Label: NUPHARMISTO TATTOO NUMBING CREAM 80G- lidocaine cream
- NDC Code(s): 71331-116-04
- Packager: Orange Lab, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 1, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
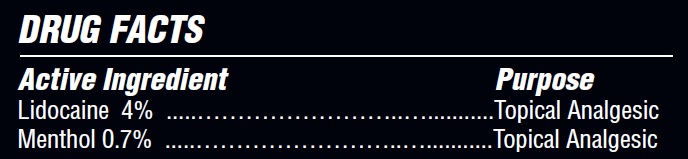
- Active Ingredient
- ask doctor
- Warning do not use if
- Keep out of reach of children
- if pregnant or breast feeding
- Purpose
- questions
- Stop use warnings
- When using
- Directions
- inactive Ingredients
- warnings sections
- How to use it correctly
- Label
- bottle label
-
INGREDIENTS AND APPEARANCE
NUPHARMISTO TATTOO NUMBING CREAM 80G
lidocaine creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71331-116 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 0.7 g in 100 g LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 4 g in 100 g Inactive Ingredients Ingredient Name Strength ACRYLATES/C10-30 ALKYL ACRYLATE CROSSPOLYMER (60000 MPA.S) (UNII: 8Z5ZAL5H3V) ETHYLHEXYL PALMITATE (UNII: 2865993309) HYDROGENATED POLYDECENE (1500 CST) (UNII: 4YI0729529) SODIUM POLYACRYLATE (8000 MW) (UNII: 285CYO341L) ALLANTOIN (UNII: 344S277G0Z) TRIDECETH-6 (UNII: 3T5PCR2H0C) EMU OIL (UNII: 344821WD61) PHENOXYETHANOL (UNII: HIE492ZZ3T) EDETATE SODIUM TETRAHYDRATE (UNII: L13NHD21X6) DIMETHYL ISOSORBIDE (UNII: SA6A6V432S) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SUNFLOWER OIL (UNII: 3W1JG795YI) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) WATER (UNII: 059QF0KO0R) ARNICA MONTANA FLOWER (UNII: OZ0E5Y15PZ) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71331-116-04 80 g in 1 PACKAGE; Type 0: Not a Combination Product 05/01/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 05/01/2024 Labeler - Orange Lab, Inc (004862271)