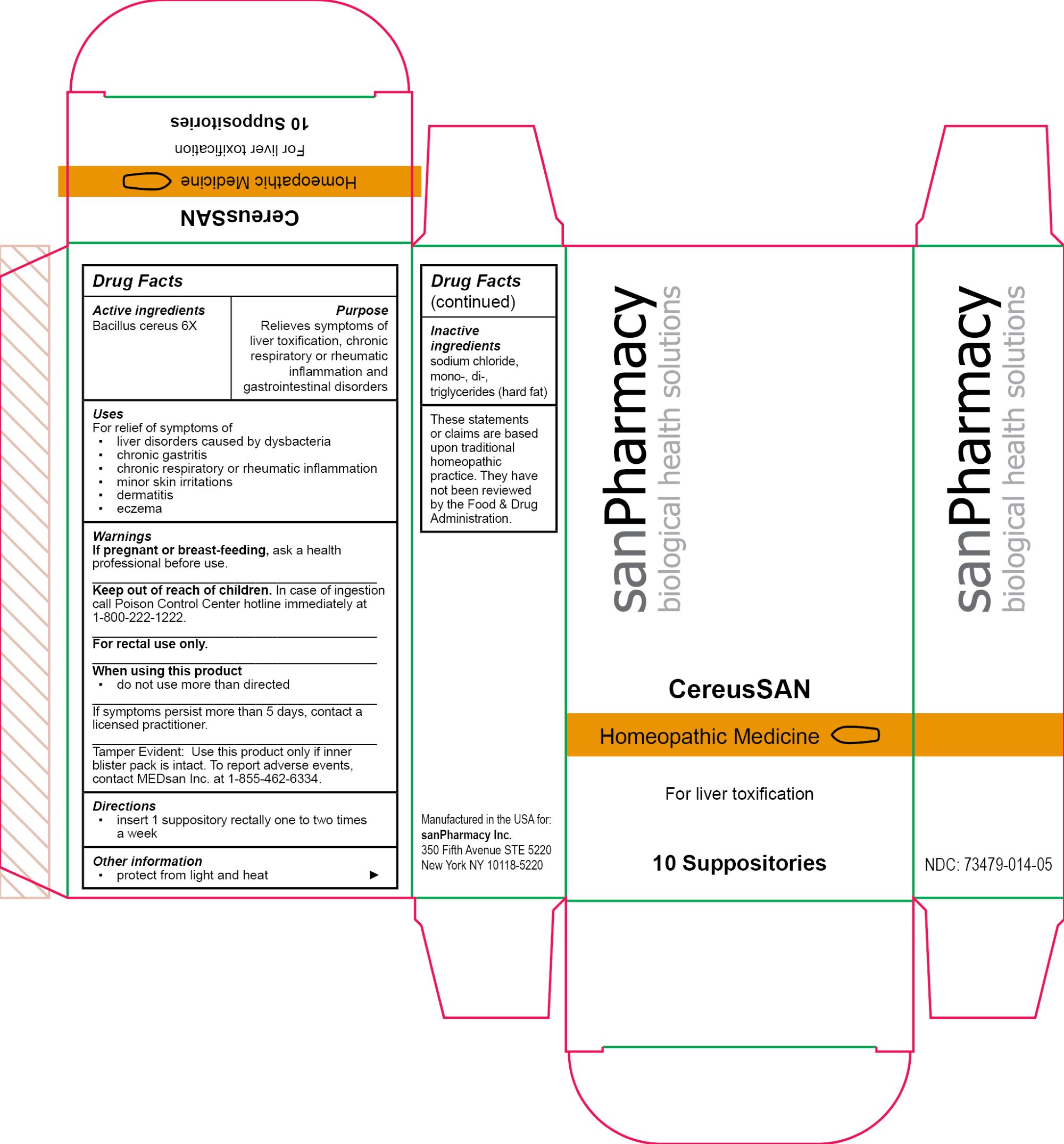
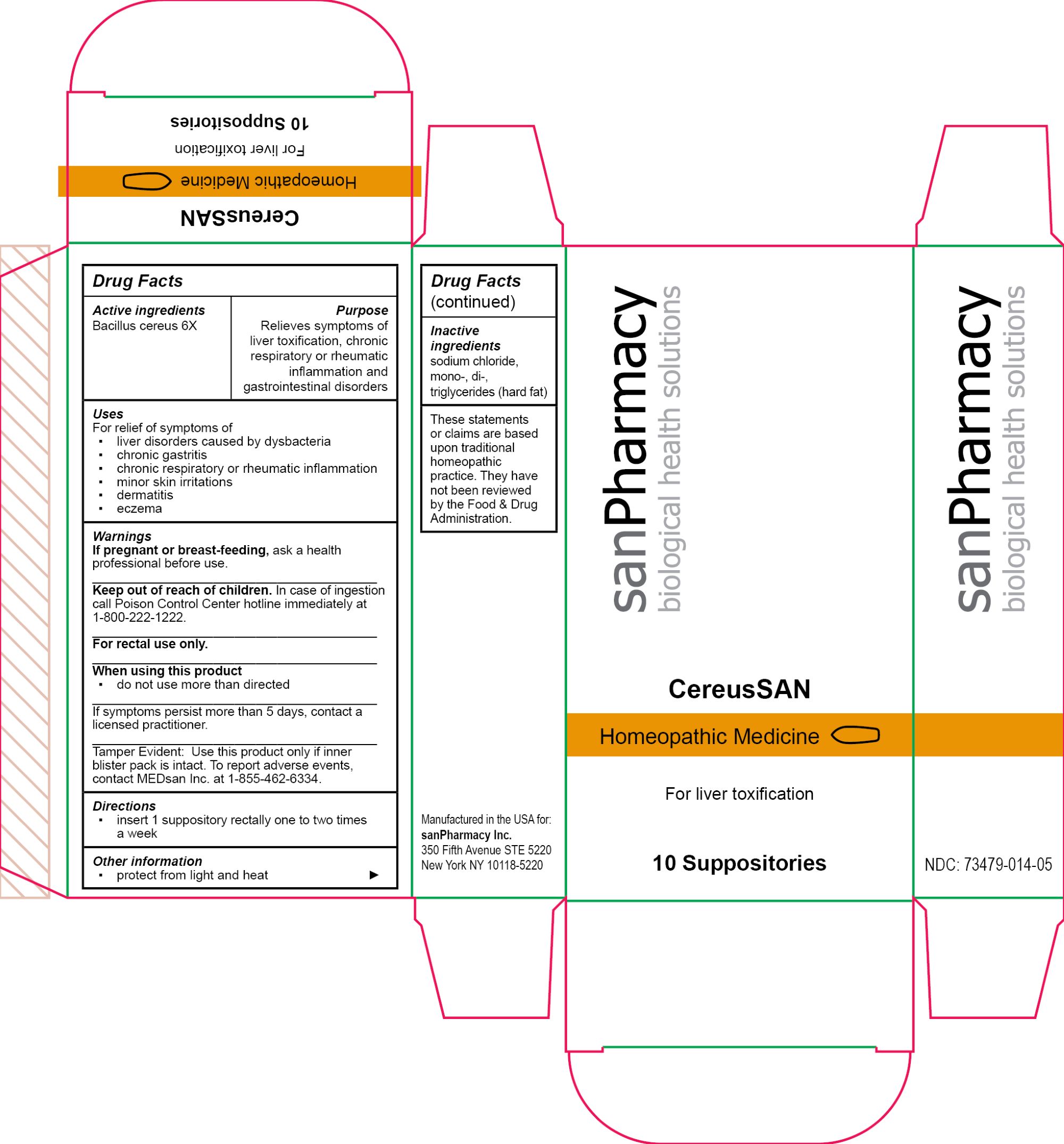
Label: CEREUSSAN- bacillus cereus suppository
- NDC Code(s): 73479-014-05
- Packager: sanPharmacy Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated April 25, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- SPL UNCLASSIFIED SECTION
- Liability statement
-
WARNINGS
If pregnant or breast-feeding, ask a health professional before use.
______________________________
Keep out of reach of children. In case of ingestion call Poison Control Center hotline immediately at 1-800-222-1222.
______________________________
For rectal use only.
______________________________
When using this product
▪ do not use more than directed
______________________________
If symptoms persist more than 5 days, contact a licensed practitioner.
______________________________
Tamper Evident: Use this product only if inner blister pack is intact. To report adverse events, contact MEDsan Inc. at 1-855-462-6334. - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CEREUSSAN
bacillus cereus suppositoryProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73479-014 Route of Administration RECTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACILLUS CEREUS (UNII: 7YC151N82L) (BACILLUS CEREUS - UNII:7YC151N82L) BACILLUS CEREUS 6 [hp_X] Inactive Ingredients Ingredient Name Strength HARD FAT (UNII: 8334LX7S21) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73479-014-05 1 in 1 CARTON; Type 0: Not a Combination Product 04/25/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 04/25/2024 Labeler - sanPharmacy Inc. (117235152) Establishment Name Address ID/FEI Business Operations MEDsan Inc. 004326784 manufacture(73479-014)