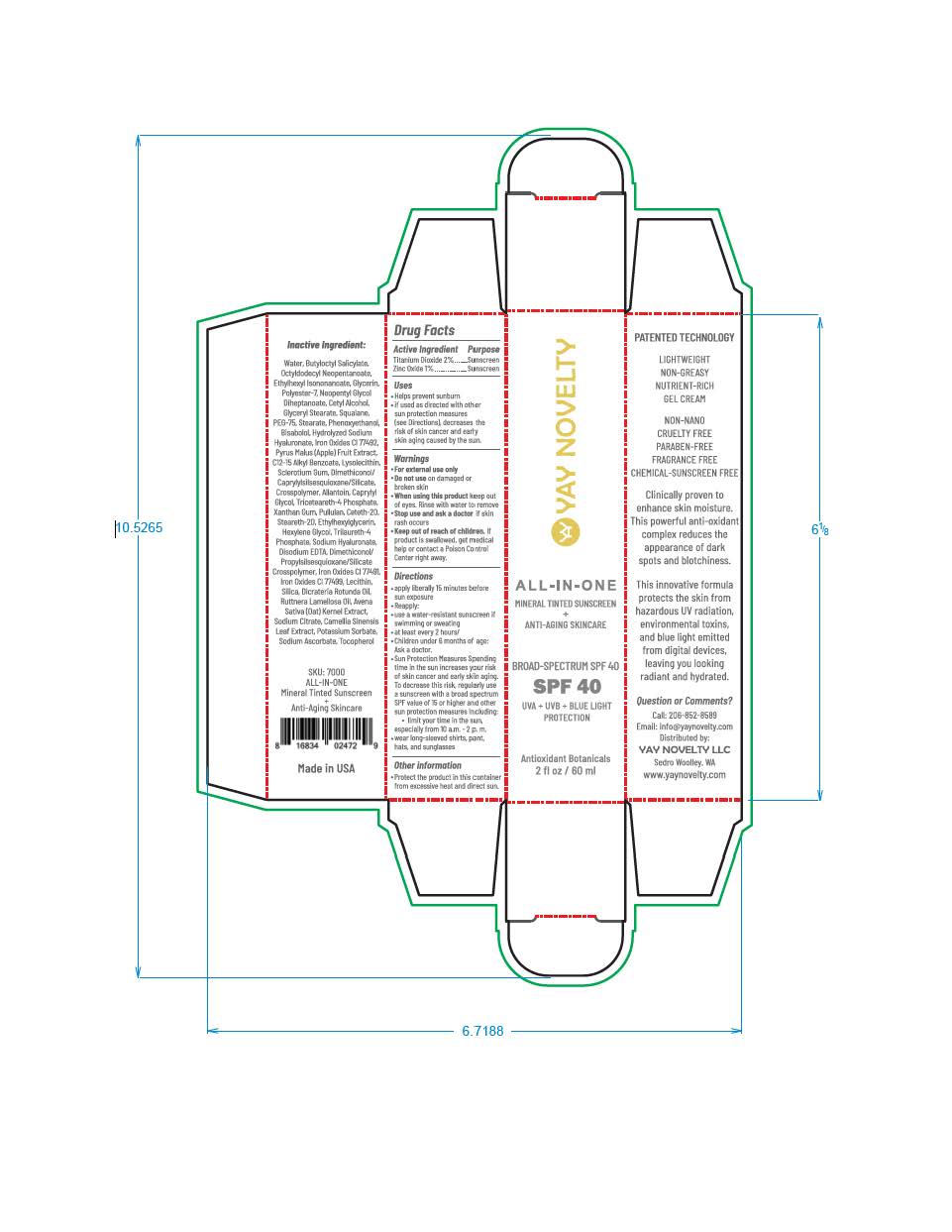
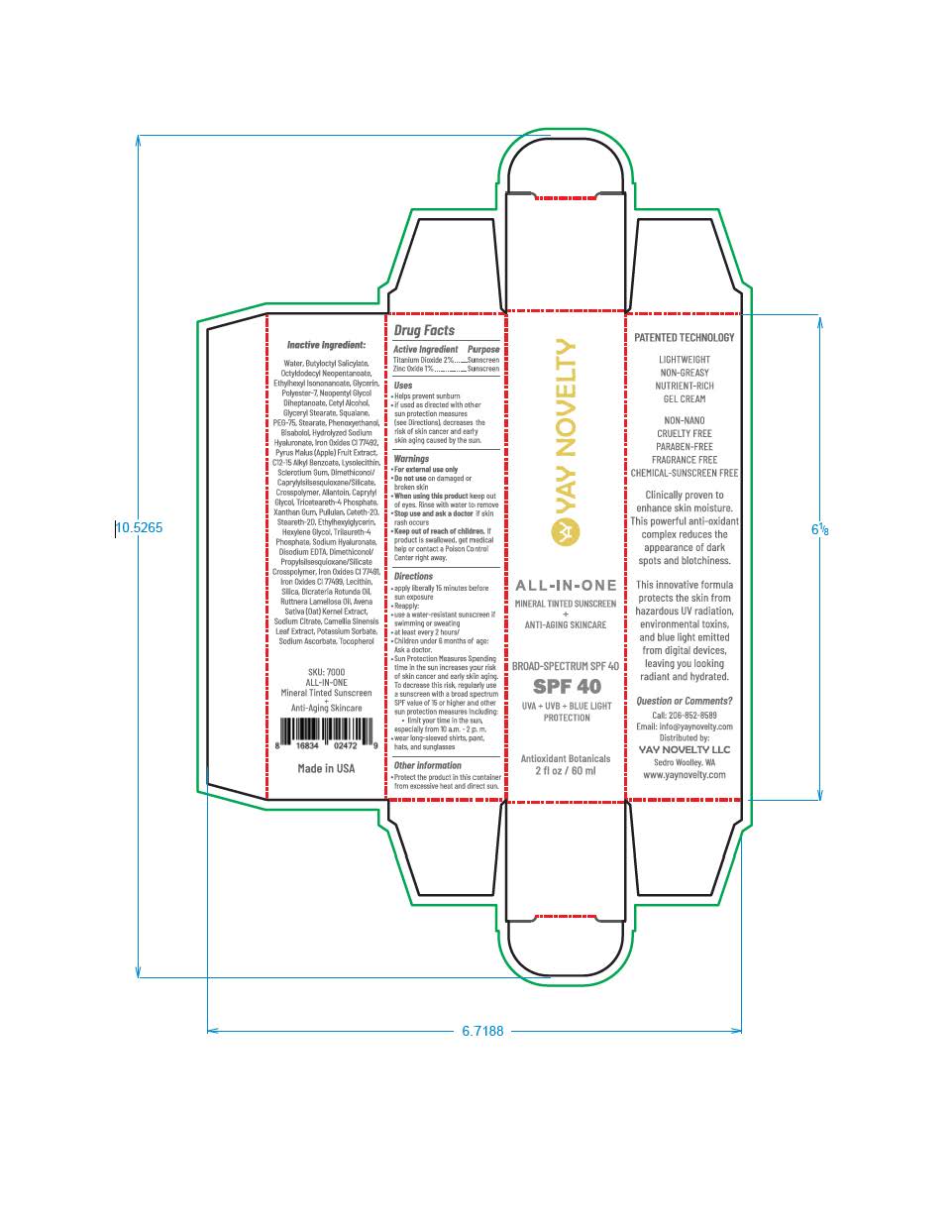
Label: YAY NOVELTY ALL-IN-ONE- titanium dioxide, zinc oxide lotion
- NDC Code(s): 83336-0391-1, 83336-0391-2
- Packager: YAY Novelty
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 12, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- YAY NOVELTY ALL-IN-ONE MINERAL TINTED SUNSCREEN
-
INGREDIENTS AND APPEARANCE
YAY NOVELTY ALL-IN-ONE
titanium dioxide, zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83336-0391 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 9.7 mg in 1 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 19.4 mg in 1 mL Inactive Ingredients Ingredient Name Strength FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) POLYESTER-7 (UNII: 0841698D2F) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DIHEPTANOATE (UNII: 5LKW3C543X) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) ALLANTOIN (UNII: 344S277G0Z) CAPRYLYL GLYCOL (UNII: 00YIU5438U) AVENA SATIVA WHOLE (UNII: 5P8D0Z74RG) DICRATERIA ROTUNDA OIL (UNII: Z0DWU9R4GM) DISODIUM EDTA-COPPER (UNII: 6V475AX06U) CAMELLIA SINENSIS WHOLE (UNII: C5M4585ZBZ) TRICETEARETH-4 PHOSPHATE (UNII: 69534Y66NO) PULLULAN (UNII: 8ZQ0AYU1TT) XANTHAN GUM (UNII: TTV12P4NEE) STEARETH-20 (UNII: L0Q8IK9E08) HEXYLENE GLYCOL (UNII: KEH0A3F75J) TRILAURETH-4 PHOSPHATE (UNII: M96W2OLL2V) SODIUM ASCORBATE (UNII: S033EH8359) OCTYLDODECYL NEOPENTANOATE (UNII: X8725R883T) CETYL ALCOHOL (UNII: 936JST6JCN) SQUALANE (UNII: GW89575KF9) PEG-75 STEARATE (UNII: OT38R0N74H) PHENOXYETHANOL (UNII: HIE492ZZ3T) .ALPHA.-BISABOLOL, (+)- (UNII: 105S6I733Z) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) DIMETHICONOL/PROPYLSILSESQUIOXANE/SILICATE CROSSPOLYMER (450000000 MW) (UNII: 9KB5R958PB) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ETHYLHEXYL ISONONANOATE (UNII: I6KB4GE3K4) CETETH-20 (UNII: I835H2IHHX) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) TOCOPHEROL (UNII: R0ZB2556P8) RUTTNERA LAMELLOSA OIL (UNII: 5XZ38R4SUT) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color brown Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83336-0391-2 1 in 1 CARTON 04/24/2023 1 NDC:83336-0391-1 60 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 04/24/2023 Labeler - YAY Novelty (107893510) Registrant - Nanophase Technologies Corporation (623502044)