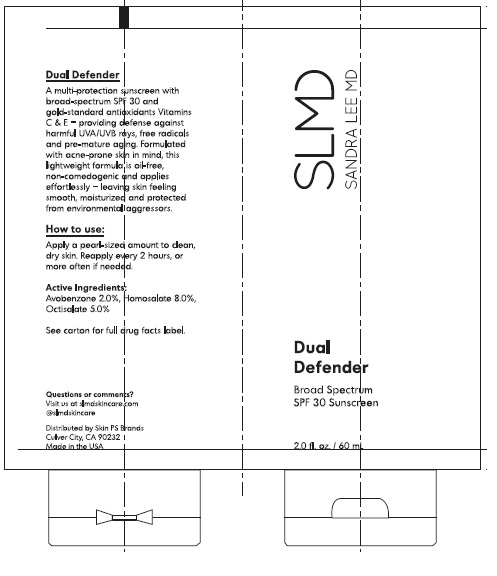
Label: SLMD DUAL DEFENDER SPF 30- avobenzone, homosalate, octisalate lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 73318-4321-2 - Packager: Skin PS Brands
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 4, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions
• Apply liberally and every 15 minutes
before sun exposure.
• Reapply at least every 2 hours.
• Use a water resistant sunscreen if
swimming or sweating:
• immediately after towel drying
• at least every 2 hours
• children under 6 months of age:
ask a doctor• Sun Protection Measures.
Sun exposure may increase your
risk of skin cancer and early aging.
To decrease this risk, regularly use
a sunscreen with a Broad Spectrum
SPF of 15 or higher and take other
sun protection measures. - OTHER SAFETY INFORMATION
-
INACTIVE INGREDIENT
Inactive Ingredients
Water, Cetearyl Alcohol,
Butyloctyl Salicylate, Glycerin,
Trimethoxybenzylidene
Pentanedione, Dimethicone,
Glyceryl Stearate, Poly-
sorbate 60, Behentrimonium
Methosulfate, Silica,
Phenoxyethanol, Caprylyl
Glycol, Hydroxyethylcellulose,
Allantoin, Ethylhexylglycerin,
Panthenol, Sodium Phytate,
Tocopheryl Acetate, Camellia
Sinensis Leaf Extract, Citrus
Grandis (Grapefruit) Fruit
Extract, Hamamelis Virginiana
(Witch Hazel) Leaf Extract,
Sodium Hydroxide, Tetra-
hexyldecyl Ascorbate,
1,2-Hexanediol, Beta-Glucan,
Rosa Canina Fruit Extract. - Product Package for Dual Defender
-
INGREDIENTS AND APPEARANCE
SLMD DUAL DEFENDER SPF 30
avobenzone, homosalate, octisalate lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73318-4321 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 2.0 g in 100 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 8.0 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5.0 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) GLYCERIN (UNII: PDC6A3C0OX) TRIMETHOXYBENZYLIDENE PENTANEDIONE (UNII: 322V0ACF25) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) POLYSORBATE 60 (UNII: CAL22UVI4M) BEHENTRIMONIUM METHOSULFATE (UNII: 5SHP745C61) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) PHENOXYETHANOL (UNII: HIE492ZZ3T) CAPRYLYL GLYCOL (UNII: 00YIU5438U) HYDROXYETHYL CELLULOSE, UNSPECIFIED (UNII: T4V6TWG28D) ALLANTOIN (UNII: 344S277G0Z) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) PANTHENOL (UNII: WV9CM0O67Z) PHYTATE SODIUM (UNII: 88496G1ERL) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) GREEN TEA LEAF (UNII: W2ZU1RY8B0) PUMMELO (UNII: ET1TN5W71X) HAMAMELIS VIRGINIANA LEAF (UNII: T07U1161SV) SODIUM HYDROXIDE (UNII: 55X04QC32I) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) 1,3-BETA-GLUCAN SYNTHASE COMPONENT FKS1 (UNII: 4FRY3LJG0V) ROSA CANINA FRUIT (UNII: 3TNW8D08V3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73318-4321-2 1 in 1 CARTON 02/01/2022 1 60 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 02/01/2022 Labeler - Skin PS Brands (081085221) Registrant - Skin PS Brands (081085221)