Label: NIGHTTIME SLEEP AID- diphenhydramine hcl solution
- NDC Code(s): 37835-319-30
- Packager: Bi-Mart
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 2, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each 30 mL dose cup)
- Purpose
- Uses
-
Warnings
Do not use
- for children under 12 years of age
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
-
Directions
- do not take more than directed
- mL = milliliter; FL OZ = fluid ounce
- use only enclosed dosing cup designed for use with this product. Do not use any other dosing device.
- take only one dose per day (24 hours)
- adults and children 12 years and over: take 30 mL at bedtime if needed or as directed by a doctor
- children under 12 years: do not use
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
BI-MART
NDC 37835-319-30
Compare to the active ingredient in
Vicks® ZzzQuil® NIGHTTIME SLEEP-AID*Nighttime
Sleep-Aid
Diphenhydramine HCl 50 mg
Nighttime Sleep-Aid• Non-Habit Forming
• Alcohol FreeNot for treating Cold or Flu
Berry Flavor
12 FL OZ (355 mL)
TAMPER EVIDENT: DO NOT USE IF PRINTED NECK
WRAP IS BROKEN OR MISSING*This product is not manufactured or distributed by The Procter &
Gamble Company, owner of the registered trademark Vicks® ZzzQuil®
Nighttime Sleep-Aid. 50844 ORG112000202BI-MART UNCONDITIONAL MONEY BACK GUARANTEE
If you are not completely satisfied with your BI-MART
QUALITY PRODUCT, regardless of the reason, return
the unused portion and your entire purchase price will
be cheerfully refunded.Distributed by:
BI-MART
Eugene, OR 97402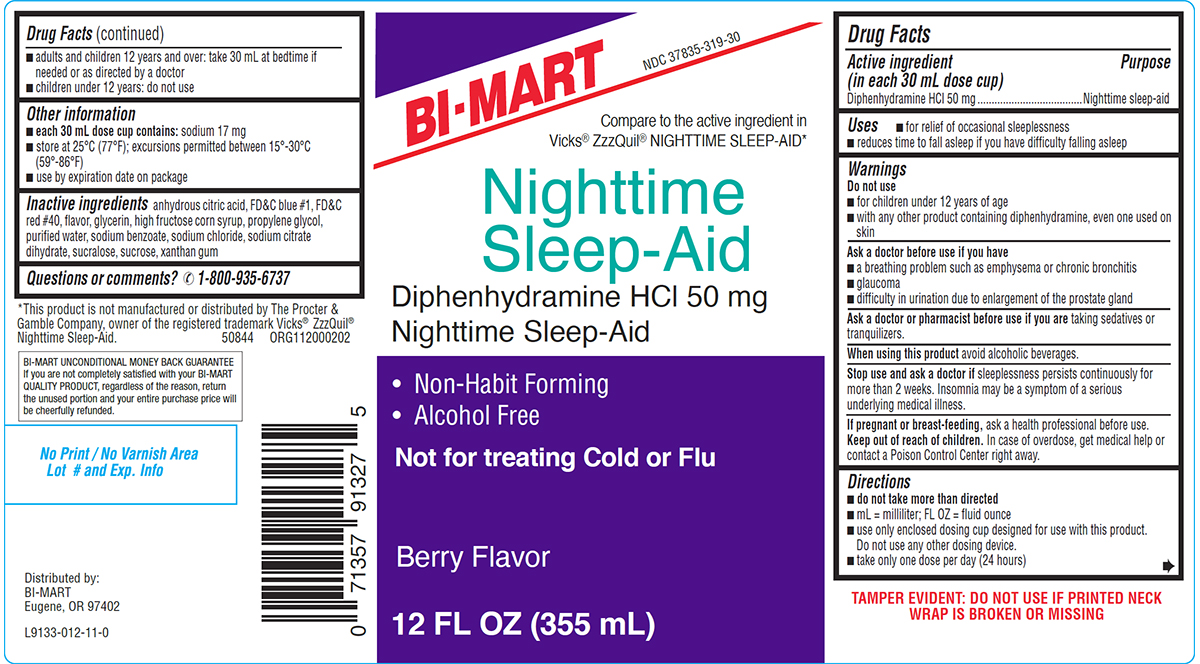
Bi-Mart 44-002A
-
INGREDIENTS AND APPEARANCE
NIGHTTIME SLEEP AID
diphenhydramine hcl solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:37835-319 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 50 mg in 30 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) HIGH FRUCTOSE CORN SYRUP (UNII: XY6UN3QB6S) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CHLORIDE (UNII: 451W47IQ8X) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) SUCRALOSE (UNII: 96K6UQ3ZD4) SUCROSE (UNII: C151H8M554) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color purple Score Shape Size Flavor BERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:37835-319-30 355 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/29/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M010 04/29/2021 Labeler - Bi-Mart (027630078) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 manufacture(37835-319) , pack(37835-319)

