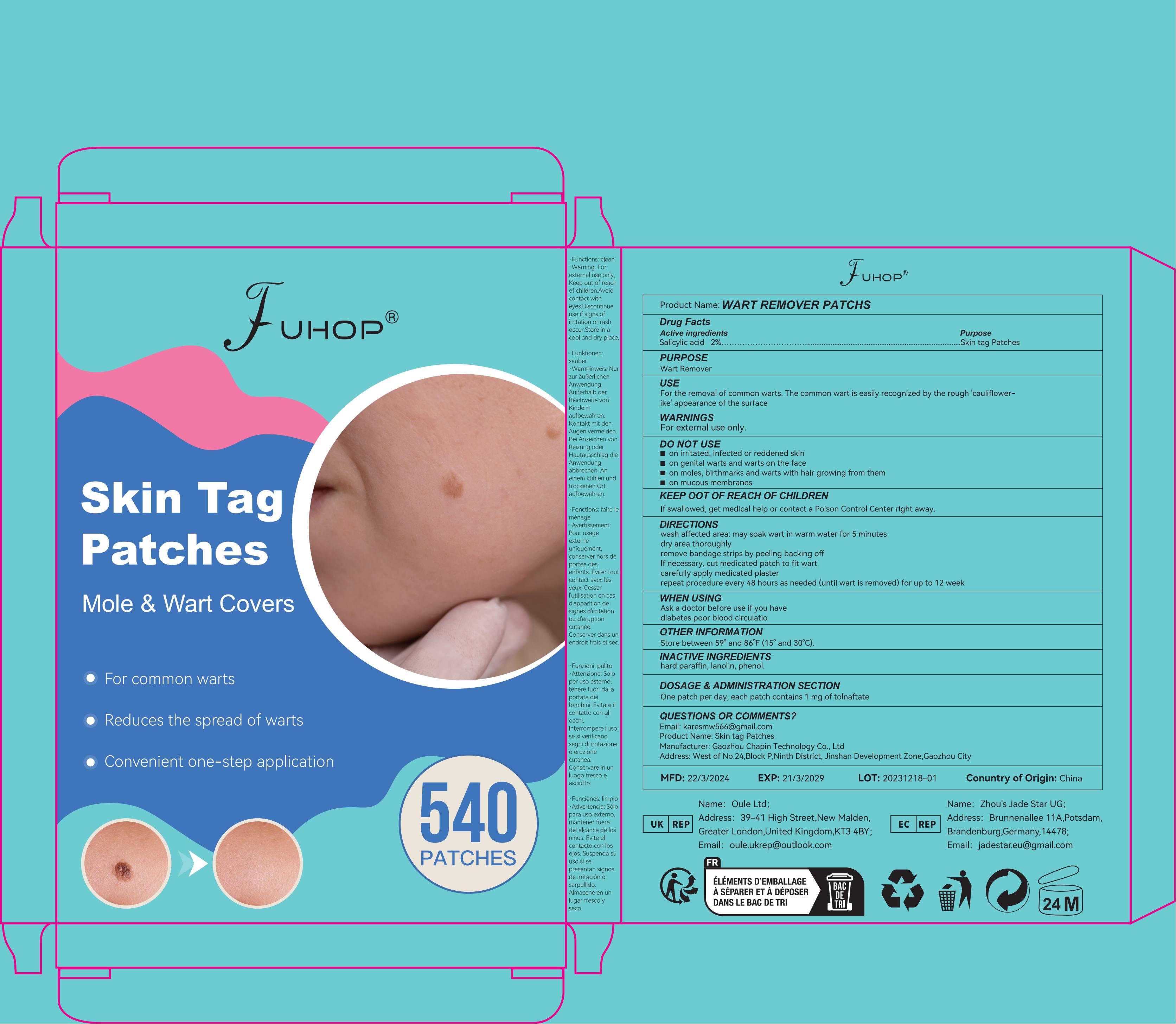
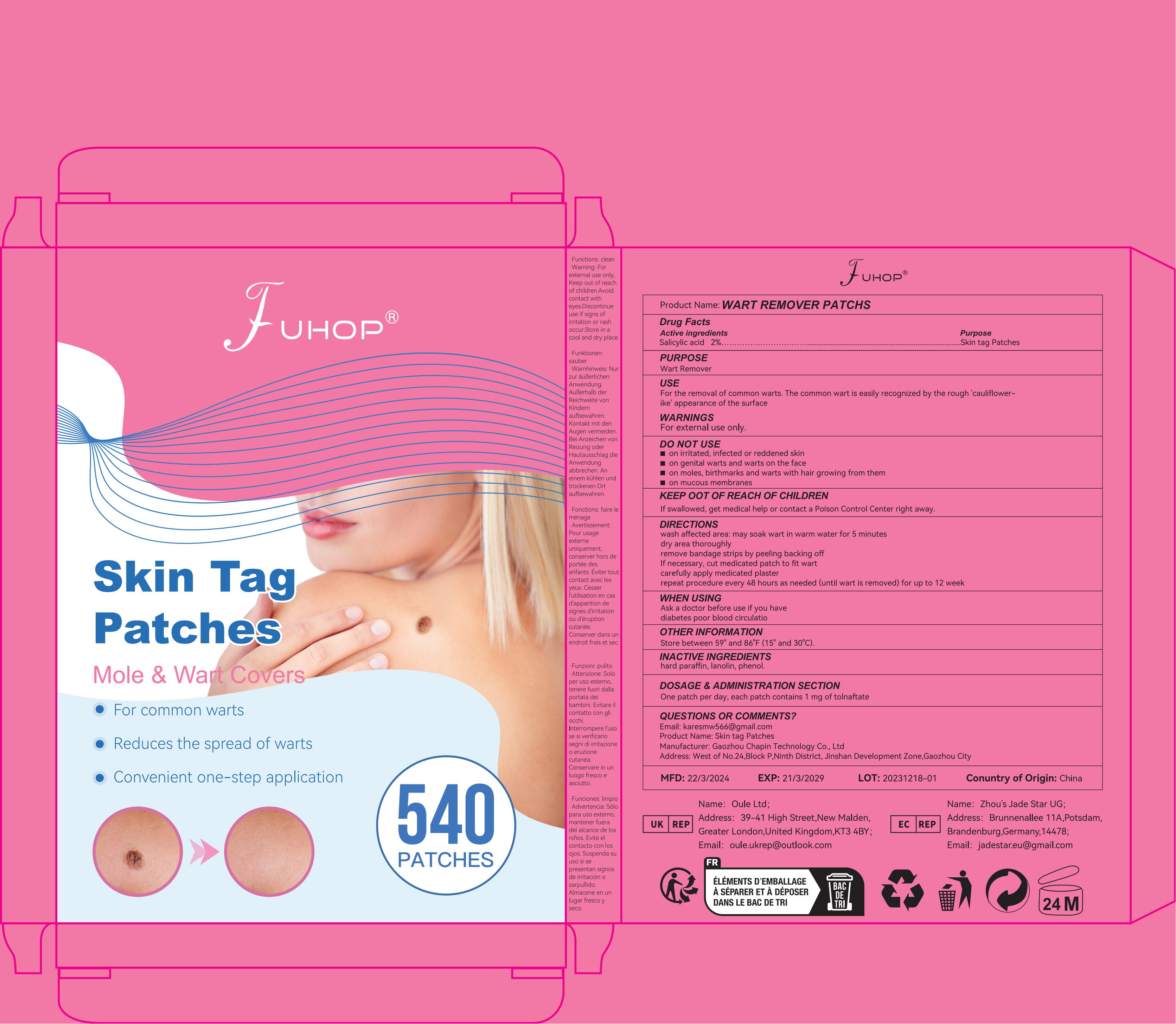
Label: WART REMOVER PATCHS- wart remover patch
- NDC Code(s): 84168-015-11
- Packager: Gaozhou Chapin Technology Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 25, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- USE
- WARNINGS
- DO NOT USE
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- WHEN USING
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- DOSAGE & ADMINISTRATION SECTION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
WART REMOVER PATCHS
wart remover patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84168-015 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 200 mg in 1 g Inactive Ingredients Ingredient Name Strength ETHYLHEXYL ACRYLATE (UNII: HR49R9S6XG) 380 mg in 1 g POLYETHYLENE TEREPHTHALATE (INTRINSIC VISCOSITY 0.40-0.70) (UNII: Y78AJD3DED) 420 mg in 1 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84168-015-11 16.8 g in 1 BOX; Type 0: Not a Combination Product 05/09/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M028 05/09/2024 Labeler - Gaozhou Chapin Technology Co., Ltd (715573719) Establishment Name Address ID/FEI Business Operations Gaozhou Chapin Technology Co., Ltd 715573719 manufacture(84168-015)