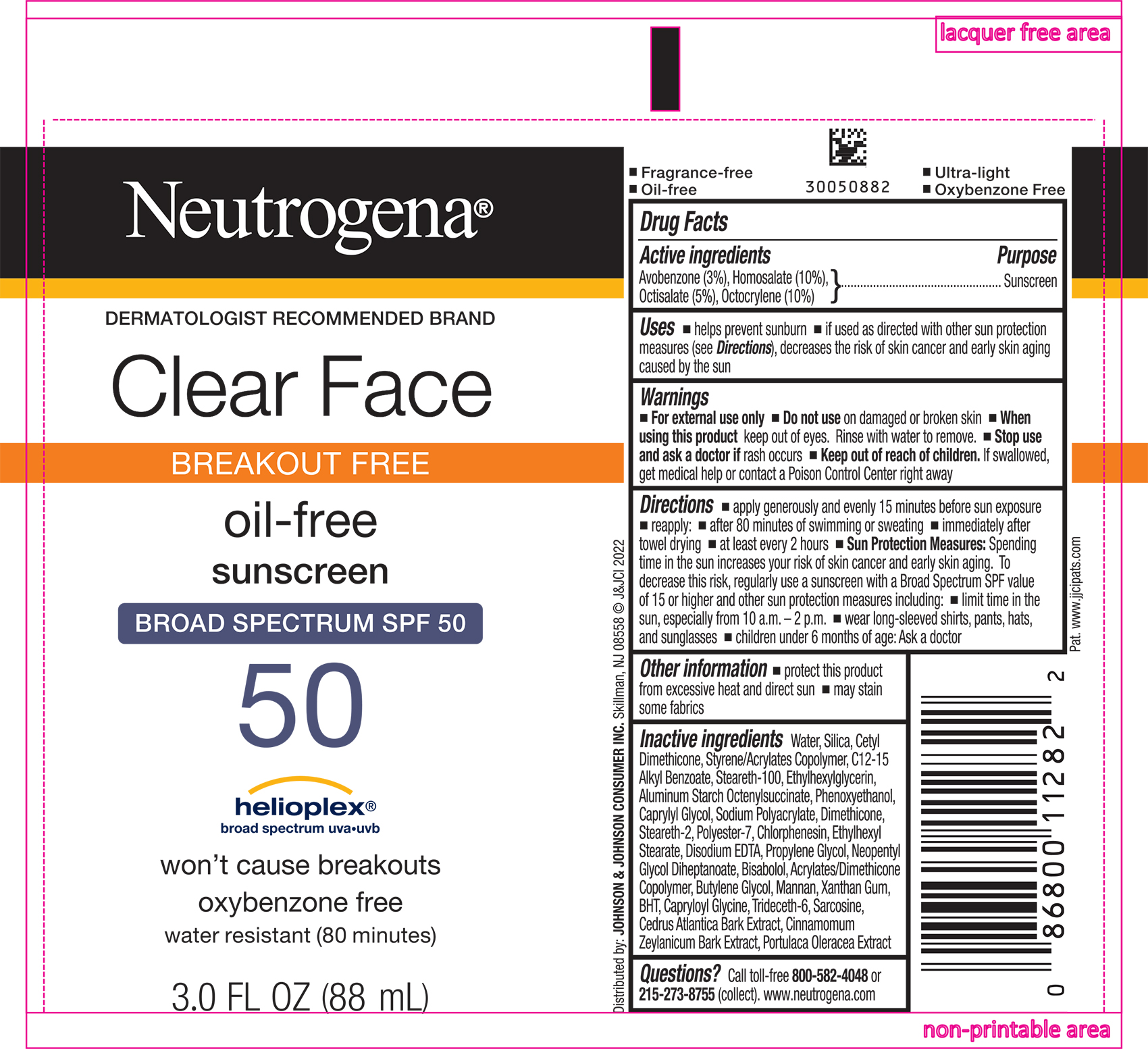
Label: NEUTROGENA CLEAR FACE BREAKOUT FREE OIL FREE SUNSCREEN BROAD SPECTRUM SPF 50- avobenzone, homosalate, octisalate, and octocrylene lotion
- NDC Code(s): 69968-0662-2, 69968-0662-3
- Packager: Kenvue Brands LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 12, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
- Warnings
-
Directions
- apply generously and evenly 15 minutes before sun exposure
- reapply:
- after 80 minutes of swimming or sweating
- immediately after towel drying
- at least every 2 hours
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
- children under 6 months of age: Ask a doctor
- Other Information
-
Inactive Ingredients
Water, Silica, Cetyl Dimethicone, Styrene/Acrylates Copolymer, C12-15 Alkyl Benzoate, Steareth-100, Ethylhexylglycerin, Aluminum Starch Octenylsuccinate, Phenoxyethanol, Caprylyl Glycol, Sodium Polyacrylate, Dimethicone, Steareth-2, Polyester-7, Chlorphenesin, Ethylhexyl Stearate, Disodium EDTA, Propylene Glycol, Neopentyl Glycol Diheptanoate, Bisabolol, Acrylates/Dimethicone Copolymer, Butylene Glycol, Mannan, Xanthan Gum, BHT, Capryloyl Glycine, Trideceth-6, Sarcosine, Cedrus Atlantica Bark Extract, Cinnamomum Zeylanicum Bark Extract, Portulaca Oleracea Extract
- Questions?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 88 mL Tube Label
-
INGREDIENTS AND APPEARANCE
NEUTROGENA CLEAR FACE BREAKOUT FREE OIL FREE SUNSCREEN BROAD SPECTRUM SPF 50
avobenzone, homosalate, octisalate, and octocrylene lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69968-0662 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 100 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 100 mg in 1 mL Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CETYL DIMETHICONE 25 (UNII: U4AS1BW4ZB) BUTYL METHACRYLATE/METHYL METHACRYLATE/METHACRYLIC ACID/STYRENE CROSSPOLYMER (UNII: V5RS026Q0H) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) WATER (UNII: 059QF0KO0R) STEARETH-100 (UNII: 4OH5W9UM87) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) ALUMINUM STARCH OCTENYLSUCCINATE (UNII: I9PJ0O6294) PHENOXYETHANOL (UNII: HIE492ZZ3T) CAPRYLYL GLYCOL (UNII: 00YIU5438U) DIMETHICONE (UNII: 92RU3N3Y1O) STEARETH-2 (UNII: V56DFE46J5) POLYESTER-7 (UNII: 0841698D2F) CHLORPHENESIN (UNII: I670DAL4SZ) ETHYLHEXYL STEARATE (UNII: EG3PA2K3K5) EDETATE DISODIUM (UNII: 7FLD91C86K) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) NEOPENTYL GLYCOL DIHEPTANOATE (UNII: 5LKW3C543X) LEVOMENOL (UNII: 24WE03BX2T) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) YEAST MANNAN (UNII: 91R887N59P) XANTHAN GUM (UNII: TTV12P4NEE) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) CAPRYLOYL GLYCINE (UNII: 8TY5YO42NJ) TRIDECETH-6 (UNII: 3T5PCR2H0C) CEDRUS ATLANTICA BARK (UNII: ITP1Q41UPF) SARCOSINE (UNII: Z711V88R5F) CINNAMON BARK OIL (UNII: XE54U569EC) PURSLANE (UNII: M6S840WXG5) 2-ETHYLHEXYL ACRYLATE, METHACRYLATE, METHYL METHACRYLATE, OR BUTYL METHACRYLATE/HYDROXYPROPYL DIMETHICONE COPOLYMER (30000-300000 MW) (UNII: S7ZA3CCJ4M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69968-0662-3 88 mL in 1 TUBE; Type 0: Not a Combination Product 10/05/2020 2 NDC:69968-0662-2 2 in 1 CARTON 10/05/2020 2 88 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 10/05/2020 Labeler - Kenvue Brands LLC (118772437)