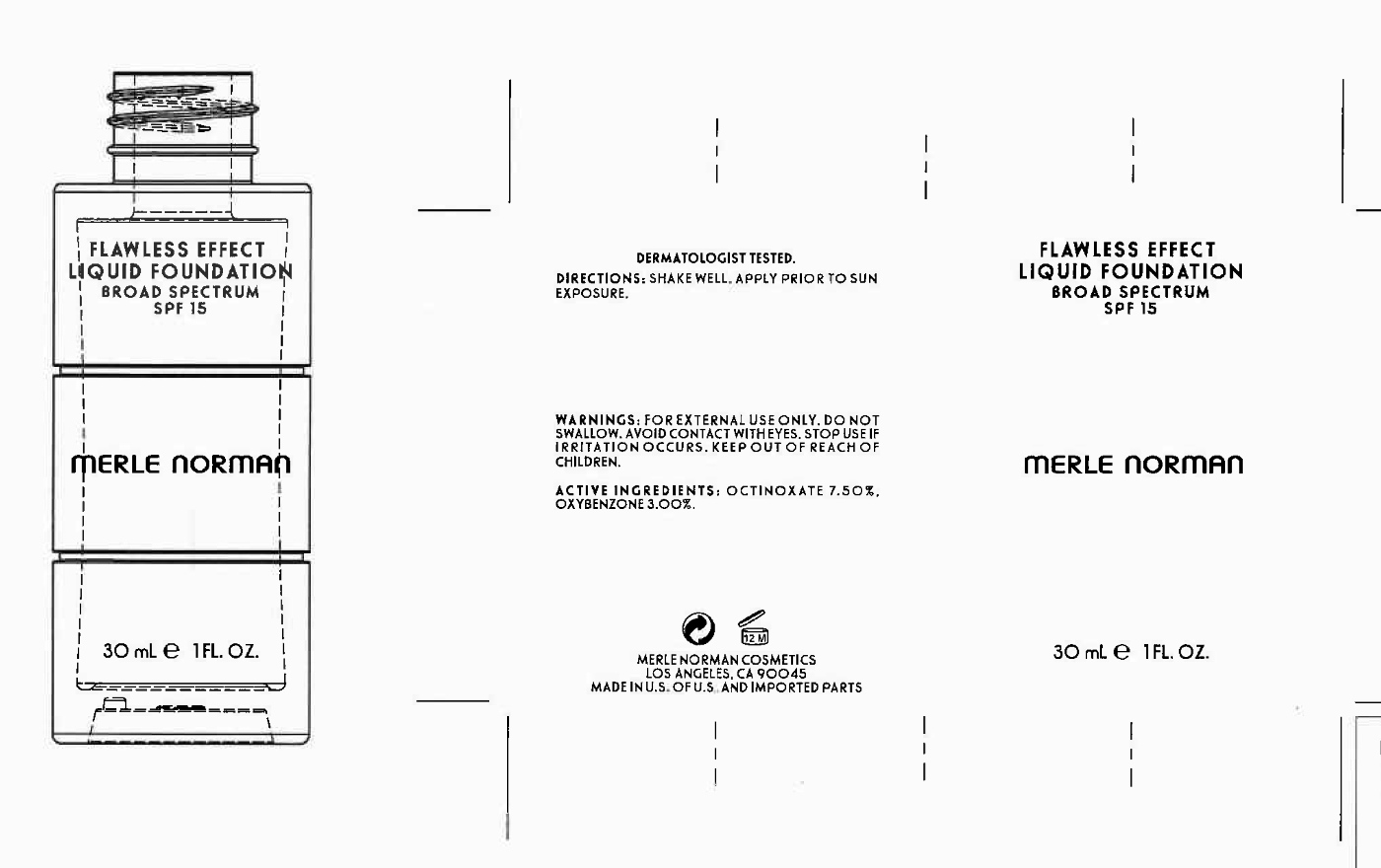
Label: FLAWLESS EFFECT LIQUID FOUNDATION SPF 15 MERLE NORMAN- octinoxate, oxybenzone liquid
- NDC Code(s): 57627-113-01, 57627-113-02
- Packager: Merle Norman
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 27, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active Ingredients
Octinoxate 7.50%
Oxybenzone 3.00%
Uses
· helps prevent sunburn
· If used as directed with other sun protection measures (see Directions),
decreases the risk of skin cancer and early skin aging caused by the sunKeep out of reach of children. If product is swallowed, get medical help or
contact a Poison Control Center right awayWarnings
· For external use only
· Do not use on damaged or broken skin
· When using this product keep out of eyes. Rinse with water to remove.
Directions
For Sunscreen Use:
· Apply liberally 15 minutes before sun exposure
· Use a water resistant sunscreen if swimming or sweating
· Reapply at least every 2 hours
· Sun Protection Measures. Spending time in the sun increases your risk of skin
cancer and early skin aging. To decrease this risk, regularly use a sunscreen
with broad spectrum SPF of 15 or higher and other sun protection measures
including:
· limit time in the sun, especially from 10 a.m. - 2 p.m.
· wear long-sleeve shirts, pants, hats, and sunglasses
· Children under 6 months: Ask a doctorWater(aqua), cyclopentasiloxane , mica, titanium dioxide, diisostearyl malate, ethylhexyl palmitate, glycerin, pentylene glycol, peg-30 dipolyhydroxystearate, iron oxides, propylene glycol, cyclohexasiloxane, aluminum starch octenylsuccinate, disteardimonium hectorite, hdi/ppg/polycaprolactone crosspolymer, glyceryl oleate, glyceryl stearate, myristyl myristate, polyglyceryl-3 beeswax, lecithin, disodium stearoyl glutamate, Butylene Glycol, sodium hyaluronate, camellia oleifera leaf extract, ginkgo biloba leaf extract, chamomilla recutita (matricaria) flower extract, tetrahexyldecyl ascorbate, tocopheryl acetate, aluminum hydroxide (Cl 77002), silica, alcohol denat., BHT, phenoxyethanol, methylparaben, propylparaben
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FLAWLESS EFFECT LIQUID FOUNDATION SPF 15 MERLE NORMAN
octinoxate, oxybenzone liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57627-113 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.9 g in 30 g OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 2.25 g in 30 g Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) MICA (UNII: V8A1AW0880) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ETHYLHEXYL PALMITATE (UNII: 2865993309) GLYCERIN (UNII: PDC6A3C0OX) PENTYLENE GLYCOL (UNII: 50C1307PZG) PEG-30 DIPOLYHYDROXYSTEARATE (UNII: 9713Q0S7FO) FERROUS OXIDE (UNII: G7036X8B5H) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) CYCLOMETHICONE 6 (UNII: XHK3U310BA) ALUMINUM STARCH OCTENYLSUCCINATE (UNII: I9PJ0O6294) GLYCERYL 1-OLEATE (UNII: D3AEF6S35P) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) MYRISTYL MYRISTATE (UNII: 4042ZC00DY) DISODIUM STEAROYL GLUTAMATE (UNII: 45ASM2L11M) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) HYALURONATE SODIUM (UNII: YSE9PPT4TH) CAMELLIA OLEIFERA LEAF (UNII: 5077EL0C60) GINKGO (UNII: 19FUJ2C58T) CHAMOMILE (UNII: FGL3685T2X) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) PHENOXYETHANOL (UNII: HIE492ZZ3T) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57627-113-02 1 in 1 CARTON 04/25/2011 1 NDC:57627-113-01 30 g in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 04/25/2011 Labeler - Merle Norman (008479388) Registrant - Merle Norman (008479388) Establishment Name Address ID/FEI Business Operations Merle Norman 008479388 manufacture(57627-113)