Label: NYSTATIN ointment
- NDC Code(s): 33342-481-15, 33342-481-30
- Packager: Macleods Pharmaceuticals Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated January 29, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Nystatin is a polyene antifungal antibiotic drug obtained from Streptomyces nursei
Nystatin Ointment USP, for topical use only, contains 100,000 USP Nystatin Units per gram, in an ointment base of light mineral oil and white petrolatum. The structural formula is as follows:
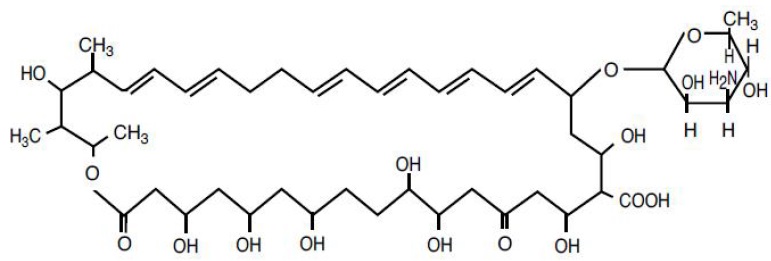
Molecular Weight 926.13
Molecular Formula C47H75NO17
-
CLINICAL PHARMACOLOGY
Pharmacokinetics
Nystatin is not absorbed from intact skin or mucous membrane.
Microbiology
Nystatin is an antibiotic which is both fungistatic and fungicidal in vitro against a wide variety of yeasts and yeast-like fungi, including Candida albicans, C. parapsilosis, C. tropicalis, C. guilliermondi, C. pseudotropicalis, C. krusei, Torulopsis glabrata, Tricophyton rubrum, T. mentagrophytes.
Nystatin acts by binding to sterols in the cell membrane of susceptible species resulting in a change in membrane permeability and the subsequent leakage of intracellular components. On repeated subculturing with increasing concentrations of nystatin, Candida albicans does not develop resistance to nystatin.Generally, resistance to nystatin does not develop during therapy. However, other species of 47 75 17 Candida (C. tropicalis, C. guilliermondi, C. krusei, and C. stellatoides) become quite resistant on treatment with nystatin and simultaneously become cross resistant to amphotericin as well. This resistance is lost when the antibiotic is removed.
Nystatin exhibits no appreciable activity against bacteria, protozoa, or viruses. - INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
PRECAUTIONS
General
Nystatin Ointment should not be used for the treatment of systemic, oral, intravaginal or ophthalmic infections.
If irritation or sensitization develops, treatment should be discontinued and appropriate measures taken as indicated. It is recommended that KOH smears, cultures, or other diagnostic methods be used to confirm the diagnosis of cutaneous or mucocutaneous candidiasis and to rule out infection caused by other pathogens.INFORMATION FOR THE PATIENTS
Patients using these medications should receive the following information and instructions :
1. The patient should be instructed to use these medications as directed (including the replacement of missed doses). These medications are not for any disorder other than that for which they are prescribed.
2. Even if symptomatic relief occurs within the first few days of treatment, the patient should be advised not to interrupt or discontinue therapy until the prescribed course of treatment is completed.
3. If symptoms of irritation develop, the patient should be advised to notify the physician promptly.Laboratory Tests
If there is a lack of therapeutic response, KOH smears, cultures, or other diagnostic methods should be repeated.
Carcinogenisis, Mutagenisis, Impairment of Fertility
No long-term animal studies have been performed to evaluate the carcinogenic potential of nystatin. No studies have been performed to determine the mutagenicity of nystatin or its effects on male or female fertility.
Pregnancy: Teratogenic Effects
Animal reproduction studies have not been conducted with any nystatin topical preparation. It also is not known whether these preparations can cause fetal harm when used by a pregnant woman or can affect reproductive capacity. Nystatin topical preparations should be prescribed for a pregnant woman only if the potential benefit to the mother outweighs the potential risk to the fetus.
-
ADVERSE REACTIONS
The frequency of adverse events reported in patients using Nystatin Ointment preparations is less than 0.1%. The more common events that were reported include allergic reactions, burning, itching, rash, eczema, and pain on application.
(SeePRECAUTIONS: General.)
To report SUSPECTED ADVERSE REACTIONS, contact Macleods Pharma USA, Inc., at 1-888-943-3210 or 1-855-926-338 or FDA at 1-800-FDA-1088 (www.fda.gov/medwatch). - DOSAGE & ADMINISTRATION
-
HOW SUPPLIED
Nystatin Ointment (100,000 USP Nystatin Units per gram) is a yellowish ointment available as follows:
NDC 33342-481-15 15 gram tube
NDC 33342-481-30 30 gram tube
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Manufactured for:
Macleods Pharma USA, Inc.Princeton, NJ 08540
Manufacturer:
Macleods Pharmaceuticals Limited
At Oxalis Labs
Baddi, Himachal Pradesh, INDIARev. 01/2021
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Nystatin Ointment (100,000 USP Nystatin Units per gram)
Pack Count: 15 g Tube
NDC 33342-481-15

Nystatin Ointment (100,000 USP Nystatin Units per gram)
Pack Count: 15 g Carton
NDC 33342-481-15

Nystatin Ointment (100,000 USP Nystatin Units per gram)
Pack Count: 30 g Tube
NDC 33342-481-30

Nystatin Ointment (100,000 USP Nystatin Units per gram)
Pack Count: 30 g Carton
NDC 33342-481-30
-
INGREDIENTS AND APPEARANCE
NYSTATIN
nystatin ointmentProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:33342-481 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NYSTATIN (UNII: BDF1O1C72E) (NYSTATIN - UNII:BDF1O1C72E) NYSTATIN 100000 in 1 g Inactive Ingredients Ingredient Name Strength LIGHT MINERAL OIL (UNII: N6K5787QVP) PETROLATUM (UNII: 4T6H12BN9U) Product Characteristics Color YELLOW Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:33342-481-15 1 in 1 CARTON 01/29/2021 1 15 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:33342-481-30 1 in 1 CARTON 01/29/2021 2 30 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA213826 01/29/2021 Labeler - Macleods Pharmaceuticals Limited (862128535) Establishment Name Address ID/FEI Business Operations OXALIS LABS 860120472 ANALYSIS(33342-481) , LABEL(33342-481) , MANUFACTURE(33342-481) , PACK(33342-481)




