Label: SOTALOL HYDROCHLORIDE injection
- NDC Code(s): 69724-112-10
- Packager: ALTATHERA Pharmaceuticals, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated October 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use SOTALOL HYDROCHLORIDE injection safely and effectively. See full prescribing information for SOTALOL HYDROCHLORIDE.
SOTALOL HYDROCHLORIDE injection, for intravenous use
Initial U.S. Approval: 1992WARNING: LIFE THREATENING PROARRHYTHMIA
See full prescribing information for complete boxed warning.
- Sotalol can cause life threatening ventricular tachycardia associated with QT interval prolongation. ( 5.1)
- Initiate or reinitiate in a facility that can provide cardiac resuscitation and continuous electrocardiographic monitoring. ( 2.3)
- Do not initiate intravenous sotalol therapy if the baseline QTc is longer than 450 ms. If the QTc prolongs to 500 ms or greater, reduce the dose or discontinue. ( 2.3)
RECENT MAJOR CHANGES
Warnings and Precautions, Hypoglycemia ( 5.7) 05/2023
INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
(2)
- Intravenous sotalol can substitute for oral sotalol using a regimen that mimics oral exposure ( 2.2)
- Under close medical monitoring, intravenous sotalol can be used to achieve near steady-state exposure to sotalol prior to initiating or increasing oral dosing. ( 2.3)
- Recommended dosage depends on target oral dose and creatinine clearance. Refer to full prescribing information. ( 2.2, 2.3, 2.4)
(2)
DOSAGE FORMS AND STRENGTHS
Injection: 10-mL vial containing sotalol hydrochloride 150 mg ( 3)
CONTRAINDICATIONS
- Bradyarrhythmia, sick sinus syndrome or 2 nd or 3 rd degree AV block without a pacemaker ( 4)
- Congenital or acquired long QT syndrome ( 4)
- Cardiogenic shock or decompensated heart failure ( 4)
- Serum potassium < 4mEq ( 4)
- Bronchial asthma or related bronchospastic conditions ( 4)
- Hypersensitivity to sotalol ( 4)
WARNINGS AND PRECAUTIONS
(5)
- Risk of life-threatening ventricular arrhythmias, particularly torsade de pointes ( 5.1)
- Bradyarrhythmia, heart block, sick sinus syndrome ( 5.2, 5.3)
- Negative inotropy: hypotension, heart failure ( 5.4, 5.5)
- Bronchospasm ( 5.6)
- Masked hypoglycemia ( 5.7)
- Masked hyperthyroidism ( 5.8)
- Anaphylaxis ( 5.9)
- Diabetes: May mask symptoms of hypoglycemia and alter glucose levels; monitor ( 5.5)
(5)
ADVERSE REACTIONS
DRUG INTERACTIONS
Revised: 5/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: LIFE THREATENING PROARRHYTHMIA
RECENT MAJOR CHANGES
1 INDICATIONS AND USAGE
1.1 Delay in Recurrence of Atrial Fibrillation/Atrial Flutter
1.2 Life-Threatening Ventricular Arrhythmia
2 DOSAGE AND ADMINISTRATION
2.1 General Considerations
2.2 Use for Substitution of Oral Sotalol
2.3 Use for Loading Dose
2.4 Use in Children
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Proarrhythmia
5.2 Bradycardia/Heart Block
5.3 Sick Sinus Syndrome
5.4 Hypotension
5.5 Heart Failure
5.6 Bronchospasm
5.7 Hypoglycemia
5.8 Thyroid Abnormalities
5.9 Anaphylaxis
5.10 Anesthesia
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 Negative Chronotropes
7.2 Calcium Blocking Drugs
7.3 Catecholamine-Depleting Agents
7.4 Insulin and Oral Antidiabetics
7.5 Beta-2-Receptor Stimulants
7.6 Clonidine
7.7 Drug/Laboratory Test Interactions
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Clinical Studies in Ventricular Arrhythmias with Oral Sotalol
14.2 Clinical Studies in Supra-Ventricular Arrhythmias with Oral Sotalol
14.3 Clinical Studies in Patients with Myocardial Infarction
14.4 Intravenous Loading Dosage
16 HOW SUPPLIED/STORAGE AND HANDLING
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: LIFE THREATENING PROARRHYTHMIA
Sotalol can cause life threatening ventricular tachycardia associated with QT interval prolongation. To minimize the risk of drug induced arrhythmia, initiate or uptitrate intravenous sotalol in a facility that can provide continuous electrocardiographic monitoring and cardiac resuscitation [see Dosage and Administration ( 2.3) and Warnings and Precautions ( 5.1)]
Do not initiate intravenous sotalol therapy if the baseline QTc is longer than 450 ms. If the QTc prolongs to 500 ms or greater, reduce the dose or discontinue.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 General Considerations
For either indication, intravenous sotalol can substitute for oral sotalol in patients unable to take oral drugs or be used to achieve steady state concentration faster compared to the conventional oral dosing.
Intravenous sotalol must be diluted for infusion. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Dilute intravenous sotalol in saline, 5% dextrose in water (D5W), or Ringer's lactate. Choose a volume convenient for administration and consistent with fluid restriction. Use a volumetric infusion pump.
2.2 Use for Substitution of Oral Sotalol
To match the exposure to oral sotalol, use the same dosing frequency with intravenous administration and infuse the adjusted dose (see Table 1) over 5 hours.
Table 1: Intravenous doses for sotalol infusion when used as a substitute for oral sotalol Oral dose Intravenous dose Undiluted volume 80 mg 75 mg 5 mL 120 mg 112.5 mg 7.5 mL 160 mg 150 mg 10 mL 2.3 Use for Loading Dose
Initiate or uptitrate intravenous sotalol in a facility that can provide continuous ECG monitoring and cardiac resuscitation. Personnel should be trained in the management of serious ventricular arrhythmias. Withdraw other antiarrhythmic therapy before starting sotalol hydrochloride.
Measure and normalize serum potassium and magnesium levels before initiation. If the baseline QTc is >450 ms (JT >330 ms if QRS over 100 ms), sotalol is not recommended.
Infuse the loading dose over one hour. Monitor QTc interval every 15 minutes during infusion. Continue to monitor QTc around Tmax (2 to 4 hours post-dose) following the first oral dose (in all patients) and second oral dose (in patients with CrCL≥60 mL/min). If the QTc interval prolongs to >500 ms or increases 20% from baseline when initiating for an oral dose of 80 mg, discontinue drug; if initiating for an oral dose of 120 mg discontinue drug and consider a lower dose. If re-initiation at a lower dose of 80 mg is desired, wait at least 1 day (in patients with CrCL≥60 mL/min), or at least 3 days (in patients with CrCL ≥30 to <60 mL/min), or 7 days (in patients with CrCL ≥10 to <30 mL/min).
The intravenous loading dose depends on the target oral dose and creatinine clearance; the dosing interval for oral administration of sotalol and the minimum delay between the end of the infusion and the first oral dose also depend on renal function; see Table 2 [see Clinical Studies (14.4)].
Table 2: Recommended Loading Dosage Creatinine Clearance * [mL/min] Intravenous loading dose [mg] to be administered over 1 hour when the oral dose is going from Minimum delay to first oral dose [hours] Oral dosing interval [hours] Sotalol Initiation Sotalol Escalation 0 to 80 mg † 0 to 120 mg 80 to 120 mg 120 to 160 mg >90 60 90 75 90 4 12 60-90 82.5 125 82.5 105 4 12 30-60 75 112.5 82.5 105 6 24 10-30 75 112.5 82.5 105 12 48 2.4 Use in Children
Intravenous sotalol has not been studied in children. Potency is thought to be similar in children, so a dose of 30 mg/m 2 every 8 hours should give exposure similar to that with 160 mg daily in adults. Table 3 shows suggested starting doses in children with normal renal function. With impaired renal function, start lower and titrate less frequently.
Table 3. Suggesting starting doses [mg/kg] in children with normal renal function. Age Dose [mg/kg] 3 days 0.32 6 days 0.51 9 days 0.69 12 days 0.81 2 weeks 0.90 3 weeks 1.0 1 month to 6 years �1.2 6-12 years 1.1 >12 years 0.95 - 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Sotalol hydrochloride is contraindicated in patients with:
- Sinus bradycardia (<50 bpm), sick sinus syndrome or second or third degree AV block without a pacemaker
- Congenital or acquired long QT syndromes, QT interval >450 ms
- Cardiogenic shock, decompensated heart failure
- Serum potassium <4 mEq/L
- Bronchial asthma or related bronchospastic conditions
- Known hypersensitivity to sotalol
-
5 WARNINGS AND PRECAUTIONS
5.1 Proarrhythmia
Sotalol can cause serious ventricular arrhythmias, primarily Torsade de Pointes (TdP) type ventricular tachycardia, a polymorphic ventricular tachycardia associated with QTc prolongation. QTc prolongation is directly related to the concentration of sotalol. Factors such as reduced creatinine clearance, gender (female) and higher dose, bradycardia, history of sustained VT/VF, atrial fibrillation with sinus node dysfunction, heart failure increase the risk of TdP. The risk of TdP can be reduced by adjustment of the sotalol dose according to creatinine clearance and by monitoring the ECG for excessive increases in QTc.
Correct hypokalemia or hypomagnesemia prior to initiating sotalol hydrochloride, as these conditions can exaggerate the degree of QT prolongation and increase the potential for TdP. Special attention should be given to electrolyte and acid-base balance in patients experiencing severe or prolonged diarrhea or patients receiving concomitant diuretic drugs.
Avoid sotalol with other drugs known to cause QT prolongation [see Drug Interactions ( 7.1)].
5.2 Bradycardia/Heart Block
Sotalol can cause bradycardia, sinus pauses or sinus arrest. Sotalol-induced bradycardia increases the risk of Torsade de Pointe, particularly following cardioversion. Monitor the ECG in patients receiving concomitant negative chronotropes [see Drug Interactions ( 7.1, 7.2, 7.3)] .
5.3 Sick Sinus Syndrome
In general, sotalol is not recommended in patients with sick sinus syndrome associated with symptomatic arrhythmias, because it may cause sinus bradycardia, sinus pauses, or sinus arrest. Sotalol augments bradycardia and QTc prolongation following cardioversion. Patients with AFIB/AFL associated with the sick sinus syndrome may be treated with sotalol if they have an implanted pacemaker for control of bradycardia symptoms.
5.4 Hypotension
Sotalol produces significant reductions in both systolic and diastolic blood pressures. Monitor hemodynamics during administration.
5.5 Heart Failure
New onset or worsening heart failure may occur during initiation or up-titration of sotalol because of its beta-blocking effects. Monitor for signs and symptoms of heart failure and discontinue treatment if symptoms occur.
5.6 Bronchospasm
Avoid beta-blockers, like sotalol, in patients with bronchospastic diseases. If sotalol is required, use the smallest effective dose.
5.7 Hypoglycemia
Beta-blockade may prevent early warning signs of hypoglycemia, such as tachycardia, and increase the risk for severe or prolonged hypoglycemia at any time during treatment, especially in patients with diabetes mellitus or children and patients who are fasting (i.e., surgery, not eating regularly, or and vomiting). Monitor blood glucose as appropriate.
5.8 Thyroid Abnormalities
Avoid abrupt withdrawal of beta-blockade which might be followed by an exacerbation of symptoms of hyperthyroidism, including thyroid storm. Beta-blockade may mask certain clinical signs (e.g., tachycardia) of hyperthyroidism.
5.9 Anaphylaxis
While taking beta-blockers, patients with a history of anaphylactic reaction to a variety of allergens may have a more severe reaction on repeated challenge, either accidental, diagnostic, or therapeutic. Such patients may be unresponsive to the usual doses of epinephrine used to treat the allergic reaction.
-
6 ADVERSE REACTIONS
The following adverse reactions are described elsewhere:
Adverse reactions related to sotalol use are those which are typical of its Class II (beta- blocking) and Class III (cardiac action potential duration prolongation) effects. The common documented beta-blocking adverse reactions (bradycardia, dyspnea, and fatigue) and Class III effects (QT interval prolongation) are dose related.
-
7 DRUG INTERACTIONS
7.1 Negative Chronotropes
Both digitalis glycosides and beta-blockers slow atrioventricular conduction and decrease heart rate. Concomitant use can increase the risk of bradycardia.
7.2 Calcium Blocking Drugs
Sotalol and calcium blocking drugs can be expected to have additive effects on atrioventricular conduction, ventricular function, and blood pressure.
7.3 Catecholamine-Depleting Agents
Concomitant use of catecholamine-depleting drugs, such as reserpine and guanethidine, with a beta- blocker may produce an excessive reduction of resting sympathetic nervous tone. Monitor such patients for hypotension and marked bradycardia which may produce syncope.
7.4 Insulin and Oral Antidiabetics
Hyperglycemia may occur, and the dosage of insulin or antidiabetic drugs may require adjustment. Symptoms of hypoglycemia may be masked.
7.5 Beta-2-Receptor Stimulants
Beta-agonists such as albuterol, terbutaline and isoproterenol may have to be administered in increased dosages when used concomitantly with sotalol.
7.6 Clonidine
Concomitant use with sotalol increases the risk of bradycardia. Because beta-blockers may potentiate the rebound hypertension sometime observed after clonidine discontinuation, withdraw sotalol several days before the gradual withdrawal of clonidine to reduce the risk of rebound hypertension.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Both the untreated underlying condition in pregnancy and the use of sotalol in pregnancy cause adverse outcomes to the mother and fetus/neonate (see Clinical Considerations). In animal reproduction studies in rats, early resorptions were increased at 15 times the maximum recommended human dose (MRHD). In rabbits an increase in fetal death was observed at 2 times the MRHD administered as single dose. Sotalol did not reveal any teratogenic potential in rats or rabbits at 15 and 2 times the MRHD respectively (see Data).
All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the United States (U.S.) general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
The incidence of VT is increased and may be more symptomatic during pregnancy. Most tachycardia episodes are initiated by ectopic beats and the occurrence of arrhythmia episodes may, therefore, increase during pregnancy. Breakthrough arrhythmias may also occur during pregnancy, as therapeutic treatment levels may be difficult to maintain due to the increased volume of distribution and increased drug metabolism inherent in the pregnant state.
Fetal/Neonatal Adverse Reactions
Sotalol has been shown to cross the placenta and is found in amniotic fluid. From published observational studies, the potential fetal adverse effects of sotalol use during pregnancy are growth restriction, transient fetal bradycardia, hyperbilirubinemia, hypoglycemia, uterine contractions, and possible intrauterine death. Sotalol may have a greater effect on QT prolongation in the immature heart than in the adult heart, and therefore, conveys an increased risk of serious fetal arrhythmia and/or possible intrauterine death. Monitor the newborn for symptoms of beta blockade.
Labor or Delivery
Generally, risk of arrhythmias increases during the labor and delivery process; therefore, considering the proarrhythmia potential of the drug, patients treated with sotalol should be monitored continuously during labor and delivery.
Data
Animal Data
Reproduction studies in rats and rabbits administered sotalol during organogenesis at 15 times and 2 times the MRHD as mg/m 2, respectively, did not reveal any teratogenic potential associated with sotalol.
In pregnant rats, sotalol doses administered during organogenesis at approximately15 times the MRHD as mg/m 2, increased the number of early resorptions, while no increase in early resorptions was noted at 2 times the MRHD as mg/m 2.
In reproductive studies in rabbits, a sotalol dose (160 mg/kg/day) at 5 times the MRHD as mg/m 2 produced a slight increase in fetal death, and maternal toxicity. However, one study from published data reported an increase in fetal deaths in rabbits receiving a single dose (50 mg/kg) at 2 times the MRHD as mg/m 2 as on gestation day 14.
8.2 Lactation
Risk Summary
Limited available data from published literature report that sotalol is present in human milk. The estimated daily infant dose of sotalol received from breastmilk is 0.8-3.4 mg/kg, estimated at 22 to 25.5% of the maternal weight-adjusted dosage of sotalol (see Data). The amount of the drug in breast milk is similar to the neonatal therapeutic dosage. Therefore, there is potential for bradycardia and other symptoms of beta blockade such as dry mouth, skin or eyes, diarrhea or constipation in the breastfed infant. There is no information regarding the effects of sotalol on milk production. Because of the potential serious adverse reactions to the breastfed child and the high level of sotalol in breast milk, advise women not to breastfeed while on treatment with sotalol.
Data
Sotalol is present in human milk in high levels. A prospective study evaluated 20 paired samples of breast milk and maternal blood from 5 mothers who elected to breastfeed. Breast milk samples had a mean sotalol concentration of 10.5 µg/mL (± 1.1 µg/mL; range: 4.8 to 20.2 µg/mL) compared to a simultaneous mean maternal plasma concentration of 2.3 µg/mL (± 0.3 µg/mL; range: 0.8 to 5.0 µg/mL). The mean milk plasma ratio was 5.4:1 (range: 2.2 to 8.8). The estimated daily infant dose was 0.8-3.4 mg/kg, estimated at 22 to 25.5% of the maternal weight-adjusted dosage of sotalol. This is similar to recommended therapeutic dose in neonates. None of the mothers reported any adverse reactions in the breastfed infant.
8.3 Females and Males of Reproductive Potential
Infertility
Based on the published literature, beta blockers (including sotalol) may cause erectile dysfunction.
8.4 Pediatric Use
The safety and effectiveness of sotalol in children has not been established. However, the Class III electrophysiologic and beta-blocking effects, the pharmacokinetics, and the relationship between the effects (QTc interval and resting heart rate) and drug concentrations have been evaluated in children aged between 3 days and 12 years old [see Clinical Pharmacology ( 12.3) ].
-
10 OVERDOSAGE
Overdosage with sotalol may be fatal.
Symptoms and Treatment of Overdosage: The most common signs to be expected are bradycardia, congestive heart failure, hypotension, bronchospasm and hypoglycemia. In cases of massive intentional overdosage (2-16 grams) of sotalol the following clinical findings were seen: hypotension, bradycardia, cardiac asystole, prolongation of QT interval, Torsade de Pointes, ventricular tachycardia, and premature ventricular complexes. If overdosage occurs, therapy with sotalol should be discontinued and the patient observed closely. Because of the lack of protein binding, hemodialysis is useful for reducing sotalol plasma concentrations. Patients should be carefully observed until QT intervals are normalized and the heart rate returns to levels >50 bpm. The occurrence of hypotension following an overdose may be associated with an initial slow drug elimination phase (half-life of 30 hours) thought to be due to a temporary reduction of renal function caused by the hypotension. In addition, if required, the following therapeutic measures are suggested:
Bradycardia or
Cardiac Asystole:
Atropine, another anticholinergic drug, a beta-adrenergic agonist or
transvenous cardiac pacing.
Heart Block: (second and third degree) transvenous cardiac pacemaker. Hypotension: (depending on associated factors) epinephrine rather than isoproterenol or norepinephrine may be useful. Bronchospasm: Aminophylline or aerosol beta-2-receptor stimulant. Higher than normal doses of beta-2 stimulus may be required. Torsade de Pointes: DC cardioversion, magnesium sulfate, potassium replacement. Once Torsade de Pointes is terminated, transvenous cardiac pacing or an isoproterenol infusion to increase heart rate can be employed. -
11 DESCRIPTION
Sotalol hydrochloride injection is an aqueous formulation of sotalol hydrochloride for intravenous use. Sotalol is an antiarrhythmic drug with Class II (beta-adrenoreceptor blocking) and Class III (cardiac action potential duration prolongation) properties.
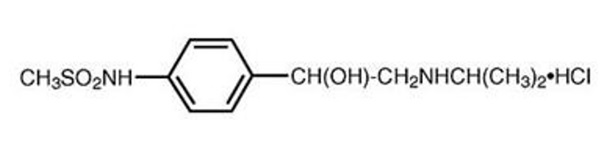
Sotalol hydrochloride is a white, crystalline solid with a molecular weight of 308.8. It is hydrophilic, soluble in water, propylene glycol and ethanol, but is only slightly soluble in chloroform. Chemically, sotalol hydrochloride is d, l- N - [4-[1-hydroxy-2-[(1-methylethyl) amino] ethyl] phenyl] methane-sulfonamide monohydrochloride. The molecular formula is C 12 H 20 N 2 O 3S•HCl and is represented by the following structural formula:
Intravenous sotalol Injection is supplied as a sterile, clear solution in a 10 mL single-dose vial, for intravenous administration after dilution. Each vial contains 150 mg racemic sotalol hydrochloride (equivalent to 132.8 mg racemic sotalol) in sodium acetate buffer. The sotalol hydrochloride concentration of the formulation is 15 mg/mL. Each mL contains 2.9 mg glacial acetic acid in water for injection as an inactive ingredient. The pH of the injection is adjusted with sodium hydroxide to be between 6.0 and 7.0.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Sotalol has both beta-adrenoreceptor blocking (Vaughan Williams Class II) and cardiac action potential duration prolongation (Vaughan Williams Class III) antiarrhythmic properties. Intravenous sotalol is a racemic mixture of d- and l-sotalol. Both isomers have similar Class III antiarrhythmic effects, while the l-isomer is responsible for virtually all of the beta-blocking activity. The beta-blocking effect of sotalol is non-cardioselective, half maximal at oral doses of about 80 mg/day and maximal at oral doses between 320 and 640 mg/day. Significant beta-blockade occurs at oral doses as low as 25 mg, significant Class III effects can be seen after a single oral or IV dose of 80 or 75 mg respectively.
12.2 Pharmacodynamics
Electrophysiology: Sotalol prolongs the plateau phase of the cardiac action potential in the isolated myocyte, as well as in isolated tissue preparations of ventricular or atrial muscle (Class III activity). In intact animals it slows heart rate, decreases AV nodal conduction and increases the refractory periods of atrial and ventricular muscle and conduction tissue.
In man, the Class II (beta-blockade) electrophysiological effects of sotalol include increased sinus cycle length (slowed heart rate), decreased AV nodal conduction, and increased AV nodal refractoriness. The Class III electrophysiological effects in man include prolongation of the atrial and ventricular monophasic action potentials, and effective refractory period prolongation of atrial muscle, ventricular muscle, and atrio-ventricular accessory pathways (when present) in both the anterograde and retrograde directions. With oral doses of 160 to 640 mg/day, the surface ECG shows dose-related mean increases of 40-100 ms in QT and 10-40 ms in QTc.
Twenty-five children in an unblinded, multicenter trial with supraventricular (SVT) and/or ventricular (VT) tachyarrhythmias, aged between 3 days and 12 years (mostly neonates and infants), received an ascending titration regimen with daily doses of 30, 90 and 210 mg/m 2 with dosing every 8 hours for a total of 9 doses. During steady-state, the respective average increases above baseline of the QTc interval were 2, 14, and 29 ms at the 3 dose levels. The respective mean maximum increases in QTc above baseline were 23, 36, and 55 ms. The steady-state percent increases in the RR interval were 3, 9 and 12%. The smallest children (BSA <0.33 m 2) showed a tendency for larger Class III effects (ΔQTc) and an increased frequency of prolongations of the QTc interval compared with children with BSA ≥0.33 m 2. The beta-blocking effects also tended to be greater in the smaller children (BSA<0.33 m 2).
Hemodynamics: In a study of systemic hemodynamic function measured invasively in 12 patients with a mean LV ejection fraction of 37% and ventricular tachycardia (9 sustained and 3 non-sustained), a median dose of 160 mg twice daily of sotalol produced a 28% reduction in heart rate and a 24% decrease in cardiac index at 2 hours post-dosing at steady-state. Concurrently, systemic vascular resistance and stroke volume showed non-significant increases of 25% and 8%, respectively.
Pulmonary capillary wedge pressure increased significantly from 6 to 12 mmHg in the 11 patients who completed the study. Mean arterial pressure, mean pulmonary artery pressure and stroke work index did not significantly change. Exercise and isoproterenol induced tachycardia are antagonized by sotalol, and total peripheral resistance increases by a small amount.
In hypertensive patients, sotalol produces significant reductions in both systolic and diastolic blood pressures. Although sotalol is usually well-tolerated hemodynamically, in patients with marginal cardiac compensation, deterioration in cardiac performance may occur [see Warnings and Precautions ( 5.3)].
12.3 Pharmacokinetics
The pharmacokinetics of the d and l isomers of sotalol are essentially identical.
Absorption
In healthy subjects, the oral bioavailability of sotalol is >90%. After oral administration, peak plasma concentrations are reached in 2.5 to 4 hours, and steady-state plasma concentrations are attained within 2-3 days (i.e., after 5-6 doses when administered twice daily). Over the oral dosage range 160-640 mg/day, sotalol displays dose proportionality with respect to plasma concentrations. When administered with a standard meal, the absorption of sotalol was reduced by approximately 20% compared to administration in the fasting state.
Distribution
Distribution occurs to a central (plasma) and to a peripheral compartment, with a mean elimination half-life of 12 hours. Dosing every 12 hours results in trough plasma concentrations which are approximately one-half of those at peak.
Sotalol does not bind to plasma proteins and is not metabolized. Sotalol shows very little intersubject variability in plasma levels. Sotalol crosses the blood brain barrier poorly.
Metabolism
Sotalol is not metabolized and is not expected to inhibit or induce any CYP 450 enzymes.
Excretion
Excretion is predominantly via the kidney in the unchanged form, and therefore lower doses are necessary in conditions of renal impairment [see Dosage and Administration ( 2.3)]. Dosing every 12 hours results in trough plasma concentration which are approximately one half of that at peak concentration.
Specific Populations
Geriatric
Age per se does not significantly alter the pharmacokinetics of sotalol, but impaired renal function in geriatric patients can increase the terminal elimination half-life, resulting in increased drug accumulation.
Hepatic Disease
Since sotalol is not subject to first-pass metabolism, patients with hepatic impairment show no alteration in clearance of sotalol.
Pediatric
The combined analysis of two unblinded, multicenter trials (a single dose and a multiple dose study) with 59 children, aged between 3 days and 12 years, showed the pharmacokinetics of sotalol to be first order. A daily dose of 30 mg/ m 2 of sotalol was administered in the single dose study and daily doses of 30, 90 and 210 mg/ m 2 were administered q8h in the multi-dose study. After rapid absorption with peak levels occurring on average between 2-3 hours following administration, sotalol was eliminated with a mean half-life of 9.5 hours. Steady-state was reached after 1-2 days. The average peak to trough concentration ratio was 2. BSA was the most important covariate and more relevant than age for the pharmacokinetics of sotalol. The smallest children (BSA <0.33 m 2) exhibited a greater drug exposure (+59%) than the larger children who showed a uniform drug concentration profile. The intersubject variation for oral clearance was 22%.
Drug-Drug Interactions
Antacids
Administration of oral sotalol within 2 hours of antacids may result in a reduction in Cmax and AUC of 26% and 20%, respectively, and consequently in a 25% reduction in the bradycardic effect at rest. Administration of the antacid two hours after oral sotalol has no effect on the pharmacokinetics or pharmacodynamics of sotalol.
No pharmacokinetic interactions were observed with hydrochlorothiazide or warfarin.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of carcinogenic potential was observed in rats during a 24-month study at 137-275 mg/kg/ day (approximately 30 times the maximum recommended human oral dose (MRHD) as mg/kg or 5 times the MRHD as mg/ m 2) or in mice, during a 24-month study at 4141-7122 mg/kg/day (approximately 450- 750 times the MRHD as mg/kg or 36-63 times the MRHD as mg/ m 2).
Sotalol has not been evaluated in any specific assay of mutagenicity or clastogenicity.
No significant reduction in fertility occurred in rats at oral doses of 1000 mg/kg/day (approximately 100 times the MRHD as mg/kg or 9 times the MRHD as mg/ m 2) prior to mating, except for a small reduction in the number of offspring per litter. Reproduction studies in rats and rabbits during organogenesis at 100 and 22 times the MRHD as mg/kg (9 and 7 times the MRHD as mg/ m 2), respectively, did not reveal any teratogenic potential associated with sotalol HCl. In rabbits, a high dose of sotalol HCl (160 mg/kg/day) at 16 times the MRHD as mg/kg (6 times the MRHD as mg/ m 2) produced a slight increase in fetal death likely due to maternal toxicity. Eight times the maximum dose (80 mg/kg/day or 3 times the MRHD as mg/ m 2) did not result in an increased incidence of fetal deaths. In rats, 1000 mg/kg/day sotalol HCl, 100 times the MRHD (18 times the MRHD as mg/ m 2), increased the number of early resorptions, while at 14 times the maximum dose (2.5 times the MRHD as mg/ m 2), no increase in early resorptions was noted. However, animal reproduction studies are not always predictive of human response.
-
14 CLINICAL STUDIES
14.1 Clinical Studies in Ventricular Arrhythmias with Oral Sotalol
Sotalol has been studied in life-threatening and less severe arrhythmias. In patients with frequent premature ventricular complexes (VPC), orally administered sotalol was significantly superior to placebo in reducing VPCs, paired VPCs and non-sustained ventricular tachycardia (NSVT); the response was dose-related through 640 mg/day with 80-85% of patients having at least a 75% reduction of VPCs. Sotalol was also superior, at the doses evaluated, to propranolol (40-80 mg TID) and similar to quinidine (200-400 mg QID) in reducing VPCs. In patients with life-threatening arrhythmias [sustained ventricular tachycardia/fibrillation (VT/VF)], sotalol was studied acutely [by suppression of programmed electrical stimulation (PES) induced VT and by suppression of Holter monitor evidence of sustained VT] and, in acute responders, chronically.
In a double-blind, randomized comparison of oral sotalol and procainamide given intravenously (total of 2 mg/kg sotalol vs. 19 mg/kg of procainamide over 90 minutes), sotalol suppressed PES induction in 30% of patients vs. 20% for procainamide (p=0.2).
In a randomized clinical trial [Electrophysiologic Study Versus Electrocardiographic Monitoring (ESVEM) Trial] comparing choice of antiarrhythmic therapy by PES suppression vs. Holter monitor selection (in each case followed by treadmill exercise testing) in patients with a history of sustained VT/VF who were also inducible by PES, the effectiveness acutely and chronically of sotalol was compared with 6 other drugs (procainamide, quinidine, mexiletine, propafenone, and imipramine). Overall response, limited to first randomized drug, was 39% for sotalol and 30% for the pooled other drugs. Acute response rate for first drug randomized using suppression of PES induction was 36% for sotalol vs. a mean of 13% for the other drugs. Using the Holter monitoring endpoint (complete suppression of sustained VT, 90% suppression of NSVT, 80% suppression of VPC pairs, and at least 70% suppression of VPCs), sotalol yielded 41% response vs. 45% for the other drugs combined. Among responders placed on long term therapy identified acutely as effective (by either PES or Holter), sotalol, when compared to the pool of other drugs, had the lowest two-year mortality (13% vs. 22%), the lowest two-year VT recurrence rate (30% vs. 60%), and the lowest withdrawal rate (38% vs. about 75-80%). The most commonly used doses of sotalol in this trial were 320-480 mg/day (66% of patients), with 16% receiving 240 mg/day or less and 18% receiving 640 mg or more.
It cannot be determined, however, in the absence of a controlled comparison of sotalol vs. no pharmacologic treatment (e.g., in patients with implanted defibrillators) whether sotalol response causes improved survival or identifies a population with a good prognosis.
Sotalol has not been shown to enhance survival in patients with ventricular arrhythmias.
14.2 Clinical Studies in Supra-Ventricular Arrhythmias with Oral Sotalol
Orally administered sotalol has been studied in patients with symptomatic AFIB/AFL in two principal studies, one in patients with primarily paroxysmal AFIB/AFL, the other in patients with primarily chronic AFIB.
In one study, a U.S. multicenter, randomized, placebo-controlled, double-blind, dose-response trial of patients with symptomatic primarily paroxysmal AFIB/AFL, three fixed dose levels of sotalol (80, 120, and 160 mg) twice daily and placebo were compared in 253 patients. In patients with reduced creatinine clearance (40-60 mL/min) the same doses were given once daily. Patients were not randomized for the following reasons: QT >450 ms; creatinine clearance <40 mL/min; intolerance to beta-blockers; bradycardia-tachycardia syndrome in the absence of an implanted pacemaker; AFIB/AFL was asymptomatic or was associated with syncope, embolic CVA or TIA; acute myocardial infarction within the previous 2 months; congestive heart failure; bronchial asthma or other contraindications to beta-blocker therapy; receiving potassium losing diuretics without potassium replacement or without concurrent use of ACE-inhibitors; uncorrected hypokalemia (serum potassium <3.5 mEq/L) or hypomagnesemia (serum magnesium <1.5 mEq/L); received chronic oral amiodarone therapy for >1 month within previous 12 weeks; congenital or acquired long QT syndromes; history of Torsade de Pointes with other antiarrhythmic agents which increase the duration of ventricular repolarization; sinus rate <50 bpm during waking hours; unstable angina pectoris; receiving treatment with other drugs that prolong the QT interval; and AFIB/AFL associated with the Wolff-Parkinson-White (WPW) syndrome. If the QT interval increased to ≥520 ms (or JT ≥430 ms if QRS >100 ms) the drug was discontinued. The patient population in this trial was 64% male, and the mean age was 62 years. No structural heart disease was present in 43% of the patients. Doses were administered once daily in 20% of the patients because of reduced creatinine clearance.
Sotalol was shown to prolong the time to the first symptomatic, ECG-documented recurrence of AFIB/AFL, as well as to reduce the risk of such recurrence at both 6 and 12 months. The 120 mg dose was more effective than 80 mg, but 160 mg did not appear to have an added benefit. Note that these doses were given twice or once daily, depending on renal function. The results are shown in Figure 1 and Tables 4 and 5.
Figure 1. Study 1—Time to First ECG-Documented Recurrence of Symptomatic AFIB/AFL Since Randomization
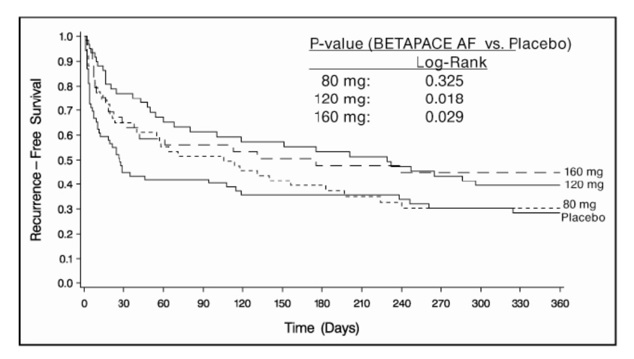
Table 4: Study 1 - Patient Status at 12 Months Placebo Oral Sotalol Dose 80 mg 120 mg 160 mg Randomized 69 59 63 62 On treatment in NSR at 12 months without recurrence * 23% 22% 29% 23% Recurrence *,† 67% 58% 49% 42% D/C for AEs 6% 12% 18% 29% * Symptomatic AFIB/AFL
† Efficacy endpoint of Study 1; study treatment stopped.Note that columns do not add up to 100% due to discontinuations (D/C) for other reasons.
Table 5: Study 1 - Median Time to Recurrence of Symptomatic AFIB/AFL and Relative Risk (vs. Placebo) at 12 Months Placebo Oral Sotalol Dose 80 mg 120 mg 160 mg p-value vs. placebo p=0.325 p=0.018 p=0.029 Relative Risk (RR) to placebo 0.81 0.59 0.59 Median Time to Recurrence (days) 27 106 229 175 Discontinuation because of adverse events was dose related.
In a second multicenter, randomized, placebo-controlled, double-blind study of 6 months duration in 232 patients with chronic AFIB, sotalol was titrated over a dose range from 80 mg/day to 320 mg/day. The patient population of this trial was 70% male with a mean age of 65 years. Structural heart disease was present in 49% of the patients. All patients had chronic AFIB for >2 weeks but <1 year at entry with a mean duration of 4.1 months. Patients were excluded if they had significant electrolyte imbalance, QTc >460 ms, QRS >140 ms, any degree of AV block or functioning pacemaker, uncompensated cardiac failure, asthma, significant renal disease (estimated creatinine clearance <50 mL/min), heart rate <50 bpm, myocardial infarction or open heart surgery in past 2 months, unstable angina, infective endocarditis, active pericarditis or myocarditis, ≥3 DC cardioversions in the past, medications that prolonged QT interval, and previous amiodarone treatment. After successful cardioversion patients were randomized to receive placebo (n=114) or sotalol (n=118), at a starting dose of 80 mg twice daily. If the initial dose was not tolerated it was decreased to 80 mg once daily, but if it was tolerated it was increased to 160 mg twice daily. During the maintenance period 67% of treated patients received a dose of 160 mg twice daily, and the remainder received doses of 80 mg once daily (17%) and 80 mg twice daily (16%).
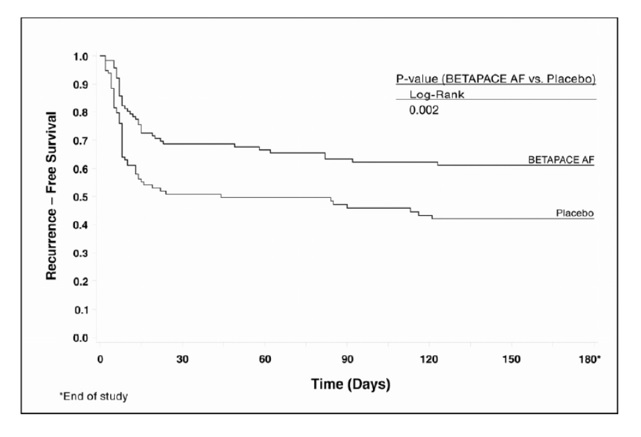
Figure 2 and Tables 6 and 7 show the results of the trial. There was a longer time to ECG-documented recurrence of AFIB and a reduced risk of recurrence at 6 months compared to placebo.
Table 6: Study 2 - Patient Status at 6 Months Randomized OralSotalol Placebo 118 114 On treatment in NSR at 6 months without recurrence * 45% 29% Recurrence *,† 49% 67% D/C for AEs 6% 3% Death 1% * Symptomatic or asymptomatic AFIB/AFL
† Efficacy endpoint of Study 2; study treatment stopped.Table 7: Study 2 - Median Time to Recurrence of Symptomatic AFIB/AFL/Death and Relative Risk (vs. Placebo) at 6 Months p-value vs. placebo Oral Sotalol Placebo p=0.002 Relative Risk (RR) to placebo Median time to recurrence (days) 0.55 >180 44
Figure 2. Study 2—Time to First ECG-Documented Recurrence of Symptomatic AFIB/AFL/Death Since Randomization
14.3 Clinical Studies in Patients with Myocardial Infarction
In a multicenter double-blind randomized study reported by Julian et al, the effect of sotalol 320 mg once daily was compared with that of placebo in 1456 patients (randomized 3:2, sotalol to placebo) surviving an acute myocardial infarction (MI). Treatment was started 5-14 days after infarction. Patients were followed for 12 months. The mortality rate was 7.3% in the sotalol group and 8.9% in the placebo group, not a statistically significant difference. Although the results do not show evidence of a benefit of sotalol in this population, they do not show an added risk in post MI patients receiving sotalol. There was, however, a suggestion of an early (i.e., first 10 days) excess mortality (3% on sotalol vs. 2% on placebo).
In a second small trial (n=17 randomized to sotalol) where sotalol was administered at high doses (e.g., 320 mg twice daily) to high-risk post-infarction patients (ejection fraction <40% and either >10 VPC/hr or VT on Holter), there were 4 fatalities and 3 serious hemodynamic/electrical adverse events within two weeks of initiating sotalol.
14.4 Intravenous Loading Dosage
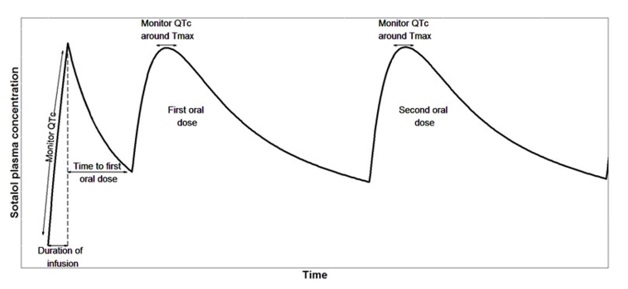
Computer-based simulations incorporating sotalol dose-exposure-QTc relationships were used to derive the intravenous loading doses. Based on these simulations, the intravenous loading dose in a typical patient across each of the renal function categories is expected to achieve steady state concentration faster compared to the conventional oral dosing. A cartoon showing the pharmacokinetic profile in a typical patient for an intravenous loading dose followed by subsequent oral doses is shown in Figure 3.
Figure 3: Illustrative pharmacokinetic profile for an intravenous loading dose followed by subsequent oral doses in a typical patient.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
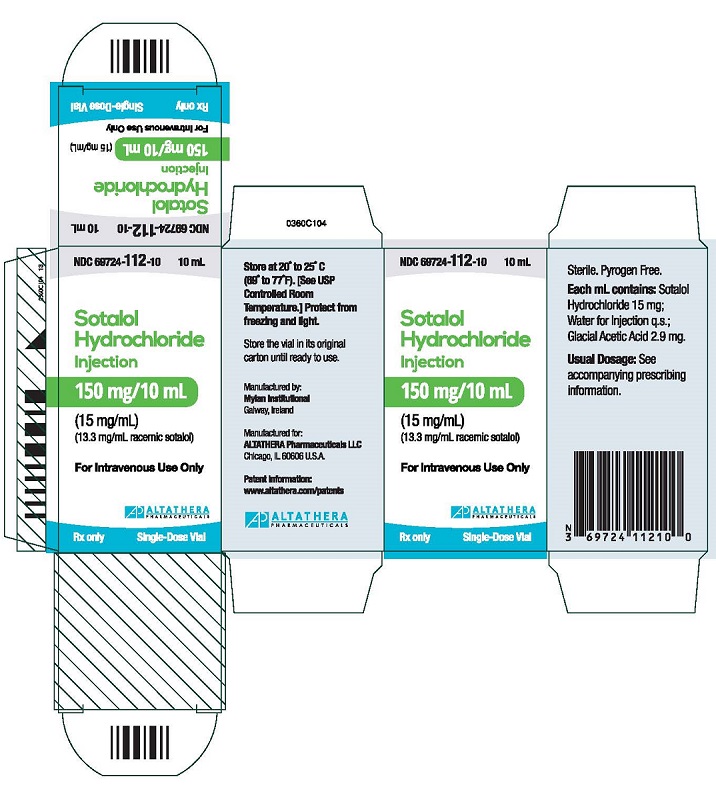
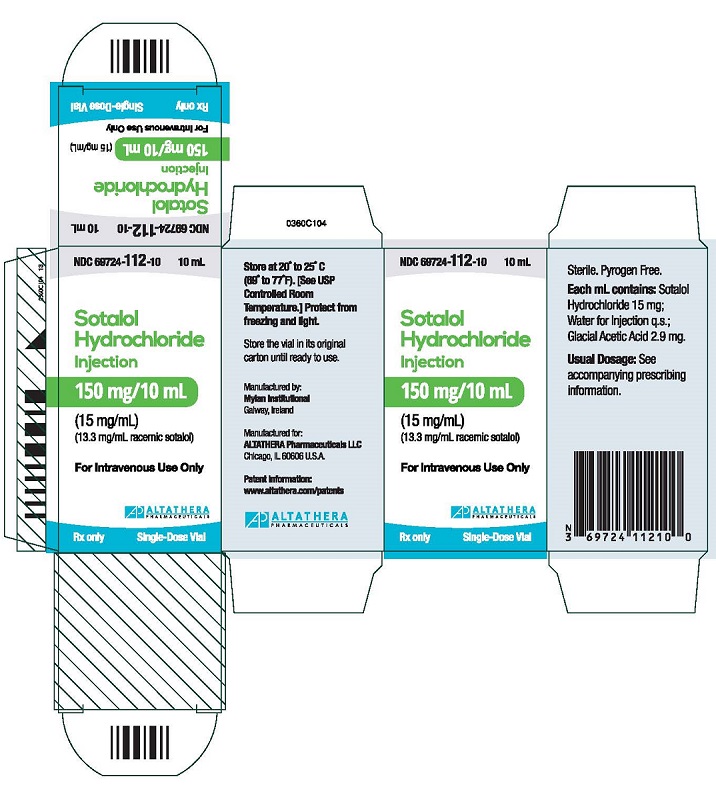
Sotalol injection is supplied in 10 mL single-dose vials, each containing 150 mg of sotalol hydrochloride as a clear solution (15 mg/mL).
NDC 69724-112-10
carton containing one 10 mL vialStore at 20° to 25°C (68° to 77°F); excursions permitted 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature.] Protect from freezing and light.
-
17 PATIENT COUNSELING INFORMATION
Risk of hypoglycemia
Inform parents or caregivers that there is a risk of hypoglycemia when sotalol is given to patients who are fasting or who are vomiting. Inform patients to notify their healthcare provider if they experience symptoms of hypoglycemia. [ See Warnings and Precautions ( 5.7) ].
Manufactured by:
Mylan Institutional
Galway, IrelandManufactured for:
ALTATHERA Pharmaceuticals LLC
Chicago, IL 60606 U.S.A. - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
SOTALOL HYDROCHLORIDE
sotalol hydrochloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:69724-112 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SOTALOL HYDROCHLORIDE (UNII: HEC37C70XX) (SOTALOL - UNII:A6D97U294I) SOTALOL HYDROCHLORIDE 15 mg in 1 mL Inactive Ingredients Ingredient Name Strength ACETIC ACID (UNII: Q40Q9N063P) 2.9 mg in 1 mL WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69724-112-10 1 in 1 CARTON 10/15/2015 1 10 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022306 10/15/2015 Labeler - ALTATHERA Pharmaceuticals, LLC (053682098) Establishment Name Address ID/FEI Business Operations Mylan Institutional 986298925 manufacture(69724-112)