Label: SHEER TINT MINERAL SUNSCREEN BROAD SPECTRUM SPF 50 - LIGHT/MEDIUM SHADE- spf 50 sunscreen broad spectrum - light/medium shade lotion
SHEER TINT MINERAL SUNSCREEN BROAD SPECTRUM SPF 50 - MEDIUM/DEEP SHADE- spf 50 sunscreen broad spectrum - medium/deep shade lotion
- NDC Code(s): 51514-0361-1, 51514-0362-1
- Packager: Autumn Harp
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- DOSAGE & ADMINISTRATION
- WARNINGS
-
INSTRUCTIONS FOR USE
Apply generously 15 minutes before sun exposure
Reapply every 2 hours
Use a water resistant sunscreen if swimming or sweating
Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10am - 2pm,
- Wear long-sleeved shirts, pants, hats, and sunglasses
Children under 6 months of age: Ask a doctor
- OTHER SAFETY INFORMATION
-
INACTIVE INGREDIENT
Caprylic/Capric Triglyceride, Water, Diisopropyl Sebacate, Polymethylsilsesquioxane, Undecane, Aloe Barbadensis Leaf Juice, Glycerin, Polyglyceryl-6 Polyricinoleate, Isododecane, Dimethicone, Mica, Polysilicone-11, Tridecane, Argania Spinosa Kernel Oil, Polyhydroxystearic Acid, Tocopheryl Acetate, Polyglyceryl-2 Isostearate, Disteardimonium Hectorite, Phenoxyethanol, Dimethicone Crosspolymer, Sodium Chloride, Triethoxycaprylylsilane, Alumina, Propanediol, Iron Oxide (CI 77492), Magnesium Stearate, Jojoba Esters, Disodium EDTA, 1,2-Hexanediol, Bisabolol, Propylene Carbonate, Titanium Dioxide (CI 77891), Niacinamide, Iron Oxides (CI 77491), Cyclohexasiloxane, Laureth-12, Caprylhydroxamic Acid, Iron Oxides (Black, CI 77499), Sodium Hyaluronate, Ethylhexylglycerin, Tocopherol, Glycyrrhiza Glabra (Licorice) Root Extract, Iron Oxides (CI77499).
- QUESTIONS
-
INDICATIONS & USAGE
Apply generously 15 minutes before sun exposure
Reapply every 2 hours
Use a water resistant sunscreen if swimming or sweating
Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10am - 2pm,
- Wear long-sleeved shirts, pants, hats, and sunglasses
Children under 6 months of age: Ask a doctor
- KEEP OUT OF REACH OF CHILDREN
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SHEER TINT MINERAL SUNSCREEN BROAD SPECTRUM SPF 50 - LIGHT/MEDIUM SHADE
spf 50 sunscreen broad spectrum - light/medium shade lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51514-0361 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 2.37 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 13.35 g in 100 g Inactive Ingredients Ingredient Name Strength LAURETH-12 (UNII: OAH19558U1) FERROSOFERRIC OXIDE (UNII: XM0M87F357) HYALURONATE SODIUM (UNII: YSE9PPT4TH) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCYRRHIZA GLABRA (UNII: 2788Z9758H) DIISOPROPYL SEBACATE (UNII: J8T3X564IH) UNDECANE (UNII: JV0QT00NUE) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) WATER (UNII: 059QF0KO0R) POLYMETHYLSILSESQUIOXANE (11 MICRONS) (UNII: Z570VEV8XK) ISODODECANE (UNII: A8289P68Y2) MICA (UNII: V8A1AW0880) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) ALUMINUM OXIDE (UNII: LMI26O6933) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) JOJOBA OIL (UNII: 724GKU717M) .ALPHA.-BISABOLOL, (+/-)- (UNII: 36HQN158VC) .ALPHA.-TOCOPHEROL, DL- (UNII: 7QWA1RIO01) TRIDECANE (UNII: A3LZF0L939) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) POLYGLYCERYL-2 ISOSTEARATE (UNII: 7B8OE71MQC) PHENOXYETHANOL (UNII: HIE492ZZ3T) SODIUM CHLORIDE (UNII: 451W47IQ8X) PROPANEDIOL (UNII: 5965N8W85T) MAGNESIUM STEARATE (UNII: 70097M6I30) EDETATE DISODIUM (UNII: 7FLD91C86K) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) PROPYLENE CARBONATE (UNII: 8D08K3S51E) NIACINAMIDE (UNII: 25X51I8RD4) FERRIC OXIDE RED (UNII: 1K09F3G675) CYCLOMETHICONE 6 (UNII: XHK3U310BA) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLHYDROXAMIC ACID (UNII: UPY805K99W) GLYCERIN (UNII: PDC6A3C0OX) POLYGLYCERYL-6 POLYRICINOLEATE (UNII: YPM0ZOC2HR) DIMETHICONE 100 (UNII: RO266O364U) ARGANIA SPINOSA SEED (UNII: 8H7X7XB54H) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (SOFT PARTICLE) (UNII: 9E4CO0W6C5) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51514-0361-1 46 g in 1 BOTTLE; Type 0: Not a Combination Product 07/15/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 07/15/2023 SHEER TINT MINERAL SUNSCREEN BROAD SPECTRUM SPF 50 - MEDIUM/DEEP SHADE
spf 50 sunscreen broad spectrum - medium/deep shade lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51514-0362 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 2.37 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 13.35 g in 100 g Inactive Ingredients Ingredient Name Strength LAURETH-12 (UNII: OAH19558U1) FERROSOFERRIC OXIDE (UNII: XM0M87F357) HYALURONATE SODIUM (UNII: YSE9PPT4TH) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCYRRHIZA GLABRA (UNII: 2788Z9758H) DIISOPROPYL SEBACATE (UNII: J8T3X564IH) UNDECANE (UNII: JV0QT00NUE) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) WATER (UNII: 059QF0KO0R) POLYMETHYLSILSESQUIOXANE (11 MICRONS) (UNII: Z570VEV8XK) ISODODECANE (UNII: A8289P68Y2) MICA (UNII: V8A1AW0880) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) ALUMINUM OXIDE (UNII: LMI26O6933) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) JOJOBA OIL (UNII: 724GKU717M) .ALPHA.-BISABOLOL, (+/-)- (UNII: 36HQN158VC) .ALPHA.-TOCOPHEROL, DL- (UNII: 7QWA1RIO01) TRIDECANE (UNII: A3LZF0L939) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) POLYGLYCERYL-2 ISOSTEARATE (UNII: 7B8OE71MQC) PHENOXYETHANOL (UNII: HIE492ZZ3T) SODIUM CHLORIDE (UNII: 451W47IQ8X) PROPANEDIOL (UNII: 5965N8W85T) MAGNESIUM STEARATE (UNII: 70097M6I30) EDETATE DISODIUM (UNII: 7FLD91C86K) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) PROPYLENE CARBONATE (UNII: 8D08K3S51E) NIACINAMIDE (UNII: 25X51I8RD4) FERRIC OXIDE RED (UNII: 1K09F3G675) CYCLOMETHICONE 6 (UNII: XHK3U310BA) ALOE VERA LEAF (UNII: ZY81Z83H0X) CAPRYLHYDROXAMIC ACID (UNII: UPY805K99W) GLYCERIN (UNII: PDC6A3C0OX) POLYGLYCERYL-6 POLYRICINOLEATE (UNII: YPM0ZOC2HR) DIMETHICONE 100 (UNII: RO266O364U) ARGANIA SPINOSA SEED (UNII: 8H7X7XB54H) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (SOFT PARTICLE) (UNII: 9E4CO0W6C5) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51514-0362-1 46 g in 1 BOTTLE; Type 0: Not a Combination Product 07/15/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 07/15/2023 Labeler - Autumn Harp (064187883)


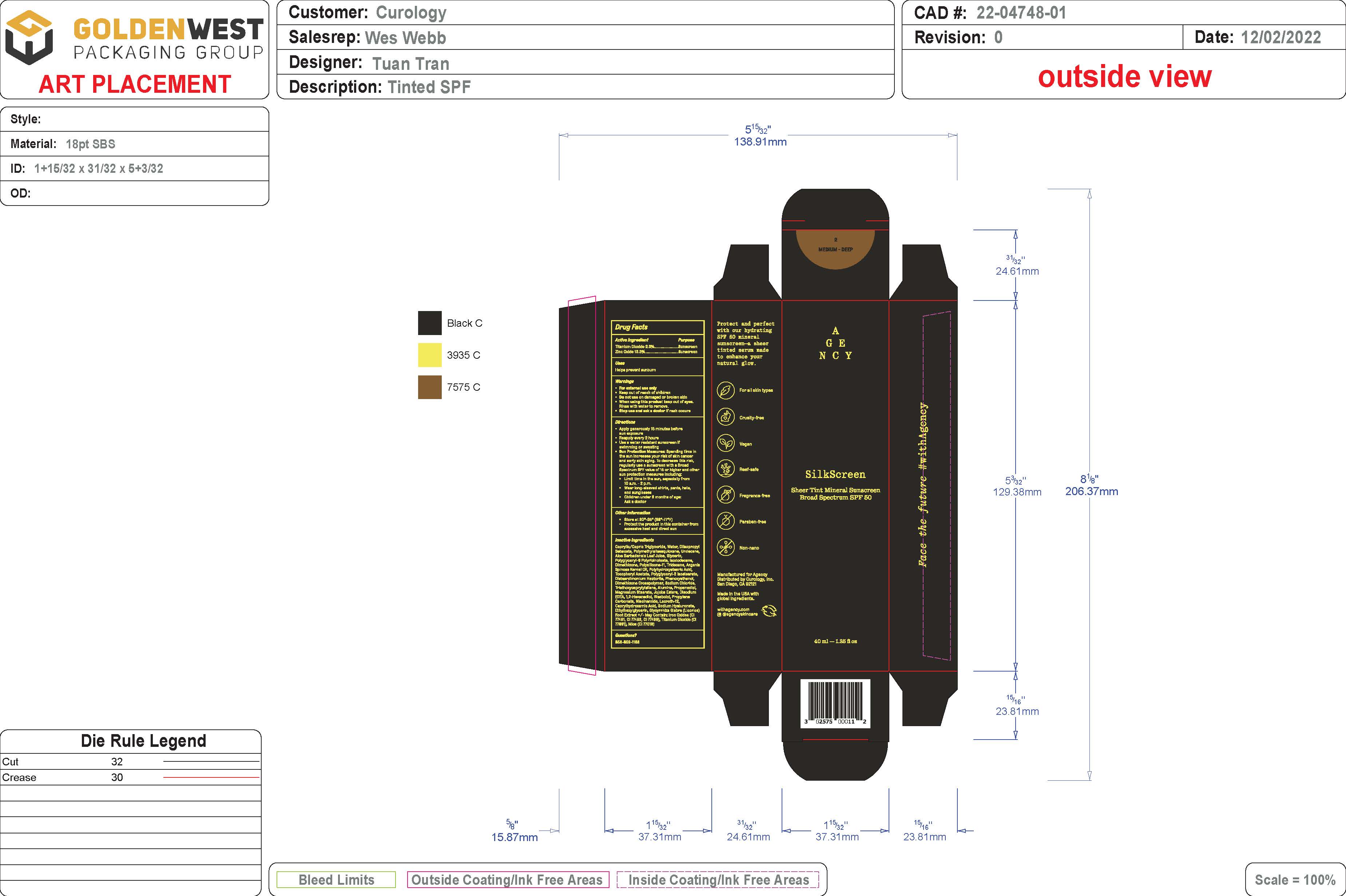
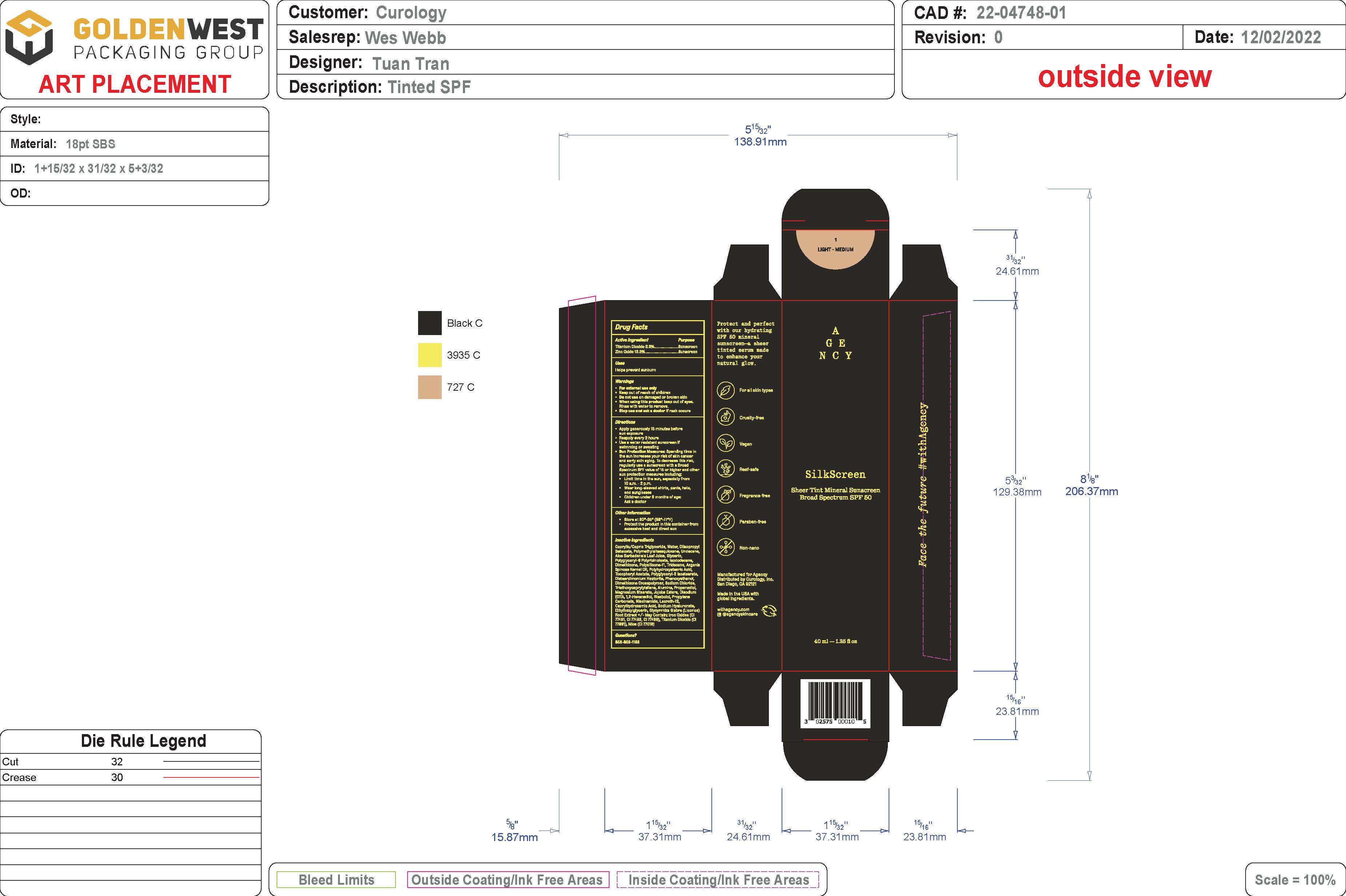

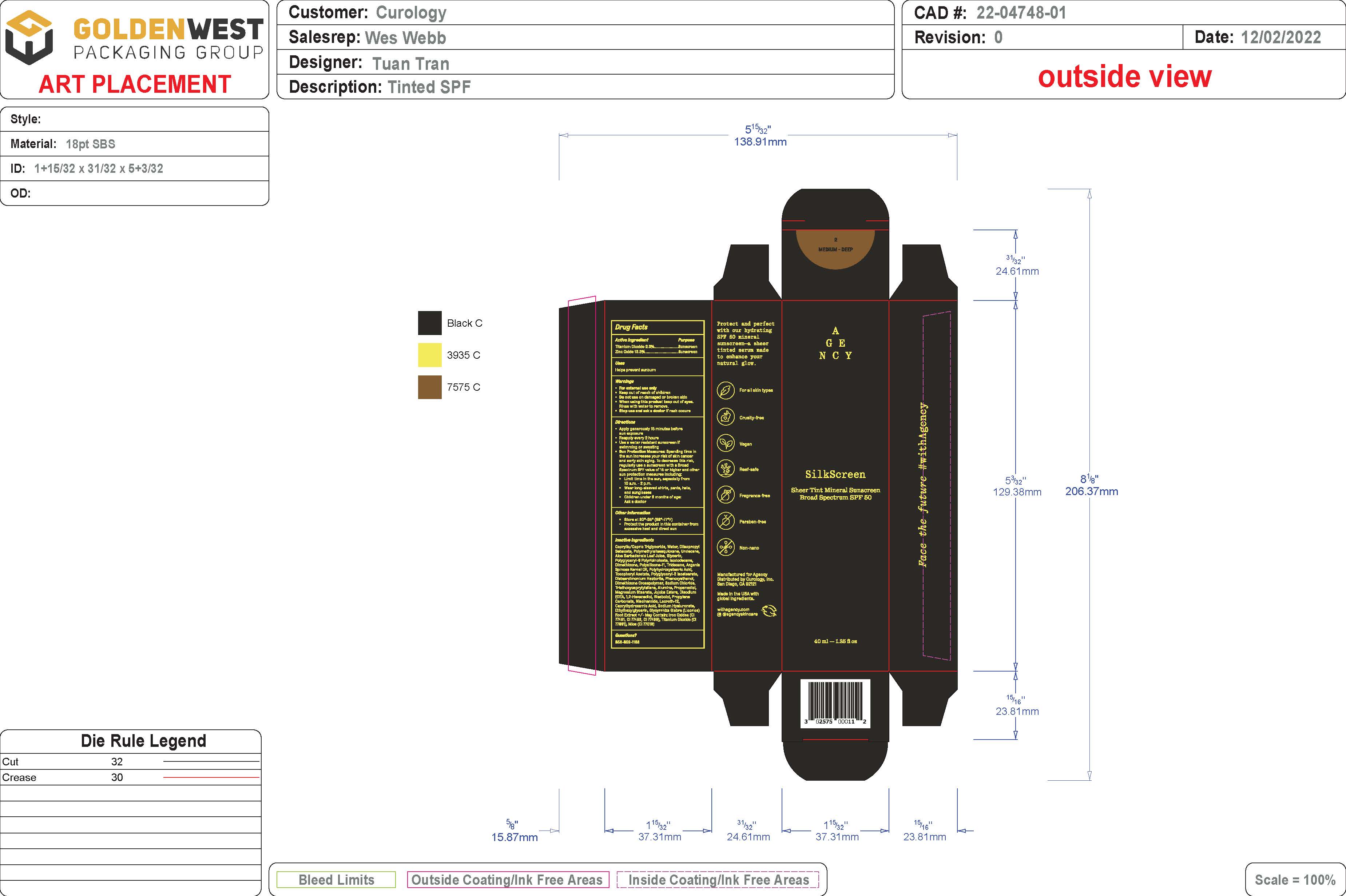
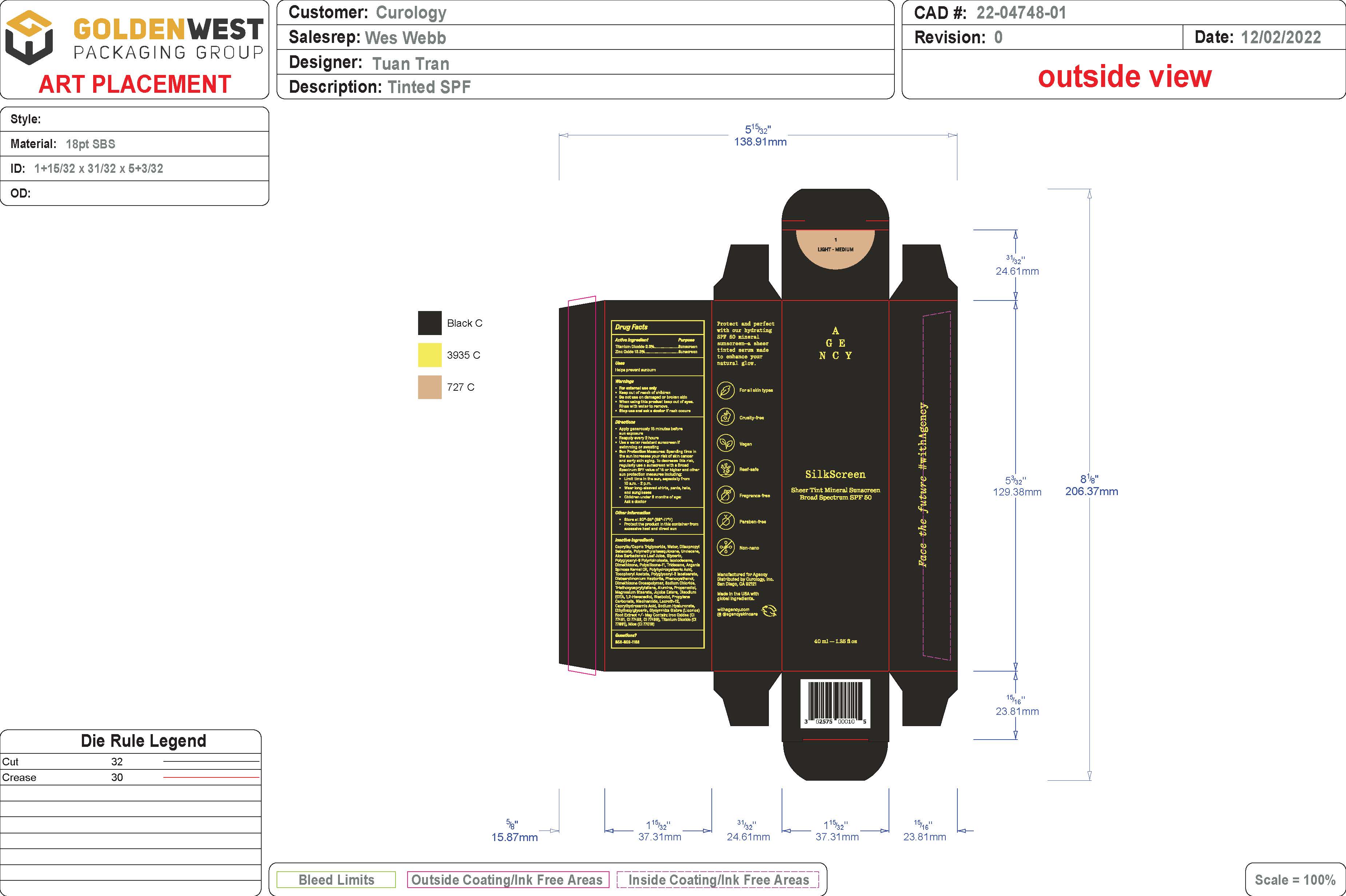 image components
image components