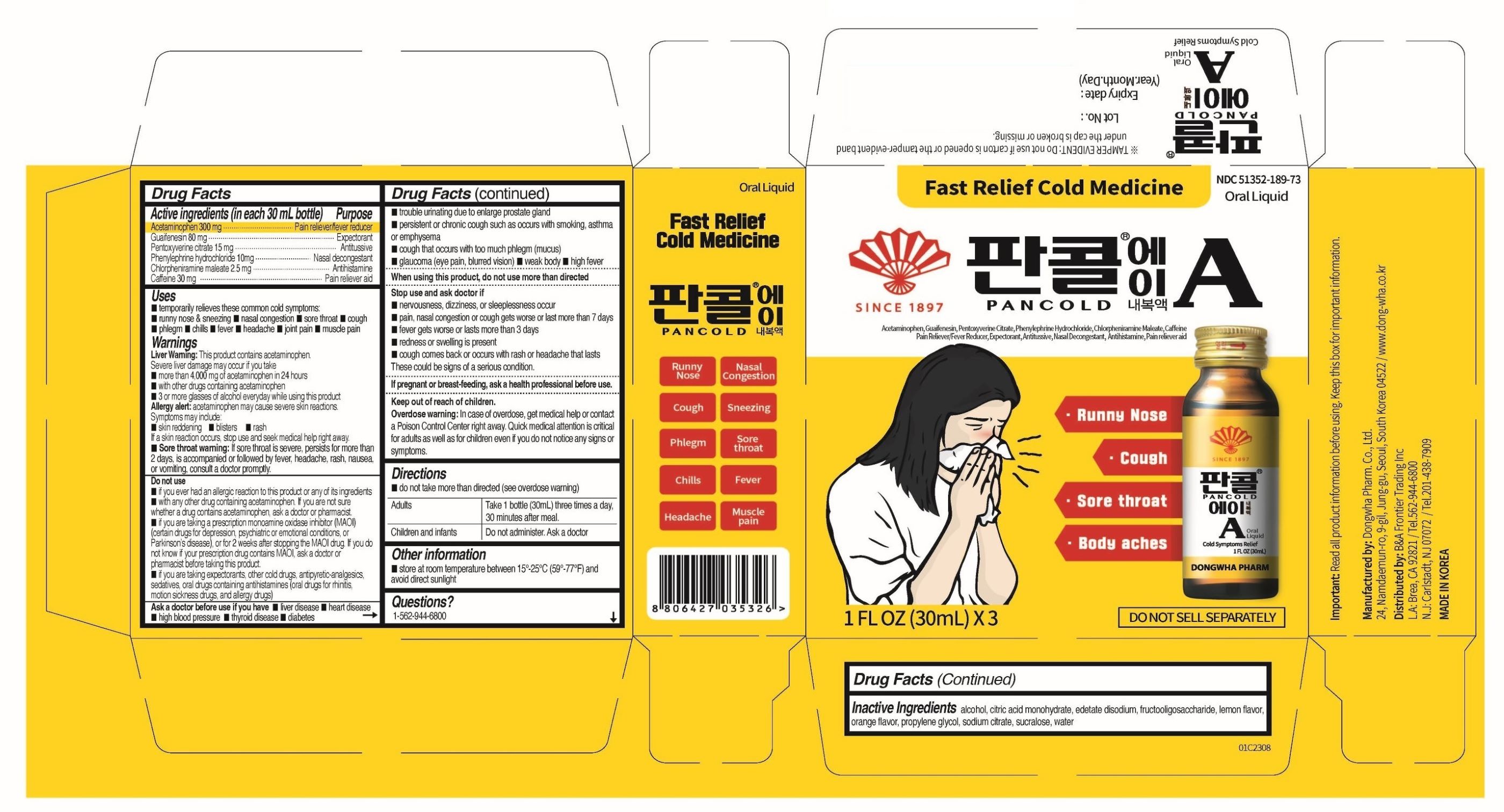
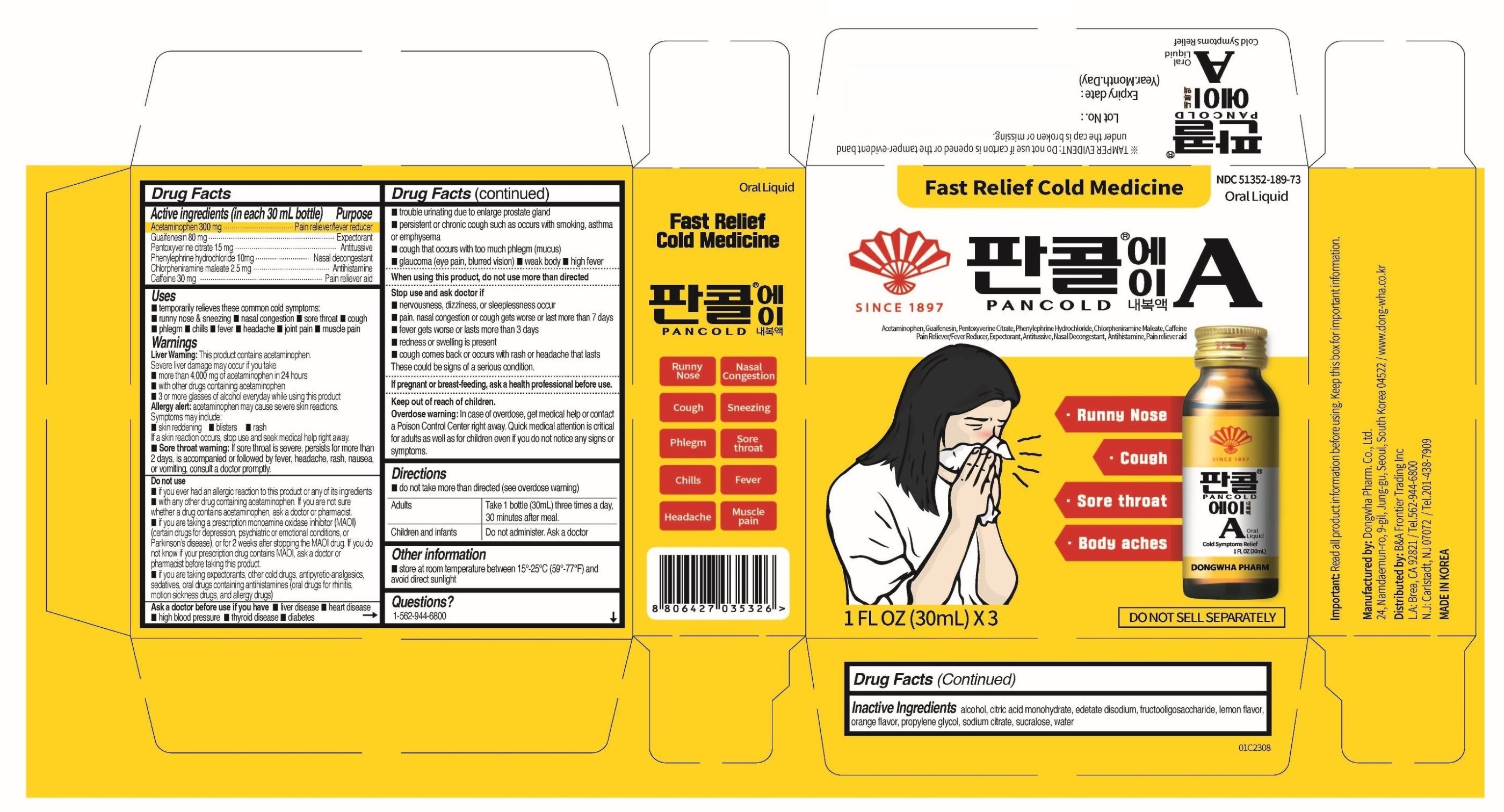
Label: PANCOLD A- acetaminophen, guaifenesin, pentoxyverine citrate, phenylpehrine hydcrochloride, chlorpheniramine maleate, caffeine anhydrous liquid
- NDC Code(s): 51352-189-73, 51352-189-75
- Packager: DONGWHA PHARM. CO. LTD.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 7, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS SECTION
Warnings
Liver Warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more glasses of alcohol everyday while using this product
Allergy alert: acetaminophen mat cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
- Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
-
DO NOT USE
Do not use
- if you ever had an allergic reaction to this product or any of its ingredients
- with any other drug containing acetaminophen. If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- If you are taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains MAOI, ask a doctor or pharmacist before taking this product.
- If you are taking expectorants, other cold drugs, antipyretic-analgesics, sedatives, oral drugs containing antihistamines (oral drugs for rhinitis, motion sickness drugs, and allergy drugs)
-
ASK A DOCTOR
Ask a doctor before use if you have
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to enlarge prostate gland
- persistent or chronic cough such as occurs with smoking, asthma or emphysema
- cough that occurs with too much phlegm (mucus)
- glaucoma (eye pain, blurred vision)
- weak body
- high fever
-
STOP USE
Stop use and ask doctor if
- nervousness, dizziness, or sleeplessness occur
- pain, nasal congestion or cough gets worse or last more than 7 days
- fever gets worse or lasts more than 3 days n redness or swelling is present
- cough comes back or occurs with rash or headache that lasts
These could be signs of a serious condition.
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
- INACTIVE INGREDIENTS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PANCOLD A
acetaminophen, guaifenesin, pentoxyverine citrate, phenylpehrine hydcrochloride, chlorpheniramine maleate, caffeine anhydrous liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51352-189 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 80 mg in 30 mL PENTOXYVERINE CITRATE (UNII: 4SH0MFJ5HJ) (PENTOXYVERINE - UNII:32C726X12W) PENTOXYVERINE CITRATE 15 mg in 30 mL ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 300 mg in 30 mL CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 2.5 mg in 30 mL CAFFEINE (UNII: 3G6A5W338E) (CAFFEINE - UNII:3G6A5W338E) CAFFEINE 30 mg in 30 mL PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 10 mg in 30 mL Inactive Ingredients Ingredient Name Strength SODIUM CITRATE (UNII: 1Q73Q2JULR) 32.4 mg in 30 mL SUCRALOSE (UNII: 96K6UQ3ZD4) 10 mg in 30 mL PROPYLENE GLYCOL (UNII: 6DC9Q167V3) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) 29 mg in 30 mL EDETATE DISODIUM (UNII: 7FLD91C86K) 3 mg in 30 mL ALCOHOL (UNII: 3K9958V90M) 0.5 mL in 30 mL SACCHARUM OFFICINARUM STEM FRUCTOOLIGOSACCHARIDES (UNII: 8LLD82AE3S) 7.5 g in 30 mL WATER (UNII: 059QF0KO0R) ORANGE (UNII: 5EVU04N5QU) LEMON (UNII: 24RS0A988O) Product Characteristics Color Score Shape Size Flavor ORANGE (Light yellowish clear oral liquid with orange-lemon flavor.) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51352-189-73 3 in 1 CASE 07/01/2023 1 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:51352-189-75 5 in 1 CASE 12/07/2023 2 30 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 07/01/2023 Labeler - DONGWHA PHARM. CO. LTD. (687745240) Registrant - DONGWHA PHARM. CO. LTD. (687745240) Establishment Name Address ID/FEI Business Operations DONGWHA PHARM. CO., LTD. 687745240 manufacture(51352-189)