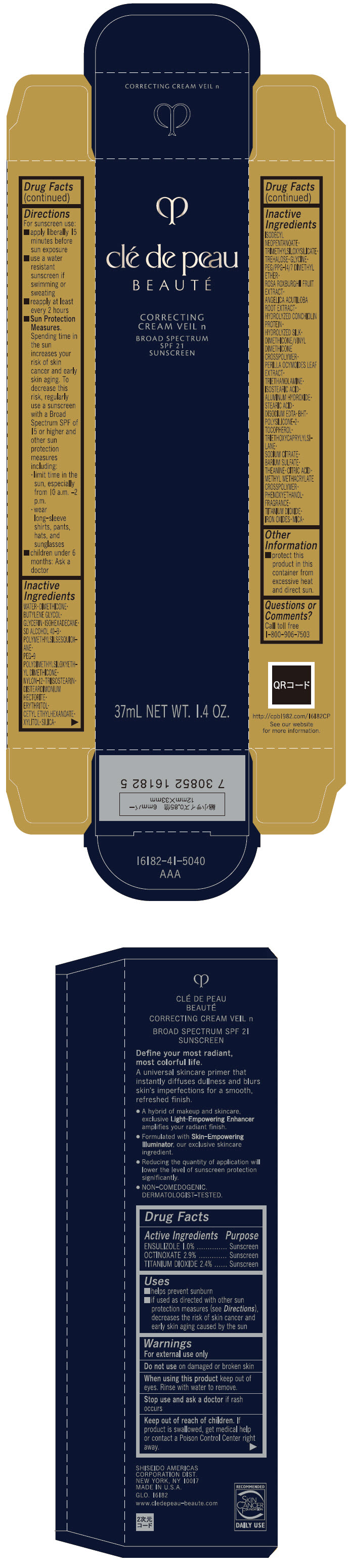
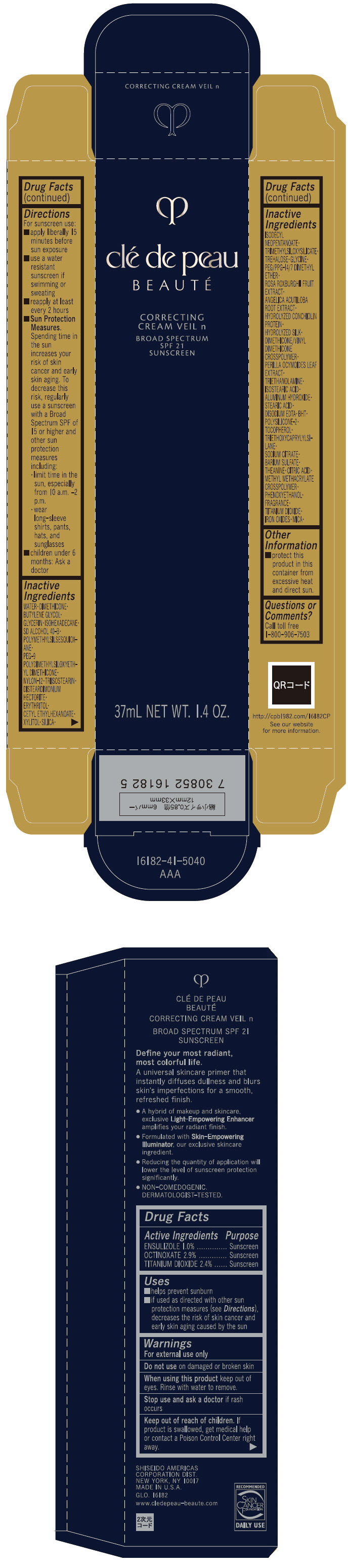
Label: CLE DE PEAU BEAUTE CORRECTING VEIL N- ensulizole, octinoxate, and titanium dioxide cream
- NDC Code(s): 58411-483-10
- Packager: SHISEIDO AMERICAS CORPORATION
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 18, 2019
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
- Warnings
-
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses
- children under 6 months: ask a doctor
-
Inactive Ingredients
WATER▪DIMETHICONE▪BUTYLENE GLYCOL▪GLYCERIN▪ISOHEXADECANE▪SD ALCOHOL 40-B▪POLYMETHYLSILSESQUIOXANE▪PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE▪NYLON-12▪TRIISOSTEARIN▪DISTEARDIMONIUM HECTORITE▪ERYTHRITOL▪CETYL ETHYLHEXANOATE▪XYLITOL▪SILICA▪ISODECYL NEOPENTANOATE▪TRIMETHYLSILOXYSILICATE▪TREHALOSE▪GLYCINE▪PEG/PPG-14/7 DIMETHYL ETHER▪ROSA ROXBURGHII FRUIT EXTRACT▪ANGELICA ACUTILOBA ROOT EXTRACT▪HYDROLYZED CONCHIOLIN PROTEIN▪HYDROLYZED SILK▪DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER▪PERILLA OCYMOIDES LEAF EXTRACT▪TRIETHANOLAMINE▪ISOSTEARIC ACID▪ALUMINUM HYDROXIDE▪STEARIC ACID▪DISODIUM EDTA▪BHT▪POLYSILICONE-2▪TOCOPHEROL▪TRIETHOXYCAPRYLYLSILANE▪SODIUM CITRATE▪BARIUM SULFATE▪THEANINE▪CITRIC ACID▪METHYL METHACRYLATE CROSSPOLYMER▪PHENOXYETHANOL▪FRAGRANCE▪TITANIUM DIOXIDE▪IRON OXIDES▪MICA▪
- Other information
- Questions or comments?
- PRINCIPAL DISPLAY PANEL - 37 mL Bottle Carton
-
INGREDIENTS AND APPEARANCE
CLE DE PEAU BEAUTE CORRECTING VEIL N
ensulizole, octinoxate, and titanium dioxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58411-483 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 0.386 g in 37 mL OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 1.119 g in 37 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 0.9264 g in 37 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) DIMETHICONE (UNII: 92RU3N3Y1O) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) GLYCERIN (UNII: PDC6A3C0OX) ISOHEXADECANE (UNII: 918X1OUF1E) POLYMETHYLSILSESQUIOXANE (4.5 MICRONS) (UNII: 59Z907ZB69) NYLON-12 (UNII: 446U8J075B) TRIISOSTEARIN (UNII: 71503RH8KG) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) ERYTHRITOL (UNII: RA96B954X6) CETYL ETHYLHEXANOATE (UNII: 134647WMX4) XYLITOL (UNII: VCQ006KQ1E) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ISODECYL NEOPENTANOATE (UNII: W60VYE24XC) TRIMETHYLSILOXYSILICATE (M/Q 0.6-0.8) (UNII: 5041RX63GN) TREHALOSE (UNII: B8WCK70T7I) GLYCINE (UNII: TE7660XO1C) PEG/PPG-14/7 DIMETHYL ETHER (UNII: 6DNW9T7YT2) ROSA ROXBURGHII FRUIT (UNII: CVT1AA87FF) ANGELICA ACUTILOBA ROOT (UNII: 3W51R3EK30) DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (SOFT PARTICLE) (UNII: 9E4CO0W6C5) PERILLA FRUTESCENS LEAF (UNII: T4L5881Y68) TROLAMINE (UNII: 9O3K93S3TK) ISOSTEARIC ACID (UNII: X33R8U0062) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) STEARIC ACID (UNII: 4ELV7Z65AP) EDETATE DISODIUM (UNII: 7FLD91C86K) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) BARIUM SULFATE (UNII: 25BB7EKE2E) THEANINE (UNII: 8021PR16QO) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) METHYL METHACRYLATE/GLYCOL DIMETHACRYLATE CROSSPOLYMER (UNII: 657L0NC5MT) PHENOXYETHANOL (UNII: HIE492ZZ3T) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) MICA (UNII: V8A1AW0880) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58411-483-10 1 in 1 CARTON 03/01/2020 1 37 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 03/01/2020 Labeler - SHISEIDO AMERICAS CORPORATION (193691821) Establishment Name Address ID/FEI Business Operations SHISEIDO AMERICAS INC. 782677132 MANUFACTURE(58411-483) , ANALYSIS(58411-483)