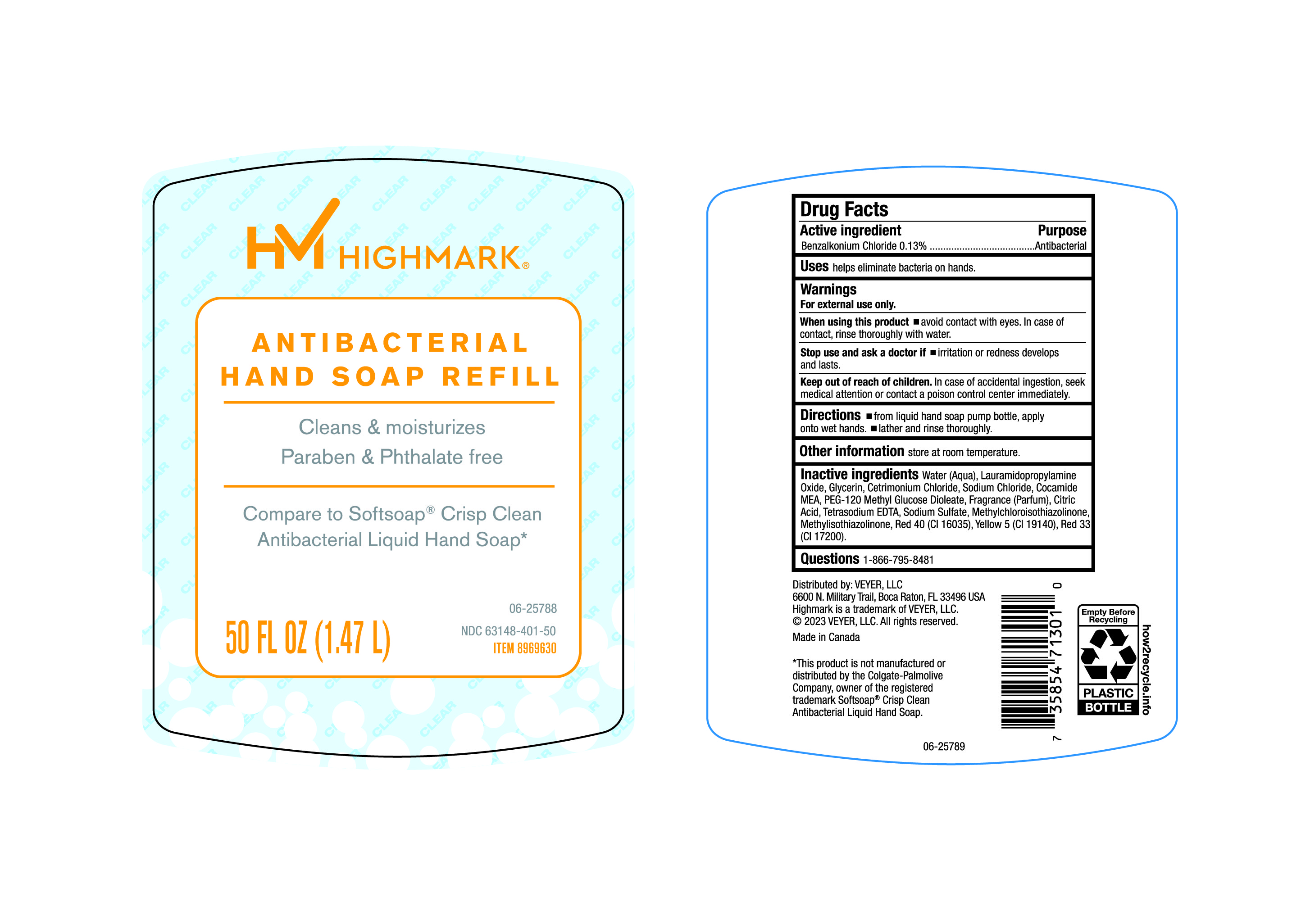
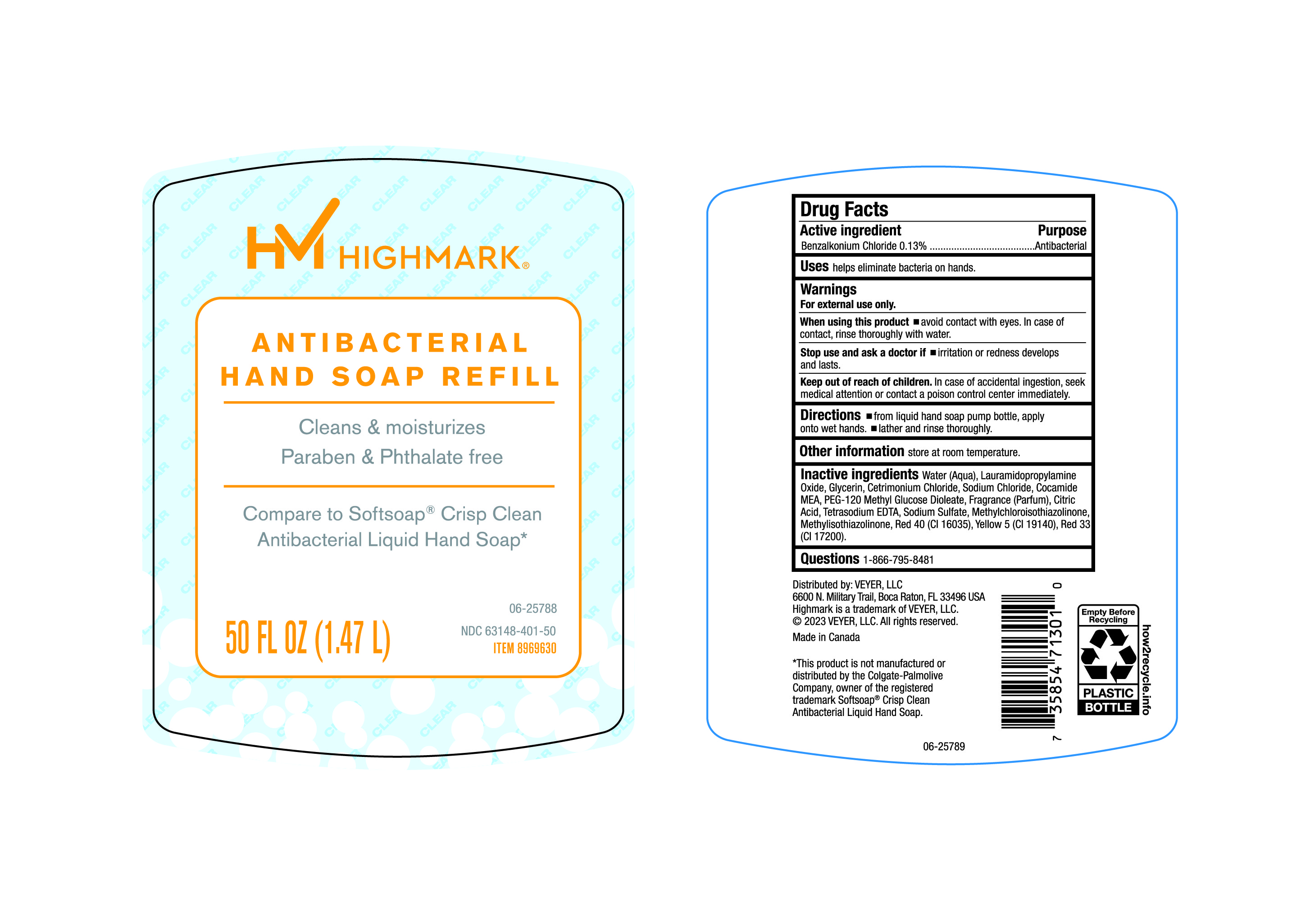
Label: HIMARK- benzalkonium chloride liquid
- NDC Code(s): 63148-401-07, 63148-401-50
- Packager: Apollo Health and Beauty Care
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 24, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
- Warnings
- Directions
- Other Information
-
Inactive Ingredients
Water (Aqua), Lauramidopropylamine Oxide, Glycerin, Cetrimonium Chloride, Sodium Chloride, Cocamide MEA, PEG-120 Methyl Glucose Dioleate, Fragrance (Parfum), Citric Acid, Tetrasodium EDTA, Sodium Sulfate, Methylchloroisothiazolinone, Methylisothiazolinone, Red 40 (CI 16035), Yellow 5(CI19140), Red 33 (CI17200).
- Questions
- Principal Display Panel- 50 Oz
- Principal Display Panel-7.5 Oz
-
INGREDIENTS AND APPEARANCE
HIMARK
benzalkonium chloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63148-401 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 130 mg in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) COCO MONOETHANOLAMIDE (UNII: C80684146D) FRAGRANCE CLEAN ORC0600327 (UNII: 329LCV5BTF) METHYLCHLOROISOTHIAZOLINONE (UNII: DEL7T5QRPN) FD&C RED NO. 40 (UNII: WZB9127XOA) BASIC YELLOW 5 (UNII: 07BP340B4T) GLYCERIN (UNII: PDC6A3C0OX) METHYL GLUCOSE DIOLEATE (UNII: FA9KFJ4Z6P) METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) D&C RED NO. 33 (UNII: 9DBA0SBB0L) LAURAMIDOPROPYLAMINE OXIDE (UNII: I6KX160QTV) CETRIMONIUM CHLORIDE (UNII: UC9PE95IBP) EDETATE SODIUM (UNII: MP1J8420LU) SODIUM SULFATE (UNII: 0YPR65R21J) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63148-401-07 221 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/01/2023 2 NDC:63148-401-50 1478 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 06/01/2023 Labeler - Apollo Health and Beauty Care (201901209) Registrant - Apollo Health and Beauty Care (201901209) Establishment Name Address ID/FEI Business Operations Apollo Health and Beauty Care 201901209 manufacture(63148-401)