Label: NICAZEL- nicotinamide, pyridoxine, zinc, copper, folic acid tablet, coated
- NHRIC Code(s): 42783-501-60
- Packager: Elorac, Inc
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated December 8, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- NicAzel® (nicotinamide, zinc, azelaic acid, pyridoxine, copper, folic acid)
-
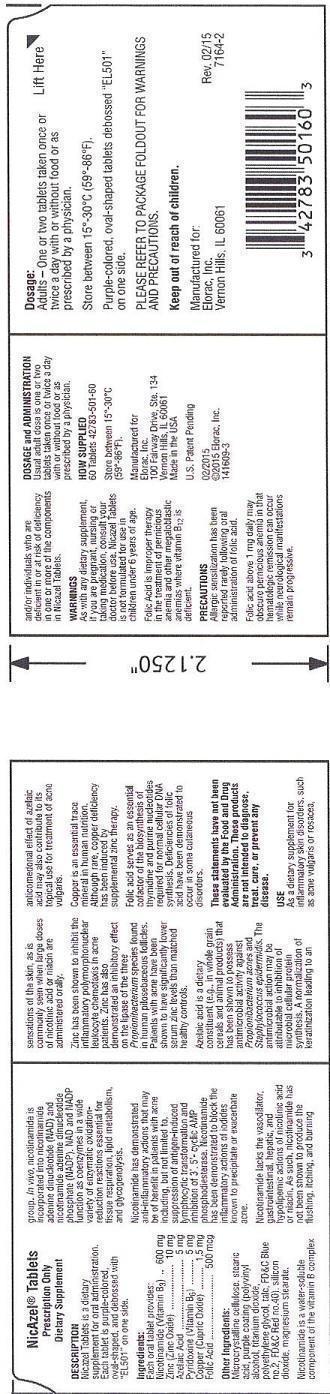
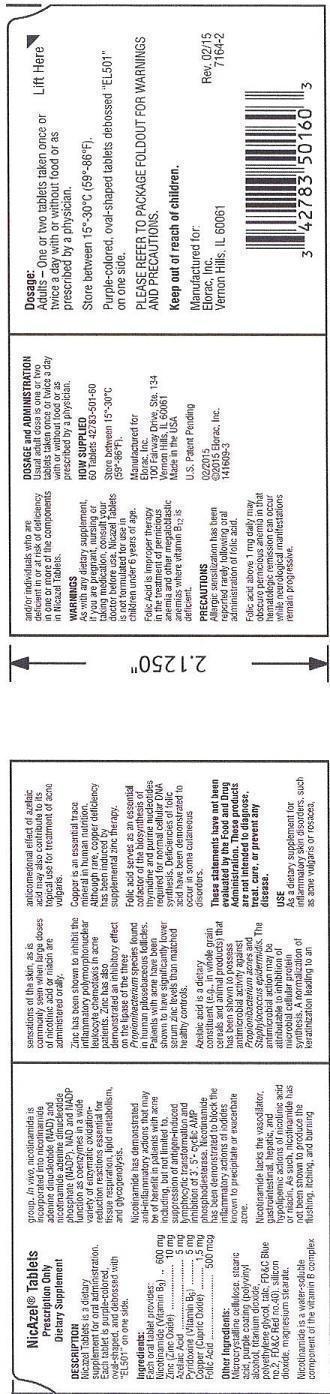
Ingredients:
Each oral tablet provides:
Nicotinamide (Vitamin B3).............600 mg
Zinc (Zinc Oxide)...........................10 mg
Azelaic Acid....................................5 mg
Pyridoxine (Vitamin B6)...................5 mg
Copper (Cupric Oxide)..................1.5 mg
Folic Acid....................................500 mcg
- Other Ingredients:
-
HEALTH CLAIM
Nicotinamide is a water-soluble component of the vitamin B complex group. In vivo, nicotinamide is incorporated into nicotinamide adenine dinucleotide (NAD), and nicotinamide adenine dinucleotide phosphate (NADP). NAD and NADP function as coenzymes in a wide variety of enzymatic oxidation-reduction reactions essential for tissue respiration, lipid metabolism, and glycogenolysis.
Nicotinamide has demonstrated anti-inflammatory actions that may be of benefit in patients with acne including, but not limited to, suppression of antigen-induced lymphocytic transformation and inhibition of 3',5'- cyclic AMP phosphodiesterase. Nicotinamide has been demonstrated to block the inflammatory actions of iodides known to precipitate or exacerbate acne.
Nicotinamide lacks the vasodilator, gastrointestinal, hepatic and hypolipemic actions of nicotinic acid or niacin. As such, nicotinamide has not been shown to produce the flushing, itching and burning sensations of the skin, as is commonly seen when large doses of nicotinic acid or niacin are administered orally.
Zinc has been shown to inhibit the inflammatory polymorphonuclear leukocyte chemotaxis in acne patients. Zinc has also demonstrated an inhibitory effect on the lipase of the three Propionibacterium species found in human pilosebaceous follicles. Patients with acne have been shown to have significantly lower serum zinc levels than matched healthy controls.
Azelaic Acid is a dietary constituent (e.g., in whole grain cereals and animal products) that has been shown to possess antimicrobial activity against Propionibacterium acnes and Staphylococcus epidermidis. The antimicrobial action may be attributable to inhibition of microbial cellular protein synthesis. A normalization of keratinization leading to an anticomedonal effect of azelaic acid may be also contributed to its topical use for treatment of acne vulgaris.
Copper is an essential trace mineral in human nutrition. Although rare, copper deficiency has been induced by supplemental zinc therapy.
Folic acid serves as an essential cofactor of the biosynthesis of thymidine and purine nucleotides required for normal cellular DNA synthesis. Deficiencies of folic acid have been demonstrated to occur in some cutaneous disorders.
These statements have not been evaluated by the Food and Drug Administration. These products are not intended to diagnose, treat, cure, or prevent any disease.
- USE
-
WARNINGS
As with any dietary supplement, if you are pregnant, nursing or taking medication, consult your doctor before use. Nicazel Tablets is not formulated for use in children under 6 years of age.
Folic Acid is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where vitamin B12 is deficient.
- PRECAUTIONS
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- DOSAGE & ADMINISTRATION
-
Dosage:
Adults - One or two tablets taken once or twice a day with or without food or as prescribed by a physician.
Store between 15o-30oC (59o-86oF).Purple-colored, oval-shaped tablets debossed "EL501" on one side.
PLEASE REFER TO PACKAGE FOLDOUT FOR WARNINGS AND PRECAUTIONS.
Keep out of reach of children.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NICAZEL
nicotinamide, pyridoxine, zinc, copper, folic acid tablet, coatedProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:42783-501 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 600 mg ZINC (UNII: J41CSQ7QDS) (ZINC - UNII:J41CSQ7QDS) ZINC 10 mg AZELAIC ACID (UNII: F2VW3D43YT) (AZELAIC ACID - UNII:F2VW3D43YT) AZELAIC ACID 5 mg PYRIDOXINE (UNII: KV2JZ1BI6Z) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE 5 mg COPPER (UNII: 789U1901C5) (COPPER - UNII:789U1901C5) COPPER 1.5 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 500 ug Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYVINYL ALCOHOL (UNII: 532B59J990) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:42783-501-60 60 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date dietary supplement 10/15/2010 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color shape size (solid drugs) 19 mm scoring 1 imprint Labeler - Elorac, Inc (832590009)