Label: SMART CLINICAL REPAIR BROAD SPECTRUM SPF 30- avobenzone,homosalate,octinoxate,octisalate cream
-
NDC Code(s):
49527-083-01,
49527-083-02,
49527-083-03,
49527-083-04, view more49527-083-05
- Packager: CLINIQUE LABORATORIES LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 15, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Use
- Warnings
-
Direction
- For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply at least every two hours
- use a water resistant sunscreen if swimming or sweating
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m.–2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: ask a doctor
-
Inactive Ingredients
water\aqua\eau, glycerin, butylene glycol, coco-caprylate/caprate, polyester-8, ethylhexyl methoxycrylene, butyloctyl salicylate, acrylates copolymer, dextrin palmitate, peg-100 stearate, silica, polyglyceryl-2 stearate, whey protein\lactis protein\protéine du petit-lait, algae extract, saccharomyces ferment, caprylyl glycol, sigesbeckia orientalis (st. paul's wort) extract, caffeine, lauroyl lysine, sodium hyaluronate, biosaccharide gum-1, 7-dehydrocholesterol, plankton extract, acetyl hexapeptide-8, palmitoyl tripeptide-1, palmitoyl hexapeptide-12, palmitoyl tetrapeptide-7, peg-8, tocopheryl acetate, pentylene glycol, hexylene glycol, sodium polyacryloyldimethyl taurate, stearyl alcohol, dehydroxanthan gum, dipropylene glycol, carbomer, potassium hydroxide, glyceryl polymethacrylate, acrylates/beheneth-25 methacrylate copolymer, lecithin, polysorbate 20, sorbitan sesquiisostearate, ppg-8-ceteth-20, glyceryl stearate, sorbeth-30 tetraisostearate, acrylates/c10-30 alkyl acrylate crosspolymer, bht, sodium citrate, disodium edta, phenoxyethanol, sodium benzoate, potassium sorbate <iln51459>
- Other Information
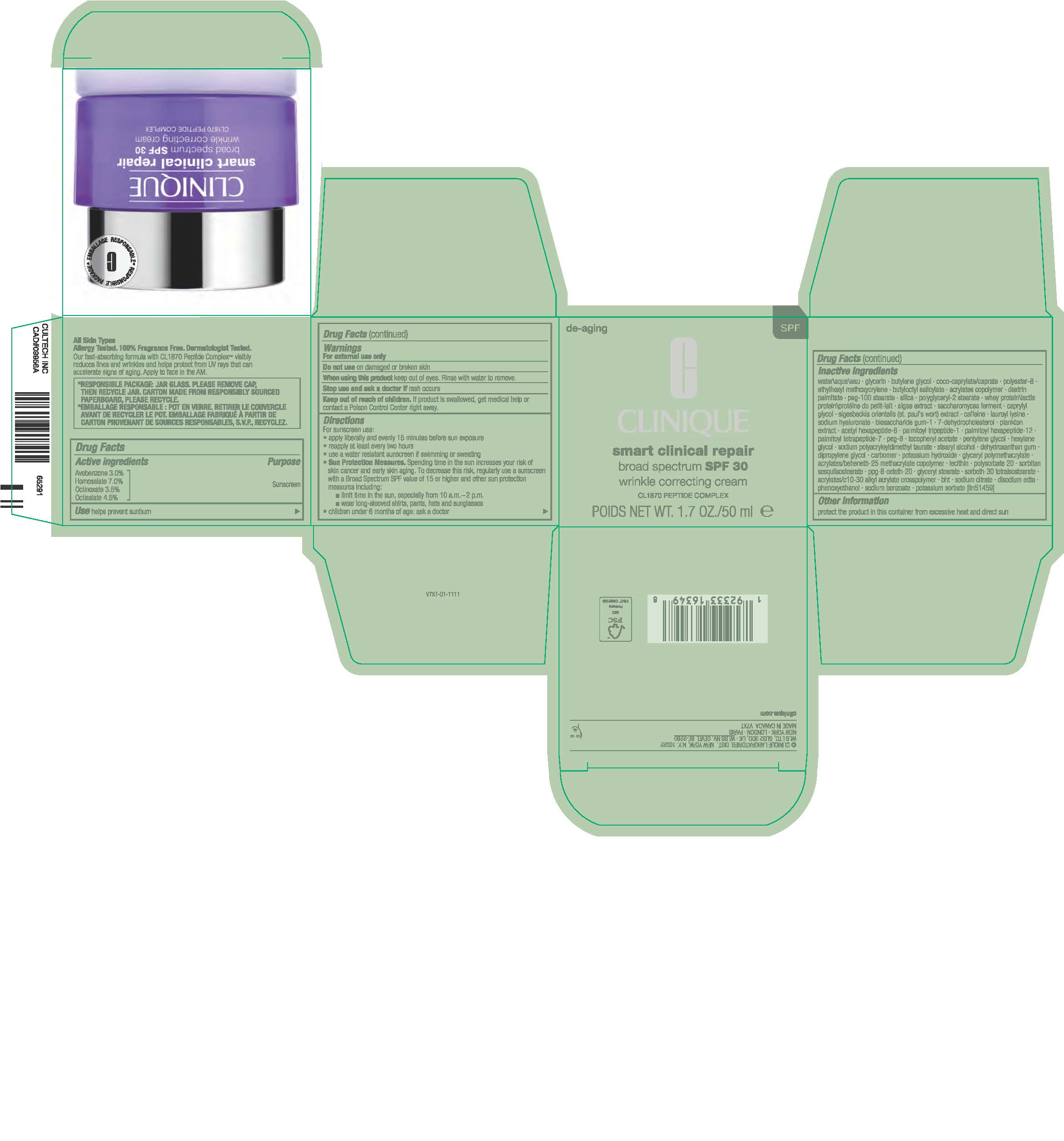
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
SMART CLINICAL REPAIR BROAD SPECTRUM SPF 30
avobenzone,homosalate,octinoxate,octisalate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49527-083 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 70 mg in 1 mL OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 35 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 45 mg in 1 mL Inactive Ingredients Ingredient Name Strength PEG-100 STEARATE (UNII: YD01N1999R) POLYGLYCERYL-2 STEARATE (UNII: 253MC0P0YV) WHEY (UNII: 8617Z5FMF6) PORPHYRIDIUM PURPUREUM (UNII: K2P8K2558N) CAPRYLYL GLYCOL (UNII: 00YIU5438U) HYALURONATE SODIUM (UNII: YSE9PPT4TH) 7-DEHYDROCHOLESTEROL (UNII: BK1IU07GKF) ACETYL HEXAPEPTIDE-8 (UNII: L4EL31FWIL) GLYCERIN (UNII: PDC6A3C0OX) BUTYL ACRYLATE/METHYL METHACRYLATE/METHACRYLIC ACID COPOLYMER (18000 MW) (UNII: JZ1374NL9E) DEXTRIN PALMITATE (CORN; 20000 MW) (UNII: 89B2BSF9I3) HEXYLENE GLYCOL (UNII: KEH0A3F75J) SODIUM POLYACRYLOYLDIMETHYL TAURATE (UNII: NG5NG5733T) DEHYDROXANTHAN GUM (UNII: 63ZP7I1BQO) POLYESTER-8 (1400 MW, CYANODIPHENYLPROPENOYL CAPPED) (UNII: T9296U138P) COCO-CAPRYLATE/CAPRATE (UNII: 8D9H4QU99H) ETHYLHEXYL METHOXYCRYLENE (UNII: S3KFG6Q5X8) SIGESBECKIA ORIENTALIS WHOLE (UNII: ZM9Q0FEI4Z) CAFFEINE (UNII: 3G6A5W338E) PALMITOYL HEXAPEPTIDE-12 (UNII: HO4ZT5S86C) PALMITOYL TETRAPEPTIDE-7 (UNII: Q41S464P1R) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) DIPROPYLENE GLYCOL (UNII: E107L85C40) CARBOXYPOLYMETHYLENE (UNII: 0A5MM307FC) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) POLYSORBATE 20 (UNII: 7T1F30V5YH) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) PHENOXYETHANOL (UNII: HIE492ZZ3T) SODIUM BENZOATE (UNII: OJ245FE5EU) WATER (UNII: 059QF0KO0R) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) PALMITOYL TRIPEPTIDE-1 (UNII: RV743D216M) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) PENTYLENE GLYCOL (UNII: 50C1307PZG) SORBITAN SESQUIISOSTEARATE (UNII: VU97D01BF9) PPG-8-CETETH-20 (UNII: MAL0SND89Q) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) LAUROYL LYSINE (UNII: 113171Q70B) BIOSACCHARIDE GUM-1 (UNII: BB4PU4V09H) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) SODIUM CITRATE (UNII: 1Q73Q2JULR) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49527-083-01 1 in 1 CARTON 05/12/2023 1 50 mL in 1 JAR; Type 0: Not a Combination Product 2 NDC:49527-083-02 1 in 1 CARTON 05/12/2023 2 75 mL in 1 JAR; Type 0: Not a Combination Product 3 NDC:49527-083-03 1 in 1 CARTON 05/12/2023 3 15 mL in 1 JAR; Type 0: Not a Combination Product 4 NDC:49527-083-04 1 in 1 CARTON 05/12/2023 4 5 mL in 1 JAR; Type 0: Not a Combination Product 5 NDC:49527-083-05 1 in 1 CARTON 05/12/2023 5 30 mL in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 05/12/2023 Labeler - CLINIQUE LABORATORIES LLC (044475127) Registrant - Estee Lauder Companies Inc. (790802086) Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics Ltd 202952982 manufacture(49527-083) Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics Ltd. 204132062 manufacture(49527-083) , pack(49527-083) , label(49527-083)


 CLINIQUE
CLINIQUE