Label: NEOMYCIN AND POLYMYXIN B SULFATES AND HYDROCORTISONE- neomycin sulfate, polymyxin b sulfate, hydrocortisone solution/ drops
- NDC Code(s): 70518-0763-0
- Packager: REMEDYREPACK INC.
- This is a repackaged label.
- Source NDC Code(s): 24208-631
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 13, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Rx only
-
DESCRIPTION
Neomycin and polymyxin B sulfates and hydrocortisone otic solution, USP is a sterile antibacterial and anti-inflammatory solution for otic use.
Each mL contains: Actives:neomycin sulfate equivalent to 3.5 mg neomycin base, polymyxin B sulfate equivalent to 10,000 polymyxin B units, and hydrocortisone 10 mg (1%); Inactives:propylene glycol, hydrochloric acid, potassium metabisulfite, cupric sulfate, glycerin, purified water. The pH range is 2.0 to 4.5.
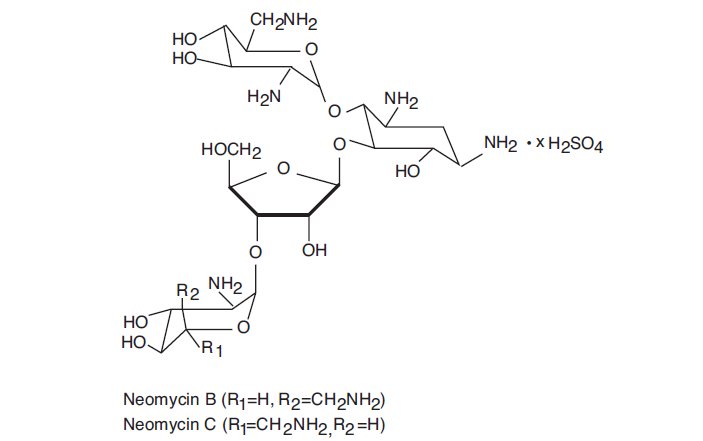
Neomycin sulfate is the sulfate salt of neomycin B and C, which are produced by the growth of Streptomyces fradiaeWaksman (Fam. Streptomycetaceae). It has a potency equivalent of not less than 600 micrograms of neomycin standard per mg, calculated on an anhydrous basis. The structural formulae are:
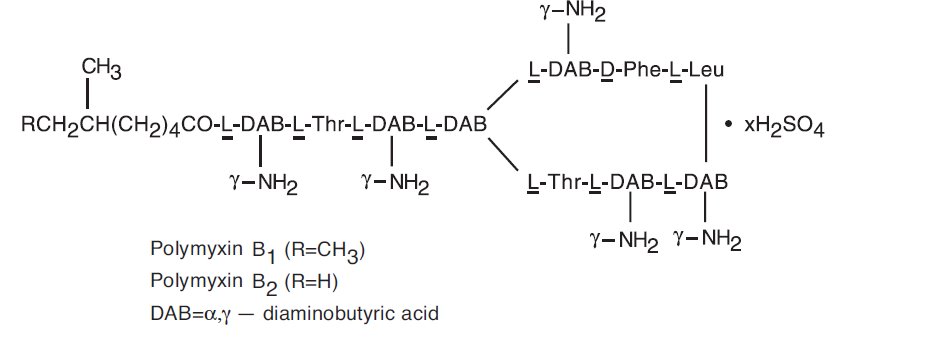
Polymyxin B sulfate is the sulfate salt of polymyxin B 1and B 2, which are produced by the growth of Bacillus polymyxa,(Prazmowski) Migula (Fam. Bacillaceae). It has a potency of not less than 6,000 polymyxin B units per mg, calculated on an anhydrous basis. The structural formulae are:
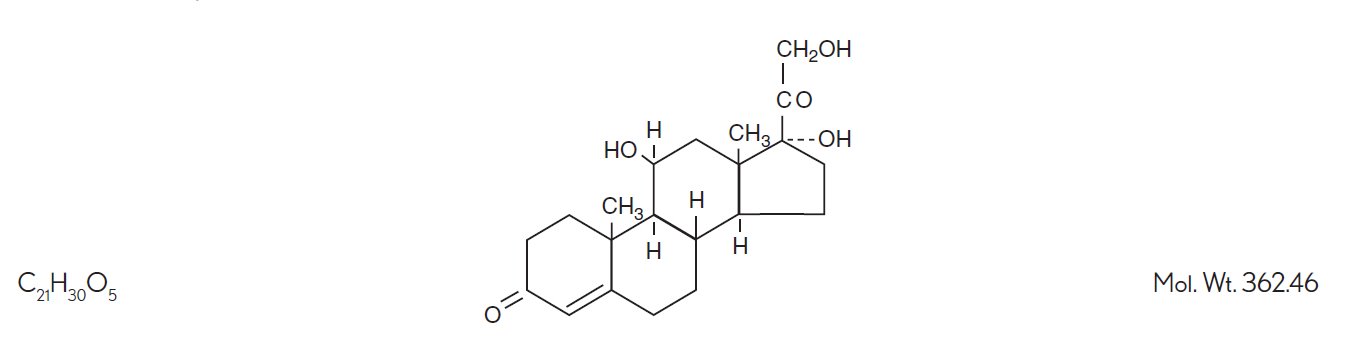
Hydrocortisone, 11β, 17, 21 - trihydroxypregn-4-ene-3, 20-dione, is an anti-inflammatory hormone. Its structural formula is:
-
CLINICAL PHARMACOLOGY
Corticoids suppress the inflammatory response to a variety of agents and they may delay healing. Since corticoids may inhibit the body's defense mechanism against infection, a concomitant antimicrobial drug may be used when this inhibition is considered to be clinically significant in a particular case.
The anti-infective components in the combination are included to provide action against specific organisms susceptible to them. Neomycin sulfate and polymyxin B sulfate together are considered active against the following microorganisms: Staphylococcus aureus, Escherichia coli, Haemophilus influenzae, Klebsiella-Enterobacterspecies, Neisseriaspecies, and Pseudomonas aeruginosa.This product does not provide adequate coverage against Serratia marcescensand streptococci, including Streptococcus pneumoniae.
The relative potency of corticosteroids depends on the molecular structure, concentration, and release from the vehicle.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
This product is contraindicated in those individuals who have shown hypersensitivity to any of its components. This product should not be used if the external auditory canal disorder is suspected or known to be due to cutaneous viral infection (for example, herpes simplex virus or varicella zoster virus).
-
WARNINGS
Neomycin can induce permanent sensorineural hearing loss due to cochlear damage, mainly destruction of hair cells in the organ of Corti. The risk of ototoxicity is greater with prolonged use; therefore, duration of therapy should be limited to 10 consecutive days (see PRECAUTIONS - General).
Patients being treated with eardrops containing neomycin should be under close clinical observation. Due to its acidity, which may cause burning and stinging, neomycin and polymyxin B sulfates and hydrocortisone otic solution should not be used in any patients with a perforated tympanic membrane.
Neomycin sulfate may cause cutaneous sensitization. A precise incidence of hypersensitivity reactions (primarily skin rash) due to topical neomycin is not known. Discontinue this product promptly if sensitization or irritation occurs.
When using neomycin-containing products to control secondary infection in the chronic dermatoses, such as chronic otitis externa or stasis dermatitis, it should be borne in mind that the skin in these conditions is more liable than is normal skin to become sensitized to many substances, including neomycin. The manifestation of sensitization to neomycin is usually a low-grade reddening with swelling, dry scaling and itching; it may be manifested simply as a failure to heal. Periodic examination for such signs is advisable, and the patient should be told to discontinue the product if they are observed. These symptoms regress quickly on withdrawing the medication. Neomycin-containing applications should be avoided for the patient thereafter.
Contains potassium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
-
PRECAUTIONS
General
As with other antibiotic preparations, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi.
If the infection is not improved after 1 week, cultures and susceptibility tests should be repeated to verify the identity of the organism and to determine whether therapy should be changed.
Treatment should not be continued for longer than 10 days.
Allergic cross-reactions may occur which could prevent the use of any or all of the following antibiotics for the treatment of future infections: kanamycin, paromomycin, streptomycin, and possibly, gentamicin.
Information for Patients
Avoid contaminating bottle tip with material from the ear, fingers or other source. This caution is necessary if the sterility of the drops is to be preserved.
If sensitization or irritation occurs, discontinue use immediately and contact your physician.
Do not use in the eyes.
Repackaged By / Distributed By: RemedyRepack Inc.
625 Kolter Drive, Indiana, PA 15701
(724) 465-8762
Laboratory Tests
Systemic effects of excessive levels of hydrocortisone may include a reduction in the number of circulating eosinophils and a decrease in urinary excretion of 17-hydroxycorticosteroids.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals (rats, rabbits, mice) showed no evidence of carcinogenicity attributable to oral administration of corticosteroids.
Pregnancy:
Teratogenic effects
Corticosteroids have been shown to be teratogenic in rabbits when applied topically at concentrations of 0.5% on days 6 to 18 of gestation and in mice when applied topically at a concentration of 15% on days 10 to 13 of gestation. There are no adequate and well-controlled studies in pregnant women. Corticosteroids should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
Hydrocortisone appears in human milk following oral administration of the drug. Since systemic absorption of hydrocortisone may occur when applied topically, caution should be exercised when neomycin and polymyxin B sulfates and hydrocortisone otic solution is used by a nursing woman.
Pediatric Use
The safety and effectiveness of neomycin and polymyxin B sulfates and hydrocortisone otic solution in otitis externa have been established in the pediatric age group 2 years to 16 years of age. There is inadequate data to establish safety and effectiveness in otitis externa for pediatric patients under 2 years of age. 1
Geriatric Use
Clinical studies of neomycin and polymyxin B sulfates and hydrocortisone otic solution did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
-
ADVERSE REACTIONS
Neomycin occasionally causes skin sensitization. Ototoxicity and nephrotoxicity have also been reported (see WARNINGS). Adverse reactions have occurred with topical use of antibiotic combinations including neomycin and polymyxin B. Exact incidence figures are not available since no denominator of treated patients is available. The reaction occurring most often is allergic sensitization. In one clinical study, using a 20% neomycin patch, neomycin-induced allergic skin reactions occurred in two of 2,175 (0.09%) individuals in the general population. 2In another study, the incidence was found to be approximately 1%. 3
The following local adverse reactions have been reported with topical corticosteroids, especially under occlusive dressings: burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, secondary infection, skin atrophy, striae, and miliaria. Stinging and burning have been reported when this product has gained access to the middle ear.
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
DOSAGE AND ADMINISTRATION
Therapy with this product should be limited to 10 consecutive days. The external auditory canal should be thoroughly cleansed and dried with a sterile cotton applicator.
For adults, four drops of the solution should be instilled into the affected ear 3 or 4 times daily. For infants and children, three drops are suggested because of the smaller capacity of the ear canal.
The patient should lie with the affected ear upward and then the drops should be instilled. This position should be maintained for 5 minutes to facilitate penetration of the drops into the ear canal.
Repeat, if necessary, for the opposite ear.
If preferred, a cotton wick may be inserted into the canal and then the cotton may be saturated with the solution. This wick should be kept moist by adding further solution every 4 hours. The wick should be replaced at least once every 24 hours.
-
HOW SUPPLIED
Neomycin and polymyxin B sulfates and hydrocortisone otic solution, USP is supplied in a white plastic dropper bottle in the following size:
NDC: 70518-0763-00
PACKAGING: 1 in 1 CARTON, 10 mL in 1 BOTTLE DROPPER TYPE 0
Storage: Store between 15°C to 25°C (59°F to 77°F).
Securely tighten cap between uses.
DO NOT USE IF IMPRINTED NECKBAND IS NOT INTACT.
Keep out of reach of children.
Repackaged and Distributed By:
Remedy Repack, Inc.
625 Kolter Dr. Suite #4 Indiana, PA 1-724-465-8762
-
REFERENCES
1. Jones RN, Milazzo J, Seidlin M. Ofloxacin Otic Solution for Treatment of Otitis Externa in Children and Adults. Arch Otolaryngol Head Neck Surg. 1997; 123:1193-1200.
2. Leyden JJ, Kligman AM. Contact dermatitis to neomycin sulfate. JAMA. 1979; 242:1276-1278.
3. Prystowsky SD, Allen AM, Smith RW, Nonomura JH, Odum RB, Akers WA. Allergic contact hypersensitivity to nickel, neomycin, ethylenediamine, and benzocaine: relationships between age, sex, history of exposure, and reactivity to standard patch tests and use tests in a general population. Arch Dermatol. 1979; 115:959-962.
Repackaged and Distributed By:
Remedy Repack, Inc.
625 Kolter Dr. Suite #4 Indiana, PA 1-724-465-8762
-
PRINCIPAL DISPLAY PANEL
DRUG: Neomycin and Polymyxin B Sulfates and Hydrocortisone
GENERIC: Neomycin Sulfate, Polymyxin B Sulfate, Hydrocortisone
DOSAGE: SOLUTION/ DROPS
ADMINSTRATION: AURICULAR (OTIC)
NDC: 70518-0763-0
PACKAGING: 10 mL in 1 BOTTLE, DROPPER
OUTER PACKAGING: 1 in 1 CARTON
ACTIVE INGREDIENT(S):
- POLYMYXIN B SULFATE 10000[USP'U] in 1mL
- NEOMYCIN SULFATE 3.5mg in 1mL
- HYDROCORTISONE 10mg in 1mL
INACTIVE INGREDIENT(S):
- PROPYLENE GLYCOL
- HYDROCHLORIC ACID
- POTASSIUM METABISULFITE
- CUPRIC SULFATE
- GLYCERIN
- WATER

-
INGREDIENTS AND APPEARANCE
NEOMYCIN AND POLYMYXIN B SULFATES AND HYDROCORTISONE
neomycin sulfate, polymyxin b sulfate, hydrocortisone solution/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70518-0763(NDC:24208-631) Route of Administration AURICULAR (OTIC) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 mL POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 10000 [USP'U] in 1 mL HYDROCORTISONE (UNII: WI4X0X7BPJ) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) HYDROCHLORIC ACID (UNII: QTT17582CB) POTASSIUM METABISULFITE (UNII: 65OE787Q7W) CUPRIC SULFATE (UNII: LRX7AJ16DT) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70518-0763-0 1 in 1 CARTON 10/06/2017 1 10 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA064053 10/06/2017 Labeler - REMEDYREPACK INC. (829572556)