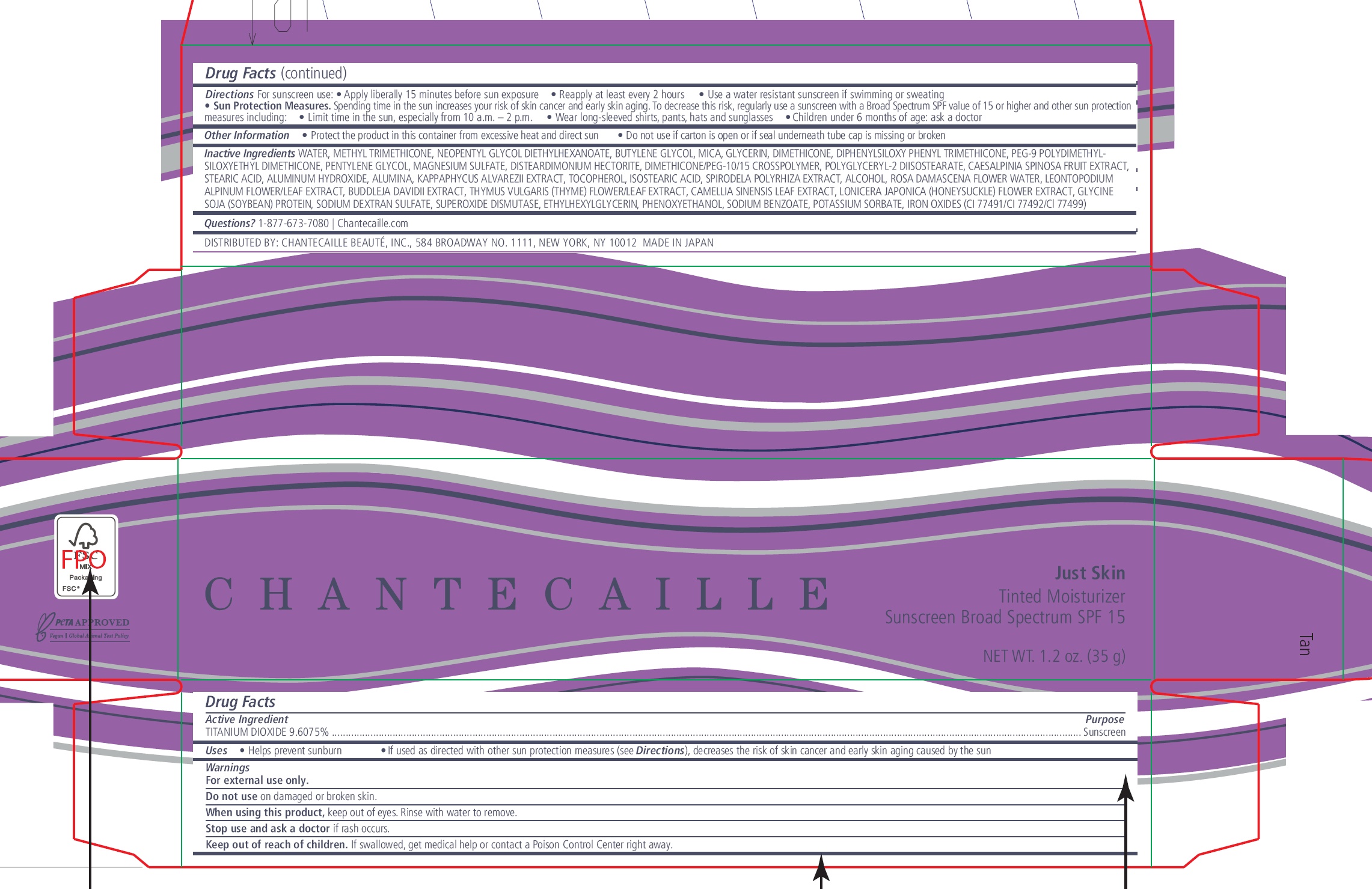
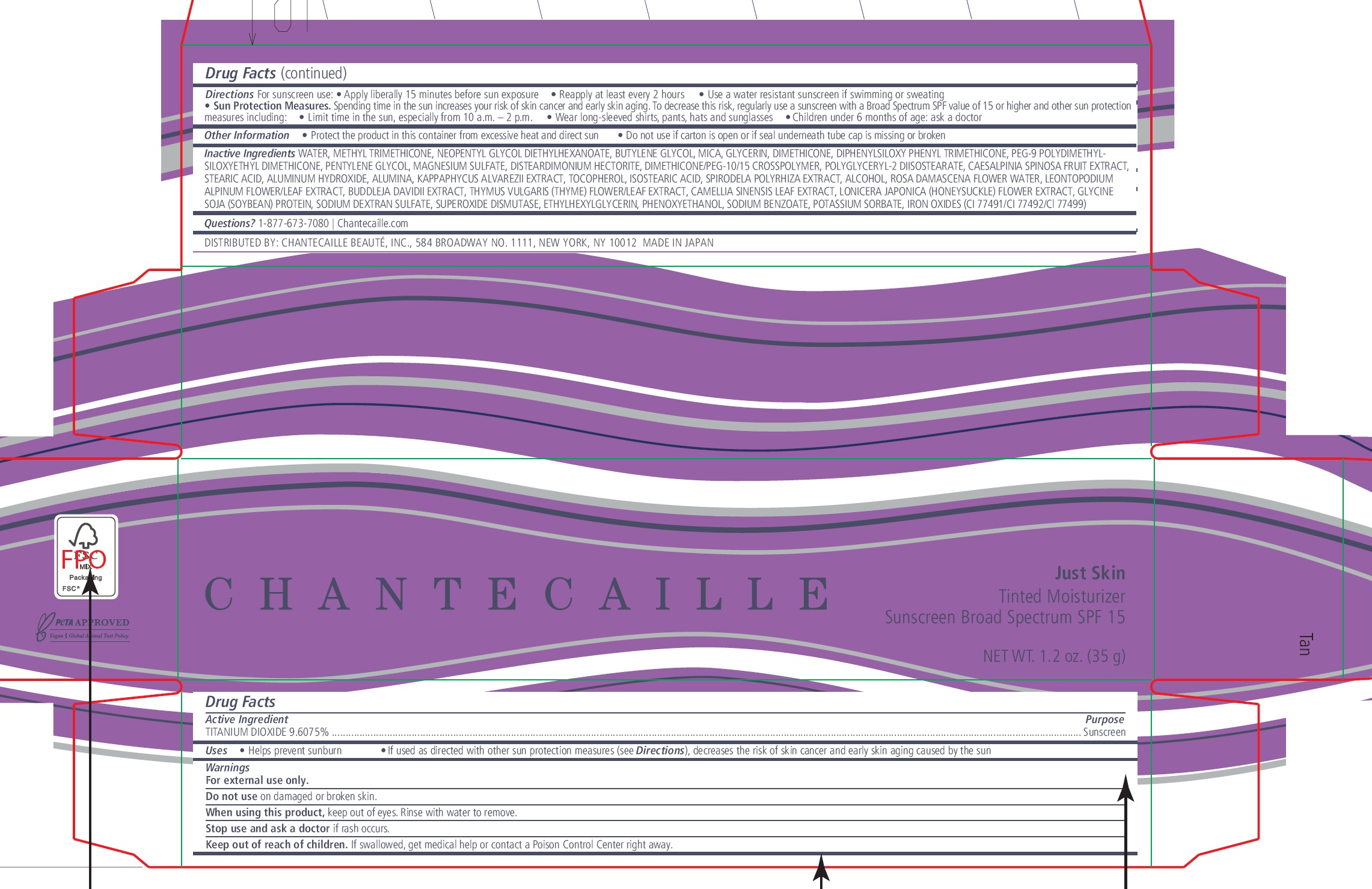
Label: CHANTECAILLE JUST SKIN TINTED MOISTURIZER SUNSCREEN BROAD SPECTRUM SPF 15 TAN- titanium dioxide cream
- NDC Code(s): 42893-033-00
- Packager: Chantecaille Beaute Inc
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated April 24, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredient
- Uses
- Warnings
-
Directions
For sunscreen use:
- Apply liberally 15 minutes before sun exposure
- Reapply at least every 2 hours
- Use a water resistant sunscreen if swimming or sweating
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10 a.m. – 2 p.m.
- Wear long-sleeved shirts, pants, hats and sunglasses
- Children under 6 months of age: ask a doctor
- Other Information
-
Inactive Ingredients
WATER, METHYL TRIMETHICONE, NEOPENTYL GLYCOL DIETHYLHEXANOATE, BUTYLENE GLYCOL, MICA, GLYCERIN, DIMETHICONE, DIPHENYLSILOXY PHENYL TRIMETHICONE, PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE, PENTYLENE GLYCOL, MAGNESIUM SULFATE, DISTEARDIMONIUM HECTORITE, DIMETHICONE/PEG-10/15 CROSSPOLYMER, POLYGLYCERYL-2 DIISOSTEARATE, CAESALPINIA SPINOSA FRUIT EXTRACT, STEARIC ACID, ALUMINUM HYDROXIDE, ALUMINA, KAPPAPHYCUS ALVAREZII EXTRACT, TOCOPHEROL, ISOSTEARIC ACID, SPIRODELA POLYRHIZA EXTRACT, ALCOHOL, ROSA DAMASCENA FLOWER WATER, LEONTOPODIUM ALPINUM FLOWER/LEAF EXTRACT, BUDDLEJA DAVIDII EXTRACT, THYMUS VULGARIS (THYME) FLOWER/LEAF EXTRACT, CAMELLIA SINENSIS LEAF EXTRACT, LONICERA JAPONICA (HONEYSUCKLE) FLOWER EXTRACT, GLYCINE SOJA (SOYBEAN) PROTEIN, SODIUM DEXTRAN SULFATE, SUPEROXIDE DISMUTASE, ETHYLHEXYLGLYCERIN, PHENOXYETHANOL, SODIUM BENZOATE, POTASSIUM SORBATE, IRON OXIDES (CI 77491/CI 77492/Cl 77499)
- Questions?
- Package Labelling:
-
INGREDIENTS AND APPEARANCE
CHANTECAILLE JUST SKIN TINTED MOISTURIZER SUNSCREEN BROAD SPECTRUM SPF 15 TAN
titanium dioxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:42893-033 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 96.075 mg in 1 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) METHYL TRIMETHICONE (UNII: S73ZQI0GXM) NEOPENTYL GLYCOL DIETHYLHEXANOATE (UNII: U68ZV6W62C) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) MICA (UNII: V8A1AW0880) GLYCERIN (UNII: PDC6A3C0OX) DIMETHICONE (UNII: 92RU3N3Y1O) DIPHENYLSILOXY PHENYL TRIMETHICONE (UNII: I445L28B12) PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE (UNII: TYP81E471F) PENTYLENE GLYCOL (UNII: 50C1307PZG) MAGNESIUM SULFATE, UNSPECIFIED FORM (UNII: DE08037SAB) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) DIMETHICONE/PEG-10/15 CROSSPOLYMER (UNII: 21AS8B1BSS) POLYGLYCERYL-2 DIISOSTEARATE (UNII: DG195GP57P) STEARIC ACID (UNII: 4ELV7Z65AP) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) ALUMINUM OXIDE (UNII: LMI26O6933) KAPPAPHYCUS ALVAREZII (UNII: T479H08K2O) TOCOPHEROL (UNII: R0ZB2556P8) ISOSTEARIC ACID (UNII: X33R8U0062) ALCOHOL (UNII: 3K9958V90M) ROSA X DAMASCENA FLOWER OIL (UNII: 18920M3T13) LEONTOPODIUM NIVALE SUBSP. ALPINUM FLOWERING TOP (UNII: QQC1AK06RK) THYME (UNII: CW657OBU4N) GREEN TEA LEAF (UNII: W2ZU1RY8B0) LONICERA JAPONICA FLOWER (UNII: 4465L2WS4Y) SOYBEAN (UNII: L7HT8F1ZOD) SUPEROXIDE DISMUTASE (SACCHAROMYCES CEREVISIAE) (UNII: W2T4YRA9AD) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) PHENOXYETHANOL (UNII: HIE492ZZ3T) SODIUM BENZOATE (UNII: OJ245FE5EU) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) FERRIC OXIDE RED (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42893-033-00 1 in 1 CARTON 04/24/2024 1 35 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 04/24/2024 Labeler - Chantecaille Beaute Inc (095270166)