Label: AGGRASTAT- tirofiban injection, solution
- NDC Code(s): 25208-001-04, 25208-002-01, 25208-002-02, 25208-002-03
- Packager: Medicure International Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated January 1, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use AGGRASTAT® (tirofiban hydrochloride) safely and effectively. See full prescribing information for AGGRASTAT.
AGGRASTAT (tirofiban hydrochloride) injection, for intravenous use
Initial U.S. Approval: 1998INDICATIONS AND USAGE
AGGRASTAT is a platelet aggregation inhibitor indicated to reduce the rate of thrombotic cardiovascular events (combined endpoint of death, myocardial infarction, or refractory ischemia/repeat cardiac procedure) in patients with non-ST elevation acute coronary syndrome (NSTE-ACS). (1)
DOSAGE AND ADMINISTRATION
- Administer intravenously 25 mcg/kg within 5 minutes and then 0.15 mcg/kg/min for up to 18 hours. In patients with creatinine clearance ≤60 mL/min, give 25 mcg/kg within 5 minutes and then 0.075 mcg/kg/min. (2)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
Bleeding is the most commonly reported adverse reaction. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Medicure at 1-800-509-0544 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Coadministration of fibrinolytics, anticoagulants and antiplatelet agents, increases the risk of bleeding. (7)
USE IN SPECIFIC POPULATIONS
- Renal Insufficiency: Reduce the dose in patients with severe renal insufficiency. (8.6)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 1/2025
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Administration
2.3 Dose Adjustment for Renal Impairment
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 General Risk of Bleeding
5.2 Thrombocytopenia
6 ADVERSE REACTIONS
6.1 Clinical Trial Experience
6.2 Post-Marketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Insufficiency
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dosage is 25 mcg/kg administered intravenously within 5 minutes and then 0.15 mcg/kg/min for up to 18 hours.
2.2 Administration
For intravenous use only. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
To open the 100 mL or 250 mL premixed bag, first tear off its foil overpouch. The plastic may be somewhat opaque because of moisture absorption during sterilization; the opacity will diminish gradually. Check for leaks by squeezing the inner bag firmly; if any leaks are found or sterility is suspect then the solution should be discarded. Do not use unless the solution is clear and the seal is intact.
Administration Instructions
- Withdraw the bolus dose of AGGRASTAT from the 15 mL premixed bolus vial into a syringe. Alternatively, the bolus dose of AGGRASTAT may be administered from the 100 mL or 250 mL premixed bag. Do not dilute. Administer the bolus dose within 5 minutes via a syringe or IV pump. For patients ≥ 167 kg, it is recommended that the bolus dose be administered via syringe from the 15 mL premixed bolus vial, to ensure that delivery time does not exceed 5 minutes.
- Immediately following the bolus dose administration, administer the maintenance infusion from the 100 mL premixed bag or the 250 mL premixed bag via an IV pump.
- Discard any unused portion left in the vial or bag.
The recommended bolus volume using the 15 mL premixed bolus vial can be calculated using the following equation:
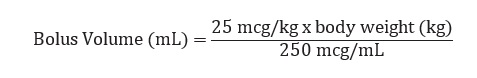
The recommended bolus volume using the 100 mL premixed bag or 250 mL premixed bag can be calculated using the following equation:
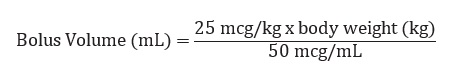
The recommended infusion rate for patients with CrCl (Creatinine Clearance) >60 mL/min using the 100 mL premixed bag or 250 mL premixed bag can be calculated using the following equation:
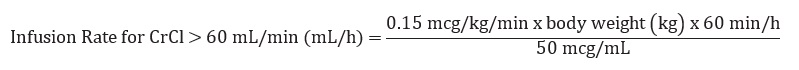
Example calculation of infusion rate for 60 kg patient with CrCl >60 mL/min using the 100 mL premixed bag or 250 mL premixed bag:
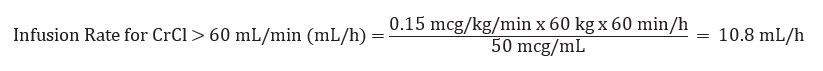
Drug Compatibilities
AGGRASTAT can be administered in the same intravenous line as heparin, atropine sulfate, dobutamine, dopamine, epinephrine hydrochloride (HCl), famotidine injection, furosemide, lidocaine, midazolam HCl, morphine sulfate, nitroglycerin, potassium chloride, and propranolol HCl. Do not administer AGGRASTAT through the same IV line as diazepam. Do not add other drugs or remove solution directly from the bag with a syringe.2.3 Dose Adjustment for Renal Impairment
The recommended dosage in patients with CrCl ≤60 mL/min (calculated using the Cockcroft-Gault equation with actual body weight) is 25 mcg/kg intravenously within 5 minutes and then 0.075 mcg/kg/min, for up to 18 hours.
The recommended infusion rate for patients with CrCl ≤ 60 mL/min using 100 mL premixed bag or 250 mL premixed bag can be calculated using the following equation:
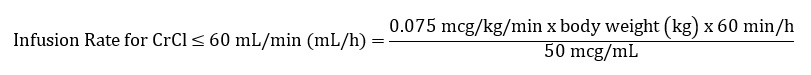
-
3 DOSAGE FORMS AND STRENGTHS
AGGRASTAT is a clear, non-preserved, colorless, isosmotic, sterile premixed injection with sodium chloride for tonicity adjustment available in the following presentations:
Table 1 AGGRASTAT Strength and Packaging Strength Volume - Packaging 50 mcg/mL 250 mL - bag 50 mcg/mL 100 mL - bag 250 mcg/mL 15 mL - bolus vial -
4 CONTRAINDICATIONS
AGGRASTAT is contraindicated in patients with:
- Severe hypersensitivity reaction to AGGRASTAT (i.e., anaphylactic reactions) [see Adverse Reactions (6.2)].
- A history of thrombocytopenia following prior exposure to AGGRASTAT [see Adverse Reactions (6.1)].
- Active internal bleeding or a history of bleeding diathesis, major surgical procedure or severe physical trauma within the previous month [see Adverse Reactions (6.1)].
-
5 WARNINGS AND PRECAUTIONS
5.1 General Risk of Bleeding
Bleeding is the most common complication encountered during therapy with AGGRASTAT. Most bleeding associated with AGGRASTAT occurs at the arterial access site for cardiac catheterization. Minimize the use of traumatic or potentially traumatic procedures such as arterial and venous punctures, intramuscular injections, nasotracheal intubation, etc.
Concomitant use of fibrinolytics, anticoagulants and antiplatelet drugs increases the risk of bleeding.
5.2 Thrombocytopenia
Profound thrombocytopenia has been reported with AGGRASTAT. Monitor platelet counts beginning about 6 hours after treatment initiation and daily thereafter. If the platelet count decreases to <90,000/mm3, monitor platelet counts to exclude pseudothrombocytopenia. If thrombocytopenia is confirmed, discontinue AGGRASTAT and heparin. Previous exposure to a glycoprotein (GP) IIb/IIIa receptor antagonist may increase the risk of developing thrombocytopenia [see Adverse Reactions (6.1)].
-
6 ADVERSE REACTIONS
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In the PRISM (Platelet Receptor Inhibition for Ischemic Syndrome Management), PRISM-PLUS (Platelet Receptor Inhibition for Ischemic Syndrome Management — Patients Limited by Unstable Signs and Symptoms) and RESTORE (Randomized Efficacy Study of Tirofiban for Outcomes and Restenosis) trials, 1946 patients received AGGRASTAT in combination with heparin and 2002 patients received AGGRASTAT alone for about 3 days. Forty-three percent of the population was >65 years of age and approximately 30% of patients were female. In clinical studies with the recommended regimen (25 mcg/kg bolus followed by a 0.15 mcg/kg/min maintenance infusion), AGGRASTAT was administered in combination with aspirin, clopidogrel and heparin or bivalirudin to over 8000 patients for typically ≤24 hours. Approximately 30% of the population was >65 years of age and approximately 25% were female.
Bleeding
PRISM-PLUS Regimen
The incidences of major and minor bleeding using the TIMI criteria in the PRISM-PLUS study are shown below.
Table 2 TIMI Major and Minor Bleeding in PRISM-PLUS - *
- Major = Hemoglobin drop of >5.0 g/dL with or without an identified site, intracranial hemorrhage, or cardiac tamponade.
- †
- Minor = Hemoglobin drop of >3.0 g/dL with bleeding from a known site, spontaneous gross hematuria, hematemesis or hemoptysis.
- ‡
- 0.4 mcg/kg/min initial infusion; 0.10 mcg/kg/min maintenance infusion.
PRISM-PLUS
(NSTE-ACS)
Bleeding
AGGRASTAT‡+ Heparin
(n=773)
Heparin alone
(n=797)
Major Bleeding
1.4%
0.8%
Minor Bleeding
10.5%
8.0%
Transfusions
4.0%
2.8%
The incidence rates of TIMI major bleeding in patients undergoing percutaneous procedures in PRISM-PLUS are shown below.
Table 3 TIMI Major Bleeding Associated with Percutaneous Procedures in PRISM-PLUS AGGRASTAT +
Heparin
Heparin alone
N
%
N
%
Prior to Procedures
773
0.3
797
0.1
Following Angiography
697
1.3
708
0.7
Following PTCA
239
2.5
236
2.2
The incidence rates of TIMI major bleeding in patients undergoing coronary artery bypass graft surgery (CABG) in PRISM-PLUS within one day of discontinuation of AGGRASTAT were 17% on AGGRASTAT plus heparin (N=29) and 35% on heparin alone (N=31).
Recommended (“High-Dose Bolus”) Regimen
Rates of major bleeds (including any intracranial, intraocular or retroperitoneal hemorrhage, clinically overt signs of hemorrhage associated with a drop in hemoglobin of >3 g/dL or any drop in hemoglobin by 4 g/dL, bleeding requiring transfusion of ≥ 2 U blood products, bleeding directly resulting in death within 7 days or hemodynamic compromise requiring intervention) were consistent with the rates observed in subjects administered the PRISM-PLUS regimen of AGGRASTAT. There was a trend toward greater bleeding in ST segment elevation myocardial infarction (STEMI) patients treated with fibrinolytics prior to administration of AGGRASTAT using the recommended regimen during rescue PCI.
Non-Bleeding
The incidences of non-bleeding adverse events that occurred at an incidence of >1% and numerically higher than control, regardless of drug relationship, are shown below:
Table 4 Non-bleeding Adverse Reactions in PRISM-PLUS AGGRASTAT + Heparin
(N=1953)
%
Heparin alone
(N=1887)
%
Body as a Whole
Edema/swelling
2
1
Pain, pelvic
6
5
Reaction, vasovagal
2
1
Cardiovascular System
Bradycardia
4
3
Dissection, coronary artery
5
4
Musculoskeletal System
Pain, leg
3
2
Nervous System/Psychiatric
Dizziness
3
2
Skin and Skin Appendage
Sweating
2
1
Thrombocytopenia
Patients treated with AGGRASTAT plus heparin, were more likely to experience decreases in platelet counts than were those on heparin alone. These decreases were reversible upon discontinuation of AGGRASTAT. The percentage of patients with a decrease of platelets to <90,000/mm3 was 1.5%, compared with 0.6% in the patients who received heparin alone. The percentage of patients with a decrease of platelets to <50,000/mm3 was 0.3%, compared with 0.1% of the patients who received heparin alone.
6.2 Post-Marketing Experience
The following additional adverse reactions have been identified during post-approval use of AGGRASTAT. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to the drug exposure.
Hypersensitivity: Severe allergic reactions including anaphylactic reactions have occurred during the first day of AGGRASTAT infusion, during initial treatment, and during readministration of AGGRASTAT. Some cases have been associated with severe thrombocytopenia (platelet counts <10,000/mm3). No information is available on the formation of antibodies to tirofiban.
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
While published data cannot definitively establish the absence of risk, available published case reports have not established an association with tirofiban use during pregnancy and major birth defects, miscarriage, or adverse maternal or fetal outcomes. Untreated myocardial infarction can be fatal to the pregnant woman and fetus (see Clinical Considerations). Studies with tirofiban HCl at intravenous doses up to 5 mg/kg/day (about 5 and 13 times the maximum recommended daily human dose for rat and rabbit, respectively, when compared on a body surface area basis) have revealed no harm to the fetus.The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2–4% and 15–20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk
Myocardial infarction is a medical emergency in pregnancy which can be fatal to the pregnant woman and fetus if left untreated.Data
Animal Data
There was no evidence of maternal or developmental toxicity in any of the studies in Table 5.Table 5 Developmental Toxicity Studies - *
- 5 mg/kg/day is ~5 and 13 times the maximum recommended daily human dose for rat and rabbit, respectively, when compared on a body surface area basis.
Type of Study
Species
Dose/Exposure*
Duration/Timing Exposure
(1) Range-finding
Rat
(N=30)
1, 2, 5 mg/kg/day IV
(N=10 per group)
Once daily from GD 6 through LD 20
(2) Developmental Toxicity
Rat
(N=66)
1, 2, 5 mg/kg/day IV
(N=22 per group)
Once daily from GD 6 through GD 20
(3) Developmental Toxicity with Postweaning Evaluation
Rat
(N=66)
1, 2, 5 mg/kg/day IV
(N=22 per group)
Once daily from GD 6 through LD 20
(4) Range-finding (non-pregnant)
Rabbit
(N=21)
1, 2, 5 mg/kg/day IV
(N=7 per group)
Once daily for 14 days
(5) Range-finding (pregnant)
Rabbit (N=30)
1, 2, 5 mg/kg/day IV
(N=10 per group)
Once daily from GD 7 through GD 20
(6) Developmental Toxicity
Rabbit
(N=60)
1, 2, 5 mg/kg/day
(N=20 per group) IV
Once daily from GD 7 through GD 20
8.2 Lactation
Risk Summary
There is no data on the presence of tirofiban in human milk, the effects of the drug on the breastfed infant, or the effects of the drug on human milk production. However, tirofiban is present in rat milk. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for AGGRASTAT and any potential adverse effects on the breastfed child from AGGRASTAT or from the underlying maternal condition.8.5 Geriatric Use
Of the total number of patients in controlled clinical studies of AGGRASTAT, 43% were 65 years and over, while 12% were 75 years and over. With respect to efficacy, the effect of AGGRASTAT in the elderly (≥65 years) appeared similar to that seen in younger patients (<65 years). Elderly patients receiving AGGRASTAT with heparin or heparin alone had a higher incidence of bleeding complications than did younger patients, but the incremental risk of bleeding in patients treated with AGGRASTAT in combination with heparin compared to the risk in patients treated with heparin alone was similar regardless of age. No dose adjustment is recommended for the elderly population [see Dosage and Administration (2)].
8.6 Renal Insufficiency
Patients with moderate to severe renal insufficiency have decreased plasma clearance of AGGRASTAT. Reduce the dosage of AGGRASTAT in patients with severe renal insufficiency [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
Safety and efficacy of AGGRASTAT has not been established in patients on hemodialysis.
-
10 OVERDOSAGE
In clinical trials, inadvertent overdosage with AGGRASTAT occurred in doses up to 2 times the recommended dose for initial infusion doses. Inadvertent overdosage occurred in doses up to 9.8 times the 0.15 mcg/kg/min maintenance infusion rate.
The most frequently reported manifestation of overdosage was bleeding, primarily minor mucocutaneous bleeding events and minor bleeding at the sites of cardiac catheterization [see Warnings and Precautions (5.1)].
Overdosage of AGGRASTAT should be treated by assessment of the patient’s clinical condition and cessation or adjustment of the drug infusion as appropriate.
AGGRASTAT can be removed by hemodialysis.
-
11 DESCRIPTION
AGGRASTAT contains tirofiban hydrochloride, a non-peptide antagonist of the platelet GP IIb/IIIa receptor, which inhibits platelet aggregation.
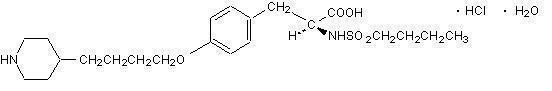
Tirofiban hydrochloride monohydrate is chemically described as N-(butylsulfonyl)-O-[4-(4-piperidinyl)butyl]-L-tyrosine monohydrochloride monohydrate.
Its molecular formula is C22H36N2O5S•HCl•H2O and its structural formula is:
Tirofiban hydrochloride monohydrate is a white to off-white, non-hygroscopic, free-flowing powder, with a molecular weight of 495.08. It is very slightly soluble in water.
AGGRASTAT Injection Premixed is supplied as a sterile solution in water for injection, for intravenous use. The pH of the solution ranges from 5.5 to 6.5 adjusted with hydrochloric acid and/or sodium hydroxide.
Each 100 mL of the premixed, isosmotic intravenous injection contains 5.618 mg tirofiban hydrochloride monohydrate equivalent to 5 mg tirofiban (50 mcg/mL) and the following inactive ingredients: 0.9 g sodium chloride, 54 mg sodium citrate dihydrate, and 3.2 mg citric acid anhydrous.
Each 250 mL of the premixed, isosmotic intravenous injection contains 14.045 mg tirofiban hydrochloride monohydrate equivalent to 12.5 mg tirofiban (50 mcg/mL) and the following inactive ingredients: 2.25 g sodium chloride, 135 mg sodium citrate dihydrate, and 8 mg citric acid anhydrous.
AGGRASTAT Injection Premixed Bolus Vial is supplied as a sterile, isosmotic, concentrated solution for intravenous bolus injection, in 15 mL vials. No dilution is required. Each 15 mL of the premixed, isosmotic intravenous injection bolus vial contains 4.215 mg of tirofiban hydrochloride monohydrate equivalent to 3.75 mg of tirofiban and the following inactive ingredients: 120 mg sodium chloride, 40.5 mg sodium citrate dihydrate, and 2.4 mg citric acid anhydrous and water for injection.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
AGGRASTAT is a reversible antagonist of fibrinogen binding to the GP IIb/IIIa receptor, the major platelet surface receptor involved in platelet aggregation. When administered intravenously, AGGRASTAT inhibits ex vivo platelet aggregation in a dose- and concentration-dependent manner.
When given according to the PRISM-PLUS regimen of 0.4 mcg/kg/min over 30 minutes followed by a 0.1 mcg/kg/min maintenance infusion, >90% inhibition of platelet aggregation is attained by the end of the 30-minute infusion. When given according to the recommended regimen of 25 mcg/kg followed by a 0.15 mcg/kg/min maintenance infusion, >90% inhibition of platelet aggregation is attained within 10 minutes. Platelet aggregation inhibition is reversible following cessation of the infusion of AGGRASTAT.
12.2 Pharmacodynamics
AGGRASTAT inhibits platelet function, as demonstrated by its ability to inhibit ex vivo adenosine phosphate (ADP)-induced platelet aggregation and prolong bleeding time in healthy subjects and patients with coronary artery disease. The time course of inhibition parallels the plasma concentration profile of the drug.
Following discontinuation of an infusion of AGGRASTAT 0.10 mcg/kg/min, ex vivo platelet aggregation returns to near baseline in 4 to 8 hours in approximately 90% of patients with coronary artery disease. The addition of heparin to this regimen does not significantly alter the percentage of subjects with >70% inhibition of platelet aggregation (IPA), but does increase the average bleeding time, as well as the number of patients with bleeding times prolonged to >30 minutes. Similar platelet aggregation recovery rates are observed following discontinuation of a 0.15 mcg/kg/min infusion.
12.3 Pharmacokinetics
Tirofiban has a half-life of approximately 2 hours. It is cleared from the plasma largely by renal excretion, with about 65% of an administered dose appearing in urine and about 25% in feces, both largely as unchanged tirofiban. Metabolism appears to be limited.
Tirofiban is not highly bound to plasma proteins and protein binding is concentration independent over the range of 0.01 to 25 mcg/mL. The unbound fraction in human plasma is 35%. The steady state volume of distribution of tirofiban ranges from 22 to 42 liters.
In healthy subjects, the plasma clearance of tirofiban ranges from 213 to 314 mL/min. Renal clearance accounts for 39 to 69% of plasma clearance.
Specific Populations
There is no effect on clearance of tirofiban by sex, race, age, or hepatic impairment.
Renal Insufficiency
Plasma clearance of tirofiban is decreased about 40% in subjects with creatinine clearance <60 mL/min and >50% in patients with creatinine clearance <30 mL/min, including patients requiring hemodialysis [see Dosage and Administration (2.3)]. Tirofiban is removed by hemodialysis.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic potential of AGGRASTAT has not been evaluated.
Tirofiban HCl was negative in the in vitro microbial mutagenesis and V-79 mammalian cell mutagenesis assays. In addition, there was no evidence of direct genotoxicity in the in vitro alkaline elution and in vitro chromosomal aberration assays. There was no induction of chromosomal aberrations in bone marrow cells of male mice after the administration of intravenous doses up to 5 mg tirofiban/kg (about 3 times the maximum recommended daily human dose when compared on a body surface area basis).
Fertility and reproductive performance were not affected in studies with male and female rats given intravenous doses of tirofiban up to 5 mg/kg/day (about 5 times the maximum recommended daily human dose when compared on a body surface area basis).
-
14 CLINICAL STUDIES
Two large-scale clinical studies established the efficacy of AGGRASTAT in the treatment of patients with NSTE-ACS (unstable angina/non-ST elevation MI). The two studies examined AGGRASTAT alone and added to heparin, prior to and after percutaneous coronary revascularization (if indicated) (PRISM-PLUS) and in comparison to heparin in a similar population (PRISM). These trials are discussed in detail below.
PRISM-PLUS (Platelet Receptor Inhibition for Ischemic Syndrome Management — Patients Limited by Unstable Signs and Symptoms)
In the double-blind PRISM-PLUS trial, 1570 patients with documented NSTE-ACS within 12 hours of entry into the study were randomized to AGGRASTAT (30 minute initial infusion of 0.4 mcg/kg/min followed by a maintenance infusion of 0.10 mcg/kg/min) in combination with heparin (bolus of 5,000 U followed by an infusion of 1,000 U/h titrated to maintain an activated partial thromboplastin time (APTT) of approximately 2 times control) or to heparin alone. All patients received concomitant aspirin unless contraindicated. Patients who were medically managed or who underwent revascularization procedures were studied. Patients underwent 48 hours of medical stabilization on study drug therapy, and they were to undergo angiography before 96 hours (and, if indicated, angioplasty/atherectomy, while continuing on AGGRASTAT and heparin for 12-24 hours after the procedure). AGGRASTAT and heparin could be continued for up to 108 hours. Exclusions included contraindications to anticoagulation, decompensated heart failure, platelet count <150,000/mm3, and serum creatinine >2.5 mg/dL. The mean age of the population was 63 years; 32% of patients were female and approximately half of the population presented with non-ST elevation myocardial infarction. On average, patients received AGGRASTAT for 71 hours.
A third group of patients was initially randomized to AGGRASTAT alone (no heparin). This arm was stopped when the group was found, at an interim look, to have greater mortality than the other two groups.
The primary endpoint of the study was a composite of refractory ischemia, new MI and death within 7 days. There was a 32% risk reduction in the overall composite primary endpoint. The components of the composite were examined separately and the results are shown in Table 6. Note that the sum of the individual components may be greater than the composite (if a patient experiences multiple component events only one event counts towards the composite).
Table 6 Primary Outcomes at 7 days in PRISM-PLUS Endpoint
AGGRASTAT+
Heparin
(n=773)
Heparin
(n=797)
Risk Reduction
p-value
Death, new MI, and refractory ischemia at 7 days
12.9%
17.9%
32%
0.004
Death
1.9%
1.9%
---
---
MI
3.9%
7.0%
47%
0.006
Refractory Ischemia
9.3%
12.7%
30%
0.023
The benefit seen at 7 days was maintained over time. The risk reduction in the composite endpoint at 30 days and 6 months is shown in the Kaplan-Meier curve below.
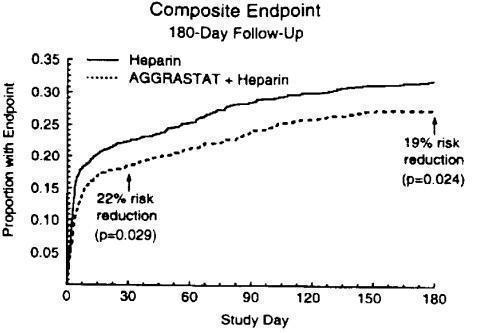
Figure 1. Time to first event of death, new MI, or refractory ischemia in PRISM-PLUS
An analysis of the results by sex suggests that women who are medically managed or who undergo subsequent percutaneous transluminal coronary angioplasty (PTCA)/atherectomy may receive less benefit from AGGRASTAT (95% confidence limits for relative risk of 0.61-1.74) than do men (0.43-0.89) (p=0.11). This difference may be a true treatment difference, the effect of other differences in these subgroups, or a chance occurrence.
Approximately 90% of patients in the PRISM-PLUS study underwent coronary angiography and 30% underwent angioplasty/atherectomy during the first 30 days of the study. The majority of these patients continued on study drug throughout these procedures. AGGRASTAT was continued for 12-24 hours (average 15 hours) after angioplasty/atherectomy. The effects of AGGRASTAT at Day 30 did not appear to differ among sub-populations that did or did not receive PTCA or CABG, both prior to and after the procedure.
PRISM (Platelet Receptor Inhibition for Ischemic Syndrome Management)
In the PRISM study, a randomized, parallel, double-blind study, 3232 patients with NSTE-ACS intended to be managed without coronary intervention were randomized to AGGRASTAT (initial dose of 0.6 mcg/kg/min for 30 minutes followed by 0.15 mcg/kg/min for 47.5 hours) or heparin (5000-unit intravenous bolus followed by an infusion of 1000 U/h for 48 hours). The mean age of the population was 62 years; 32% of the population was female and 25% had non-ST elevation MI on presentation. Thirty percent had no ECG evidence of cardiac ischemia. Exclusion criteria were similar to PRISM-PLUS. The primary endpoint was the composite endpoint of refractory ischemia, MI or death at the end of the 48-hour drug infusion. The results are shown in Table 7.
Table 7 Primary Outcomes in PRISM – Cardiac Ischemia Events Composite Endpoint (death, MI, or refractory ischemia)
AGGRASTAT
(n=1616)
Heparin
(n=1616)
Risk Reduction
p-value
2 Days (end of drug infusion)
3.8%
5.6%
33%
0.015
7 Days
10.3%
11.3%
10%
0.33
In the PRISM study, no adverse effect of AGGRASTAT on mortality at either 7 or 30 days was detected. This result is different from that in the PRISM-PLUS study, where the arm that included AGGRASTAT without heparin (N=345) was dropped at an interim analysis by the Data Safety Monitoring Committee for increased mortality at 7 days.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
AGGRASTAT is supplied as a clear, non-preserved, colorless, isosmotic, sterile premixed solution with sodium chloride for tonicity adjustment.
Table 8 AGGRASTAT Product Details Strength Total Amount Packaging NDC # 50 mcg/mL 12.5 mg/250 mL bag 25208-002-02 50 mcg/mL 5 mg/100 mL bag 25208-002-01 250 mcg/mL 3.75 mg/15 mL bolus vial 25208-001-04 FOR INTRAVENOUS USE ONLY
Store AGGRASTAT at controlled room temperature, 25°C (77°F) with excursions permitted between 15-30°C (59-86°F) [see USP Controlled Room Temperature]. Do not freeze. Protect from light during storage.
-
17 PATIENT COUNSELING INFORMATION
Advise patients to watch closely for any signs of bleeding or bruising and to report these to their health care provider when they occur.
Advise patients to discuss with their health care provider their use of any other medications, including over-the-counter or herbal products prior to AGGRASTAT use.
Patent: www.medicure.com/aggrastat/patents
AGGRASTAT is manufactured for:
MEDICURE INTERNATIONAL, INC.
by:
EMERGENT BIOSOLUTIONS
Baltimore, Maryland 21230 USA
And
LABORATORIOS GRIFOLS S.A
08150 Parets del Vallès (Barcelona), Spain
Distributed by:
MEDICURE PHARMA, INC.
Princeton, NJ 08540 USA
1-800-509-0544PIM-11
* Registered trademark of Medicure International, Inc.
© 2024
Copyright used under license.
All rights reserved. - PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- Carton 3.75 mg/15 mL - Bolus vial
- Label 3.75 mg/15 mL - Bolus vial
- Carton 5 mg/100 mL - Bag
- Pouch 5 mg/100 mL - Bag
- Label 5 mg/100 mL - Bag
-
INGREDIENTS AND APPEARANCE
AGGRASTAT
tirofiban injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:25208-002 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TIROFIBAN HYDROCHLORIDE (UNII: 6H925F8O5J) (TIROFIBAN - UNII:GGX234SI5H) TIROFIBAN 5 mg in 100 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) 3.2 mg in 100 mL TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) 54 mg in 100 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 900 mg in 100 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:25208-002-02 1 in 1 CARTON 05/14/1998 1 250 mL in 1 CONTAINER; Type 0: Not a Combination Product 2 NDC:25208-002-03 1 in 1 CARTON 10/15/2016 07/30/2024 2 100 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product 3 NDC:25208-002-01 1 in 1 CARTON 12/24/2020 3 100 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020913 05/14/1998 AGGRASTAT
tirofiban injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:25208-001 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TIROFIBAN HYDROCHLORIDE (UNII: 6H925F8O5J) (TIROFIBAN - UNII:GGX234SI5H) TIROFIBAN 3.75 mg in 15 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) 2.4 mg in 15 mL TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) 40.5 mg in 15 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 120 mg in 15 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:25208-001-04 1 in 1 CARTON 10/15/2016 1 15 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020912 10/15/2016 Labeler - Medicure International Inc (860240324) Registrant - Medicure International Inc (860240324) Establishment Name Address ID/FEI Business Operations Laboratorios Grifols, S.A. 463719725 manufacture(25208-002, 25208-001) , label(25208-002, 25208-001) , pack(25208-002, 25208-001)
















