Label: IFOSFAMIDE injection, powder, lyophilized, for solution
- NDC Code(s): 63323-142-12
- Packager: Fresenius Kabi USA, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated October 10, 2019
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
BOXED WARNING
(What is this?)
WARNING
Ifosfamide for Injection, USP should be administered under the supervision of a qualified physician experienced in the use of cancer chemotherapeutic agents. Urotoxic side effects, especially hemorrhagic cystitis, as well as CNS toxicities such as confusion and coma have been associated with the use of ifosfamide. When they occur, they may require cessation of ifosfamide therapy. Severe myelosuppression has been reported (see " ADVERSE REACTIONS").
-
DESCRIPTION
Ifosfamide for Injection, USP single-dose vials for constitution and administration by intravenous infusion each contain 1 gram or 3 grams of sterile, lyophilized ifosfamide. Ifosfamide is a chemotherapeutic agent chemically related to the nitrogen mustards and a synthetic analog of cyclophosphamide. Ifosfamide is 3-(2-chloroethyl)-2-[(2-chloroethyl)amino] tetrahydro-2H-1,3,2-oxazaphosphorine 2-oxide. Its structural formula is:
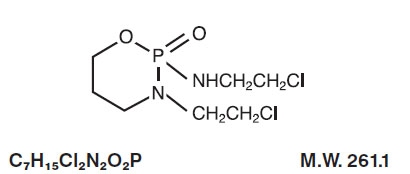
Ifosfamide is a white crystalline powder that is soluble in water.
-
CLINICAL PHARMACOLOGY
Ifosfamide has been shown to require metabolic activation by microsomal liver enzymes to produce biologically active metabolites. Activation occurs by hydroxylation at the ring carbon atom 4 to form the unstable intermediate 4-hydroxyifosfamide. This metabolite rapidly degrades to the stable urinary metabolite 4-ketoifosfamide. Opening of the ring results in formation of the stable urinary metabolite, 4-carboxyifosfamide. These urinary metabolites have not been found to be cytotoxic. N, N- bis (2-chloroethyl)-phosphoric acid diamide (ifosphoramide) and acrolein are also found. Enzymatic oxidation of the chloroethyl side chains, and subsequent dealkylation produces the major urinary metabolites, dechloroethyl ifosfamide and dechloroethyl cyclophosphamide. The alkylated metabolites of ifosfamide have been shown to interact with DNA.
In vitro incubation of DNA with activated ifosfamide has produced phosphotriesters. The treatment of intact cell nuclei may also result in the formation of DNA-DNA cross-links. DNA repair most likely occurs in G-1 and G-2 stage cells.
Pharmacokinetics
Ifosfamide exhibits dose-dependent pharmacokinetics in humans. At single doses of 3.8 to 5 g/m 2, the plasma concentrations decay biphasically and the mean terminal elimination half-life is about 15 hours. At doses of 1.6 to 2.4 g/m 2/day, the plasma decay is monoexponential and the terminal elimination half-life is about 7 hours. Ifosfamide is extensively metabolized in humans and the metabolic pathways appear to be saturated at high doses.
After administration of doses of 5 g/m 2 of 14C-labeled ifosfamide, from 70% to 86% of the dosed radioactivity was recovered in the urine, with about 61% of the dose excreted as parent compound. At doses of 1.6 to 2.4 g/m 2 only 12% to 18% of the dose was excreted in the urine as unchanged drug within 72 hours.
Two different dechloroethylated derivatives of ifosfamide, 4-carboxyifosfamide, thiodiacetic acid and cysteine conjugates of chloroacetic acid have been identified as the major urinary metabolites of ifosfamide in humans and only small amounts of 4-hydroxyifosfamide and acrolein are present. Small quantities (nmole/mL) of ifosfamide mustard and 4-hydroxyifosfamide are detectable in human plasma. Metabolism of ifosfamide is required for the generation of the biologically active species and while metabolism is extensive, it is also quite variable among patients.
In a study at Indiana University, 50 fully evaluable patients with germ cell testicular cancer were treated with Ifosfamide for Injection in combination with cisplatin and either vinblastine or etoposide after failing (47 of 50 patients) at least two prior chemotherapy regimens consisting of cisplatin/vinblastine/bleomycin, (PVB), cisplatin/vinblastine/actinomycin D/bleomycin/ cyclophosphamide, (VAB6), or the combination of cisplatin and etoposide. Patients were selected for remaining cisplatin sensitivity because they had previously responded to a cisplatin containing regimen and had not progressed while on the cisplatin containing regimen or within 3 weeks of stopping it. Patients served as their own control based on the premise that long-term complete responses could not be achieved by retreatment with a regimen to which they had previously responded and subsequently relapsed.
Ten of 50 fully evaluable patients were still alive 2 to 5 years after treatment. Four of the 10 long-term survivors were rendered free of cancer by surgical resection after treatment with the ifosfamide regimen; median survival for the entire group of 50 fully evaluable patients was 53 weeks.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
Continued use of Ifosfamide for Injection is contraindicated in patients with severely depressed bone marrow function (see WARNINGS and PRECAUTIONS sections). Ifosfamide for Injection is also contraindicated in patients who have demonstrated a previous hypersensitivity to it.
-
WARNINGS
Urinary System
Urotoxic side effects, especially hemorrhagic cystitis, have been frequently associated with the use of ifosfamide. It is recommended that a urinalysis should be obtained prior to each dose of ifosfamide. If microscopic hematuria (greater than 10 RBCs per high power field), is present, then subsequent administration should be withheld until complete resolution.
Further administration of ifosfamide should be given with vigorous oral or parenteral hydration.
Hematopoietic System
When ifosfamide is given in combination with other chemotherapeutic agents, severe myelosuppression is frequently observed. Close hematologic monitoring is recommended. White blood cell (WBC) count, platelet count and hemoglobin should be obtained prior to each administration and at appropriate intervals. Unless clinically essential, ifosfamide should not be given to patients with a WBC count below 2000/μL and/or a platelet count below 50,000/μL.
Central Nervous System
Neurologic manifestations consisting of somnolence, confusion, hallucinations and in some instances, coma, have been reported following ifosfamide therapy. The occurrence of these symptoms requires discontinuing ifosfamide therapy. The symptoms have usually been reversible and supportive therapy should be maintained until their complete resolution.
Pregnancy
Animal studies indicate that the drug is capable of causing gene mutations and chromosomal damage in vivo. Embryotoxic and teratogenic effects have been observed in mice, rats and rabbits at doses 0.05 to 0.075 times the human dose. Ifosfamide can cause fetal damage when administered to a pregnant woman. If ifosfamide is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
-
PRECAUTIONS
General
Ifosfamide should be given cautiously to patients with impaired renal function as well as to those with compromised bone marrow reserve, as indicated by: leukopenia, granulocytopenia, extensive bone marrow metastases, prior radiation therapy, or prior therapy with other cytotoxic agents.
Laboratory Tests
During treatment, the patient’s hematologic profile (particularly neutrophils and platelets) should be monitored regularly to determine the degree of hematopoietic suppression. Urine should also be examined regularly for red cells which may precede hemorrhagic cystitis.
Drug Interactions
The physician should be alert for possible combined drug actions, desirable or undesirable, involving ifosfamide even though ifosfamide has been used successfully concurrently with other drugs, including other cytotoxic drugs.
Nursing Mothers
Ifosfamide is excreted in breast milk. Because of the potential for serious adverse events and the tumorigenicity shown for ifosfamide in animal studies, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Ifosfamide has been shown to be carcinogenic in rats, with female rats showing a significant incidence of leiomyosarcomas and mammary fibroadenomas.
The mutagenic potential of ifosfamide has been documented in bacterial systems in vitro and mammalian cells in vivo. In vivo, ifosfamide has induced mutagenic effects in mice and Drosophila melanogaster germ cells, and has induced a significant increase in dominant lethal mutations in male mice as well as recessive sex-linked lethal mutations in Drosophila.
In pregnant mice, resorptions increased and anomalies were present at day 19 after 30mg/m 2 dose of ifosfamide was administered on day 11 of gestation. Embryolethal effects were observed in rats following the administration of 54 mg/m 2 doses of ifosfamide from the 6th through the 15th day of gestation and embryotoxic effects were apparent after dams received 18 mg/m 2 doses over the same dosing period. Ifosfamide is embryotoxic to rabbits receiving 88 mg/m 2/day doses from the 6th through the 18th day after mating. The number of anomalies was also significantly increased over the control group.
-
ADVERSE REACTIONS
In patients receiving ifosfamide as a single agent, the dose-limiting toxicities are myelosuppression and urotoxicity. Dose fractionation, vigorous hydration, and a protector such as mesna can significantly reduce the incidence of hematuria, especially gross hematuria, associated with hemorrhagic cystitis. At a dose of 1.2 g/m 2 daily for 5 consecutive days, leukopenia, when it occurs, is usually mild to moderate. Other significant side effects include alopecia, nausea, vomiting, and central nervous system toxicities.
Adverse Reaction
*Incidence
(%)
Alopecia
83
Nausea-Vomiting
58
Hematuria
46
Gross Hematuria
12
CNS Toxicity
12
Infection
8
Renal Impairment
6
Liver Dysfunction
3
Phlebitis
2
Fever
1
Allergic Reaction
<1
Anorexia
<1
Cardiotoxicity
<1
Coagulopathy
<1
Constipation
<1
Dermatitis
<1
Diarrhea
<1
Fatigue
<1
Hypertension
<1
Hypotension
<1
Malaise
<1
Polyneuropathy
<1
Pulmonary Symptoms
<1
Salivation
<1
Stomatitis
<1
*Based upon 2,070 patients from the published literature in 30 single agent studies.
Hematologic Toxicity
Myelosuppression was dose related and dose limiting. It consisted mainly of leukopenia and, to a lesser extent, thrombocytopenia. A WBC count <3000/μL is expected in 50% of the patients treated with ifosfamide single agent at doses of 1.2 g/m 2 per day for 5 consecutive days. At this dose level, thrombocytopenia (platelets <100,000/μL) occurred in about 20% of the patients. At higher dosages, leukopenia was almost universal, and at total dosages of 10 to 12 g/m 2/cycle, one half of the patients had a WBC count below 1000/μL and 8% of patients had platelet counts less than 50,000/μL. Myelosuppression was usually reversible and treatment can be given every 3 to 4 weeks. When ifosfamide is used in combination with other myelosuppressive agents, adjustments in dosing may be necessary. Patients who experience severe myelosuppression are potentially at increased risk for infection. Anemia has been reported as part of postmarketing surveillance.
Digestive System
Nausea and vomiting occurred in 58% of the patients who received ifosfamide. They were usually controlled by standard antiemetic therapy. Other gastrointestinal side effects include anorexia, diarrhea, and in some cases, constipation.
Urinary System
Urotoxicity consisted of hemorrhagic cystitis, dysuria, urinary frequency and other symptoms of bladder irritation. Hematuria occurred in 6% to 92% of patients treated with ifosfamide. The incidence and severity of hematuria can be significantly reduced by using vigorous hydration, a fractionated dose schedule and a protector such as mesna. At daily doses of 1.2 g/m 2 for 5 consecutive days without a protector, microscopic hematuria is expected in about one half of the patients and gross hematuria in about 8% of patients.
Renal toxicity occurred in 6% of the patients treated with ifosfamide as a single agent. Clinical signs, such as elevation in BUN or serum creatinine or decrease in creatinine clearance, were usually transient. They were most likely to be related to tubular damage. One episode of renal tubular acidosis which progressed into chronic renal failure was reported. Proteinuria and acidosis also occurred in rare instances. Metabolic acidosis was reported in 31% of patients in one study when ifosfamide was administered at doses of 2 to 2.5 g/m 2/day for 4 days. Renal tubular acidosis, Fanconi syndrome, renal rickets, and acute renal failure have been reported. Close clinical monitoring of serum and urine chemistries including phosphorus, potassium, alkaline phosphatase and other appropriate laboratory studies is recommended. Appropriate replacement therapy should be administered as indicated.
Central Nervous System
CNS side effects were observed in 12% of patients treated with ifosfamide. Those most commonly seen were somnolence, confusion, depressive psychosis, and hallucinations. Other less frequent symptoms include dizziness, disorientation, and cranial nerve dysfunction. Seizures and coma with death were occasionally reported. The incidence of CNS toxicity may be higher in patients with altered renal function.
Other
Alopecia occurred in approximately 83% of the patients treated with ifosfamide as a single agent. In combination, this incidence may be as high as 100%, depending on the other agents included in the chemotherapy regimen. Increases in liver enzymes and/or bilirubin were noted in 3% of the patients. Other less frequent side effects included phlebitis, pulmonary symptoms, fever of unknown origin, allergic reactions, stomatitis, cardiotoxicity, and polyneuropathy.
- OVERDOSAGE
-
DOSAGE AND ADMINISTRATION
Ifosfamide for Injection should be administered intravenously at a dose of 1.2 g/m 2 per day for 5 consecutive days. Treatment is repeated every 3 weeks or after recovery from hematologic toxicity (Platelets ≥100,000/μL, WBC ≥4,000/μL). In order to prevent bladder toxicity, Ifosfamide for Injection should be given with extensive hydration consisting of at least 2 liters of oral or intravenous fluid per day. A protector, such as mesna, should also be used to prevent hemorrhagic cystitis. Ifosfamide for Injection should be administered as a slow intravenous infusion lasting a minimum of 30 minutes. Although Ifosfamide for Injection has been administered to a small number of patients with compromised hepatic and/or renal function, studies to establish optimal dose schedules of Ifosfamide for Injection in such patients have not been conducted.
Preparation for Intravenous Administration/Stability
Injections are prepared for parenteral use by adding Sterile Water for Injection, USP, or Bacteriostatic Water for Injection, USP (benzyl alcohol or parabens preserved), to the vial and shaking to dissolve. Use the quantity of diluent shown below to constitute the product:
Dosage
Strength
Quantity of
Diluent
Final
Concentration
1 gram
20 mL
50 mg/mL
3 grams
60 mL
50 mg/mL
Solutions of ifosfamide may be diluted further to achieve concentrations of 0.6 to 20 mg/mL in the following fluids:
5% Dextrose Injection, USP
0.9% Sodium Chloride Injection, USP
Lactated Ringer’s Injection, USP
Sterile Water for Injection, USP
Because essentially identical stability results were obtained for Sterile Water admixtures as for the other admixtures (5% Dextrose Injection, 0.9% Sodium Chloride Injection, and Lactated Ringer’s Injection), the use of large volume parenteral glass bottles, Viaflex bags or PAB™ bags that contain intermediate concentrations or mixtures of excipients (e.g., 2.5% Dextrose Injection, 0.45% Sodium Chloride Injection, or 5% Dextrose and 0.9% Sodium Chloride Injection) is also acceptable.
Constituted or constituted and further diluted solutions of Ifosfamide for Injection should be refrigerated and used within 24 hours.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration.
-
HOW SUPPLIED
Ifosfamide for Injection, USP, lyophilized is available as:
Product
No.
NDC
No.
NP104210
63323-142-12
Ifosfamide for Injection, USP, 1 gram single-dose vial, packaged individually.
NP104300
63323-143-12
Ifosfamide for Injection, USP, 3 gram single-dose vial, packaged individually.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Vial stoppers do not contain natural rubber latex.
Protect from temperatures above 30°C (86°F).
Procedures for proper handling and disposal of anticancer drugs should be considered. Skin reactions associated with accidental exposure to Ifosfamide for Injection may occur. The use of gloves is recommended. If Ifosfamide for Injection solution contacts the skin or mucosa, immediately wash the skin thoroughly with soap and water or rinse the mucosa with copious amounts of water. Several guidelines on this subject have been published. 1-7 There is no general agreement that all of the procedures recommended in the guidelines are necessary or appropriate.
-
REFERENCES
- Recommendations for the Safe Handling of Parenteral Antineoplastic Drugs. NIH Publication No. 83-2621. For sale by the Superintendent of Documents, U.S. Government Printing Office, Washington, DC 20402.
- AMA Council Report. Guidelines for Handling Parenteral Antineoplastics. JAMA 1985; 253 (11):1590-1592.
- National Study Commission on Cytotoxic Exposure-Recommendations for Handling Cytotoxic Agents. Available from Louis P. Jeffrey, Sc. D., Chairman, National Study Commission on Cytotoxic Exposure, Massachusetts College of Pharmacy and Allied Health Sciences, 179 Longwood Avenue, Boston, Massachusetts 02115.
- Clinical Oncological Society of Australia: Guidelines and Recommendations for Safe Handling of Antineoplastic Agents. Med J Australia 1983; 1:426-428.
- Jones, RB, et al: Safe Handling of Chemotherapeutic Agents: A Report from the Mount Sinai Medical Center, CA-A Cancer Journal for Clinicians 1983; (Sept./Oct.) 258-263.
- American Society of Hospital Pharmacists Technical Assistance Bulletin on Handling Cytotoxic and Hazardous Drugs. Am J Hosp Pharm 1990;47:1033-1049.
- Controlling Occupational Exposure to Hazardous Drugs. (OSHA WORK-PRACTICE GUIDELINES). Am J Health-Syst Pharm 1996; 53:1669-1685.
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - PRINCIPAL DISPLAY - Ifosfamide 1 g Single Dose Vial Label
NDC 63323-142-12
NP104210
Ifosfamide for Injection, USP
1 g
For IV Use
Single-Dose Vial
Rx only

PACKAGE LABEL - PRINCIPAL DISPLAY - Ifosfamide 1 g Single Dose Vial Carton Panel
NDC 63323-142-12
NP104210
Ifosfamide for Injection, USP
1 g
For IV Use
Single-Dose Vial
Rx only

-
INGREDIENTS AND APPEARANCE
IFOSFAMIDE
ifosfamide injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:63323-142 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IFOSFAMIDE (UNII: UM20QQM95Y) (IFOSFAMIDE - UNII:UM20QQM95Y) IFOSFAMIDE 1 g Inactive Ingredients Ingredient Name Strength BENZYL ALCOHOL (UNII: LKG8494WBH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63323-142-12 1 in 1 BOX 01/28/2003 1 1 in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076078 01/28/2003 Labeler - Fresenius Kabi USA, LLC (608775388) Establishment Name Address ID/FEI Business Operations Fresenius Kabi USA, LLC 023648251 manufacture(63323-142)


