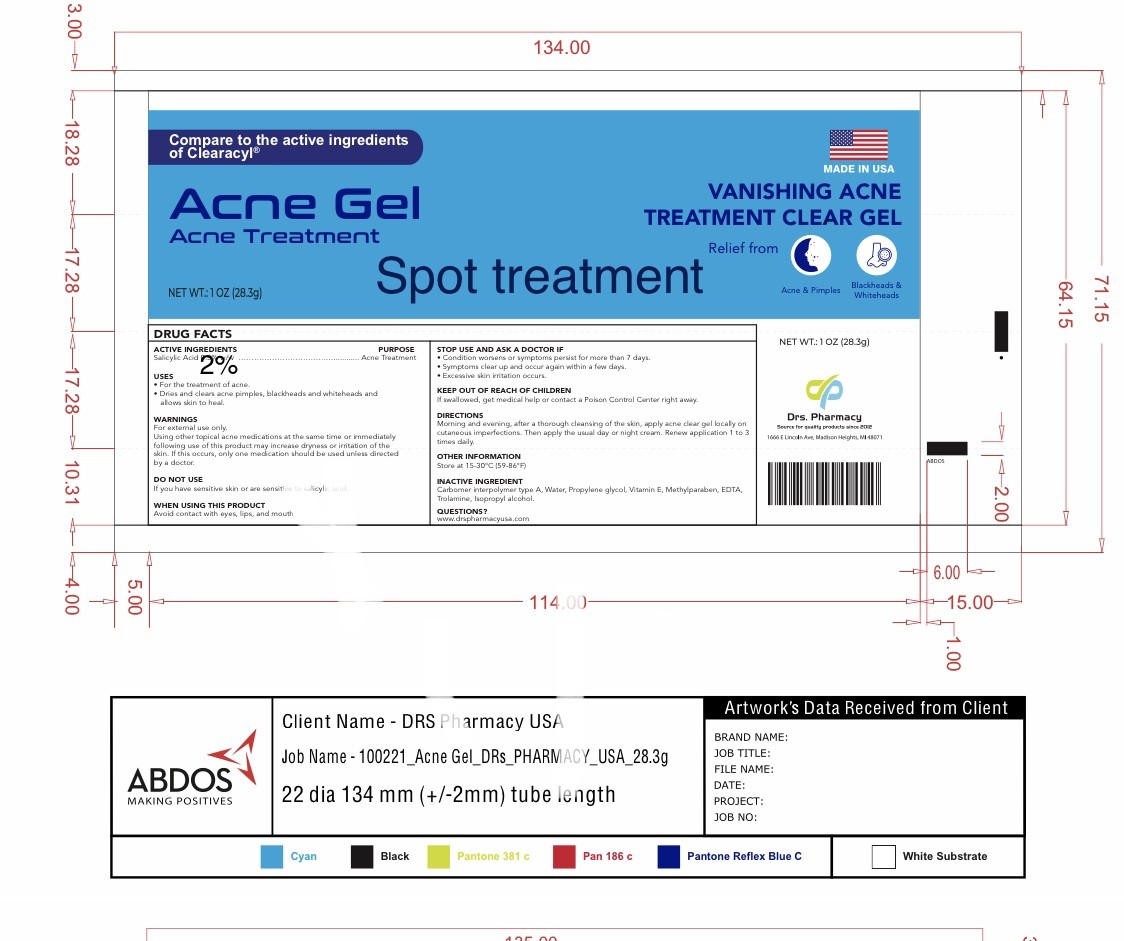
Label: DRS. ACNE SPOT TREATMENT- salicylic acid gel
- NDC Code(s): 80489-357-01, 80489-357-02
- Packager: OL PHARMA TECH, LLC Drs. PHARMACY
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 15, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- USES
- DO NOT USE
- DIRECTIONS
-
WARNINGS
For external use only. Using other topical acne medications at the same time or immediately following use of this product may increase dryness or irritation of the skin. If this occurs, only one medication should be used unless directed by a doctor. Avoid direct contact with the eyes. If product gets into the eyes, rinse liberally with water. Discontinue use if skin irritation develops or increases. If irritation persists, consult a doctor.
- WHEN USING THIS PRODUCT
- KEEP OUT OF REACH OF CHILDREN
- INACTIVE INGREDIENT
- OTHER INFORMATION
- QUESTIONS
- PACKAGE LABEL
-
INGREDIENTS AND APPEARANCE
DRS. ACNE SPOT TREATMENT
salicylic acid gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80489-357 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 20 mg in 1 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) METHYLPARABEN (UNII: A2I8C7HI9T) EDETATE DISODIUM (UNII: 7FLD91C86K) TROLAMINE (UNII: 9O3K93S3TK) ISOPROPYL ALCOHOL (UNII: ND2M416302) CARBOMER INTERPOLYMER TYPE A (55000 CPS) (UNII: 59TL3WG5CO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80489-357-01 1 in 1 CARTON 04/13/2024 1 20 g in 1 TUBE, WITH APPLICATOR; Type 0: Not a Combination Product 2 NDC:80489-357-02 1 in 1 CARTON 04/13/2024 2 30 g in 1 TUBE, WITH APPLICATOR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 02/01/2022 Labeler - OL PHARMA TECH, LLC Drs. PHARMACY (021170377) Registrant - OL PHARMA TECH, LLC Drs PHARMACY (021170377) Establishment Name Address ID/FEI Business Operations OL PHARMA TECH, LLC Drs PHARMACY 021170377 manufacture(80489-357)