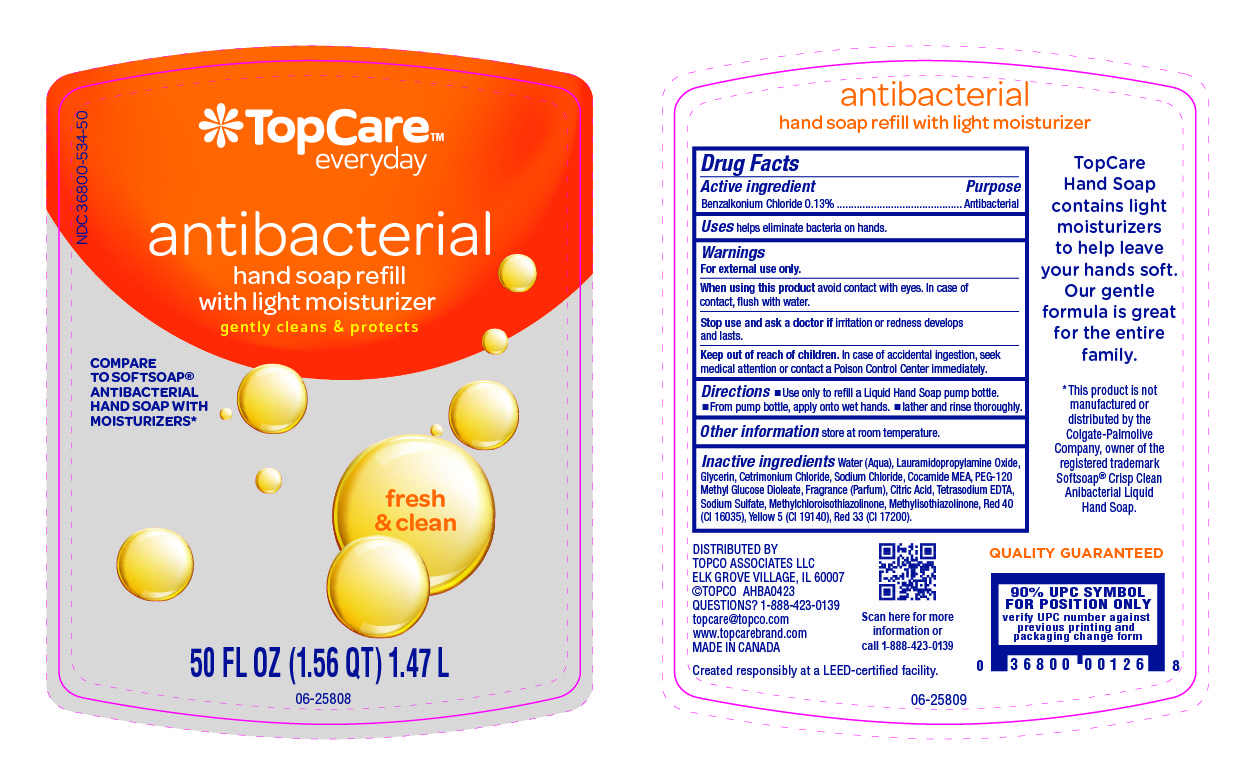
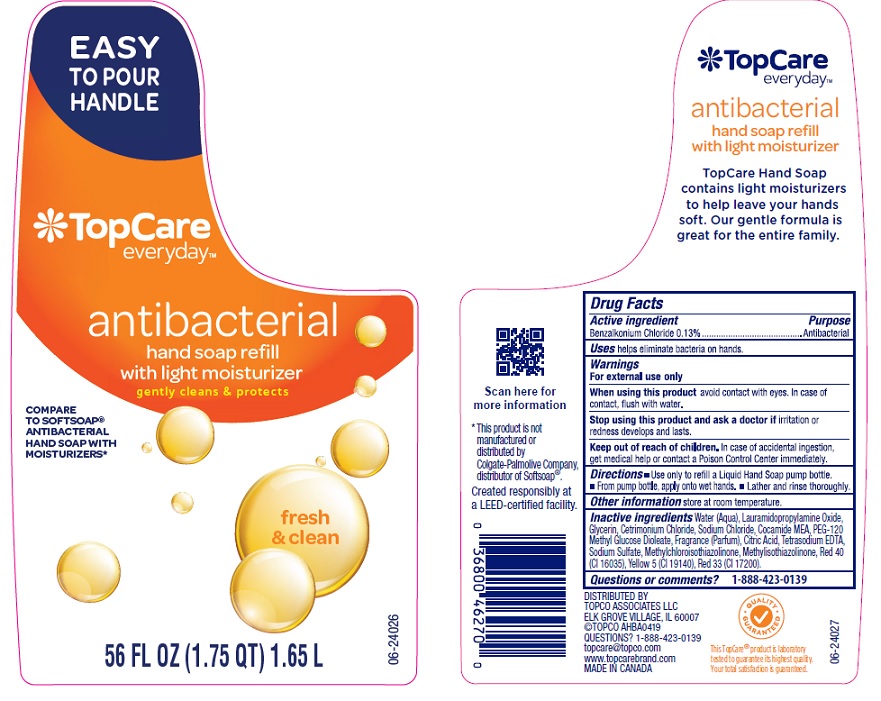
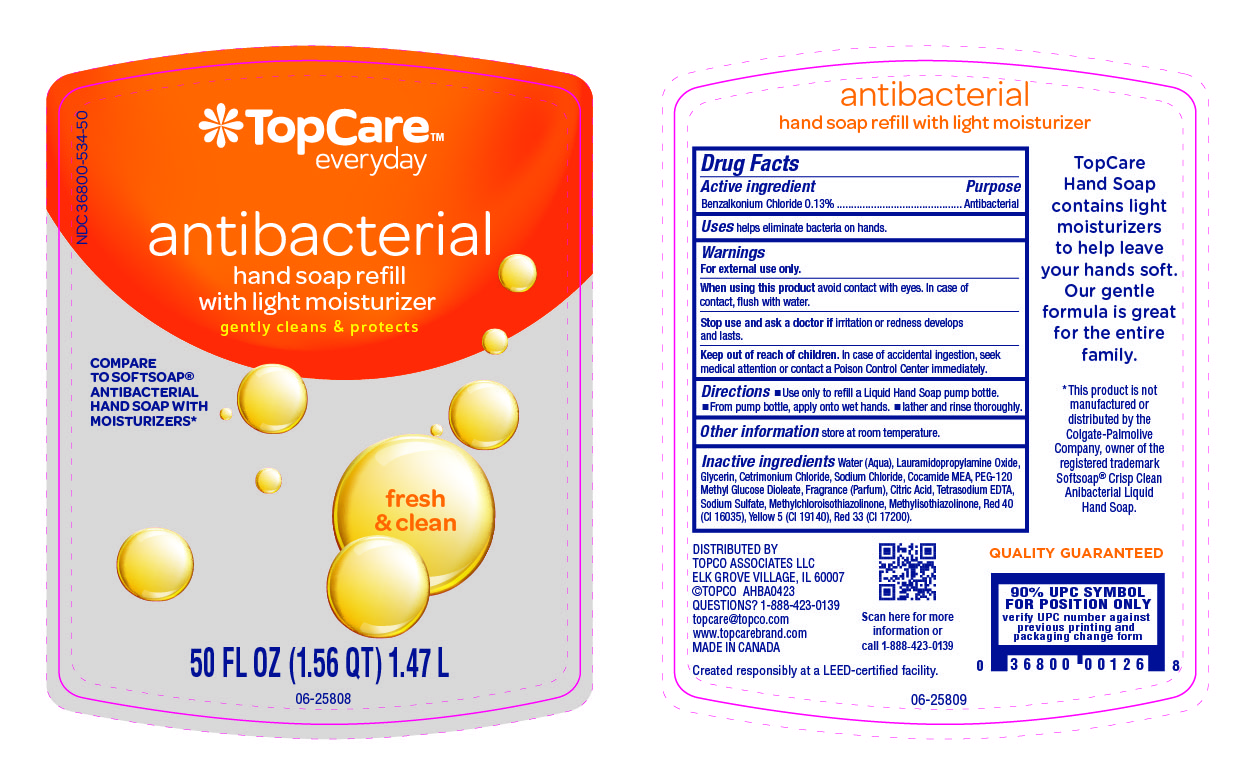
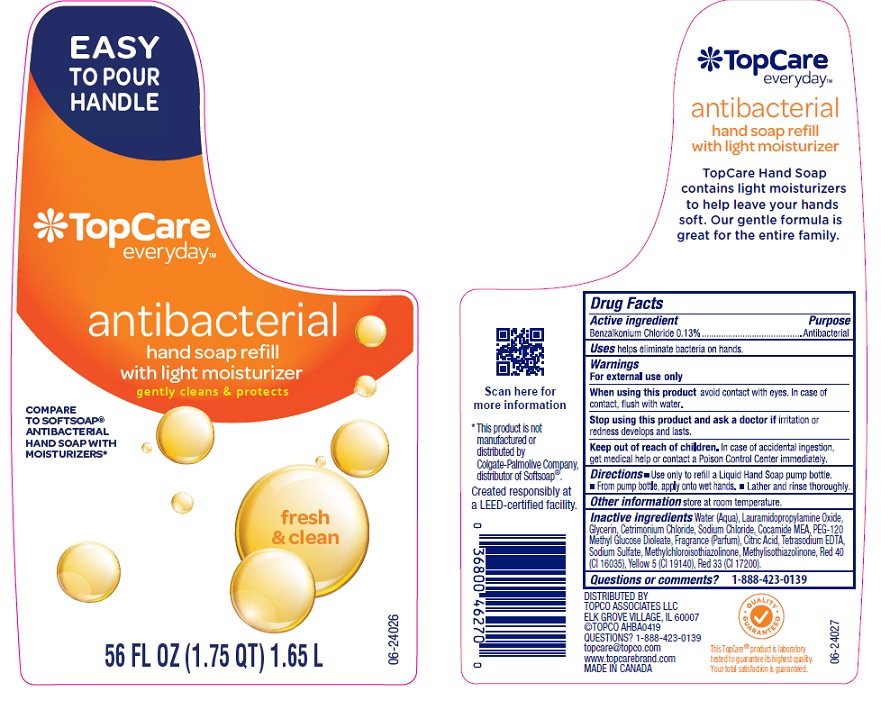
Label: TOPCARE ANTIBACTERIAL HAND FRESH AND CLEAN- benzalkonium chloride liquid
- NDC Code(s): 36800-534-50, 36800-534-56
- Packager: Topco Associates LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 9, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Uses
- Warnings
- Directions
- Other information
-
Inactive ingredients
Water (Aqua), Lauramidopropylamine Oxide, Glycerin, Cetrimonium Chloride, Sodium Chloride, Cocamide MEA, PEG-120 Methyl Glucose Dioleate, Fragrance (Parfum), Citric Acid, Tetrasodium EDTA, Sodium Sulfate, Methylchloroisothiazolinone, Methylisothiazolinone, Red 40 (CI 16035), Yellow 5 (CI 19140), Red 33 (CI 17200).
- Questions or comments?
- Principal Display Panel- 50 Oz
- Principal Display Panel- 56 Oz
-
INGREDIENTS AND APPEARANCE
TOPCARE ANTIBACTERIAL HAND FRESH AND CLEAN
benzalkonium chloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:36800-534 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1.3 mg in 1 mL Inactive Ingredients Ingredient Name Strength LAURAMIDOPROPYLAMINE OXIDE (UNII: I6KX160QTV) GLYCERIN (UNII: PDC6A3C0OX) CETRIMONIUM CHLORIDE (UNII: UC9PE95IBP) SODIUM CHLORIDE (UNII: 451W47IQ8X) COCO MONOETHANOLAMIDE (UNII: C80684146D) PEG-120 METHYL GLUCOSE DIOLEATE (UNII: YM0K64F20V) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) FRAGRANCE CLEAN ORC0600327 (UNII: 329LCV5BTF) EDETATE SODIUM (UNII: MP1J8420LU) SODIUM SULFATE (UNII: 0YPR65R21J) METHYLCHLOROISOTHIAZOLINONE (UNII: DEL7T5QRPN) METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) WATER (UNII: 059QF0KO0R) D&C RED NO. 33 (UNII: 9DBA0SBB0L) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:36800-534-56 1656 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/20/2019 2 NDC:36800-534-50 1470 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 06/20/2019 Labeler - Topco Associates LLC (006935977) Registrant - Apollo Health and Beauty Care Inc. (201901209) Establishment Name Address ID/FEI Business Operations Apollo Health and Beauty Care Inc. 201901209 manufacture(36800-534)