Label: EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (ROUGE GRANDIOSE) - RED- octinoxate lipstick
EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (ROUGE RUBI) - RED- octinoxate lipstick
EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (SOBRIETE) - BROWN- octinoxate lipstick
EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (MARRON ILIMITEE) - BROWN (oc .......T ROUGE AMPLIFIER AMPLIFYING SPF 15 (MYSTIC ROSE) - PINK- octinoxate lipstick
EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (BELLE NUDE) - BEIGE- octinoxate lipstick
EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (FUCSIA PASSION) - PURPLE- octinoxate lipstick
EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (VIN CHIC) - BROWN- octinoxate lipstick
-
Contains inactivated NDC Code(s)
NDC Code(s): 13537-800-39, 13537-800-40, 13537-801-41, 13537-801-42, view more13537-802-43, 13537-802-44, 13537-870-01, 13537-870-02, 13537-871-03, 13537-871-04, 13537-872-05, 13537-872-06, 13537-873-07, 13537-873-08, 13537-874-09, 13537-874-10, 13537-875-11, 13537-875-12, 13537-876-13, 13537-876-14, 13537-877-15, 13537-877-16, 13537-878-17, 13537-878-18, 13537-879-19, 13537-879-20, 13537-881-21, 13537-881-22, 13537-882-23, 13537-882-24, 13537-883-25, 13537-883-26, 13537-884-27, 13537-884-28, 13537-885-29, 13537-885-30, 13537-887-31, 13537-887-32, 13537-888-33, 13537-888-34, 13537-889-35, 13537-889-36, 13537-890-37, 13537-890-38 - Packager: Ventura Corporation LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 19, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredients
- Purpose
- Uses
- Warnings
- Directions
- Other information
-
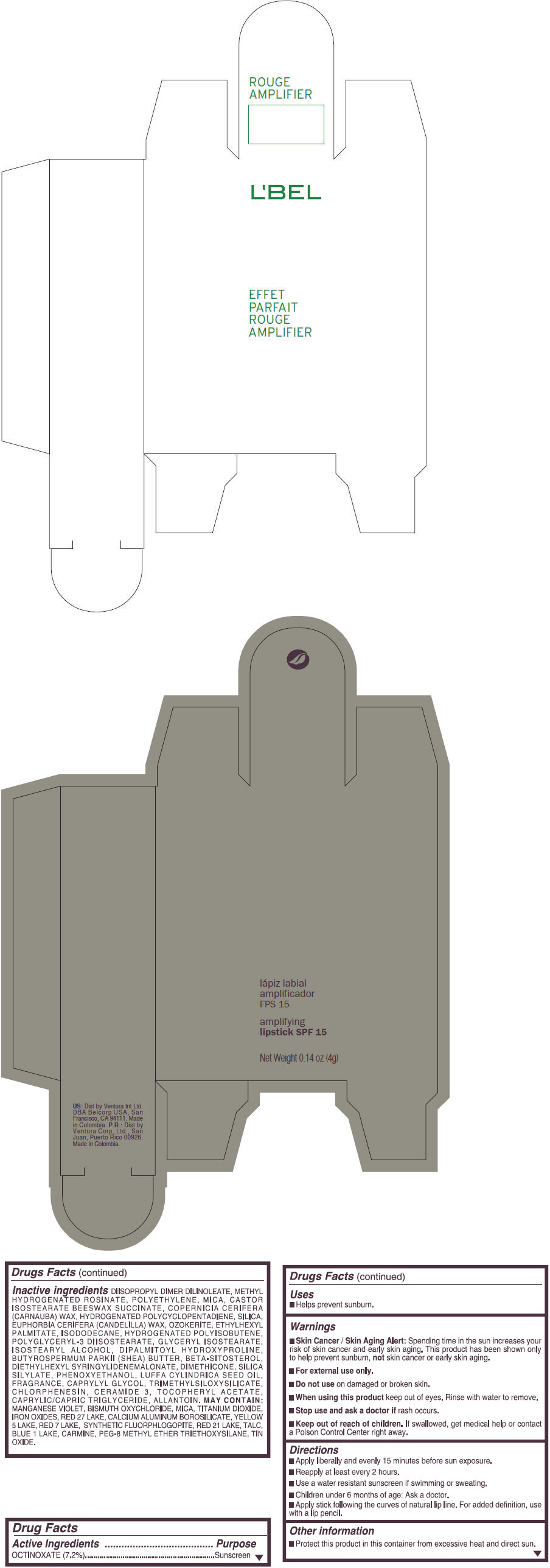
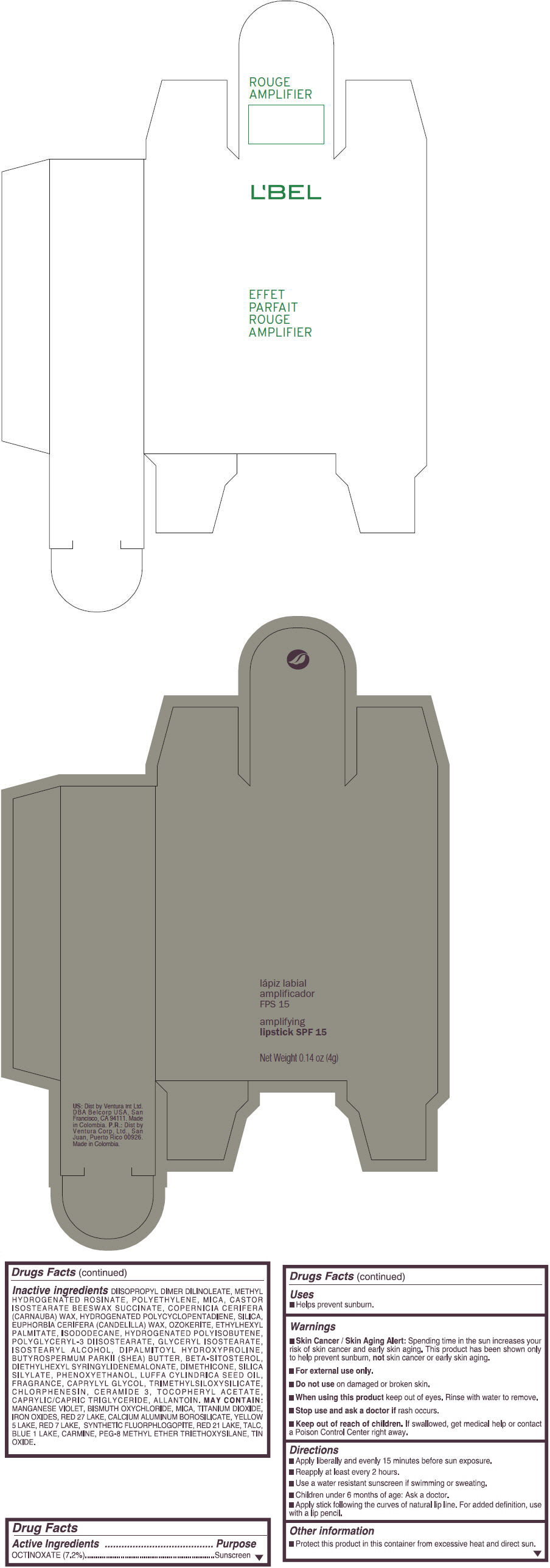
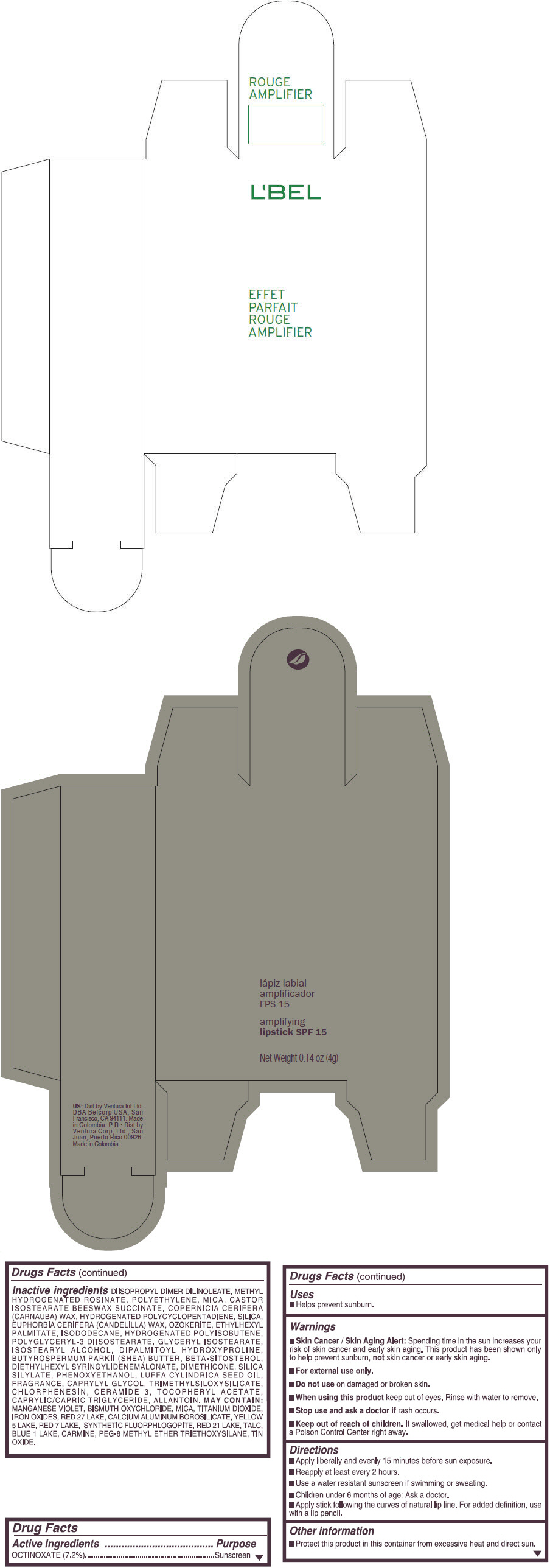
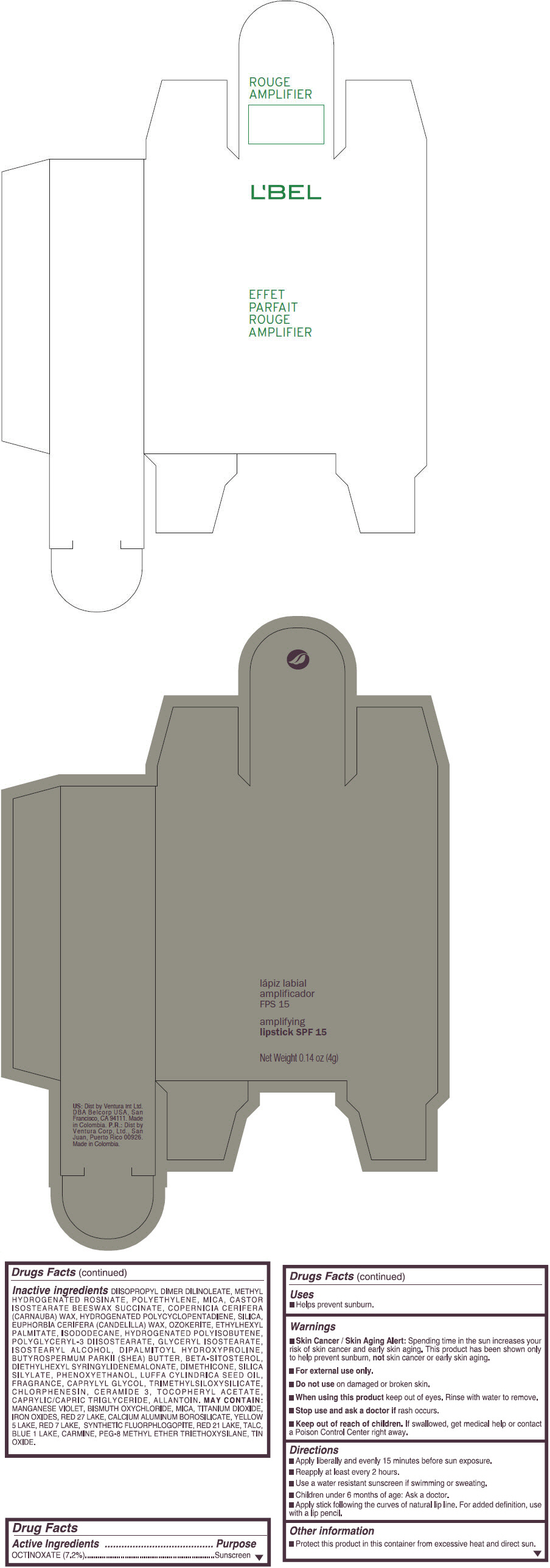
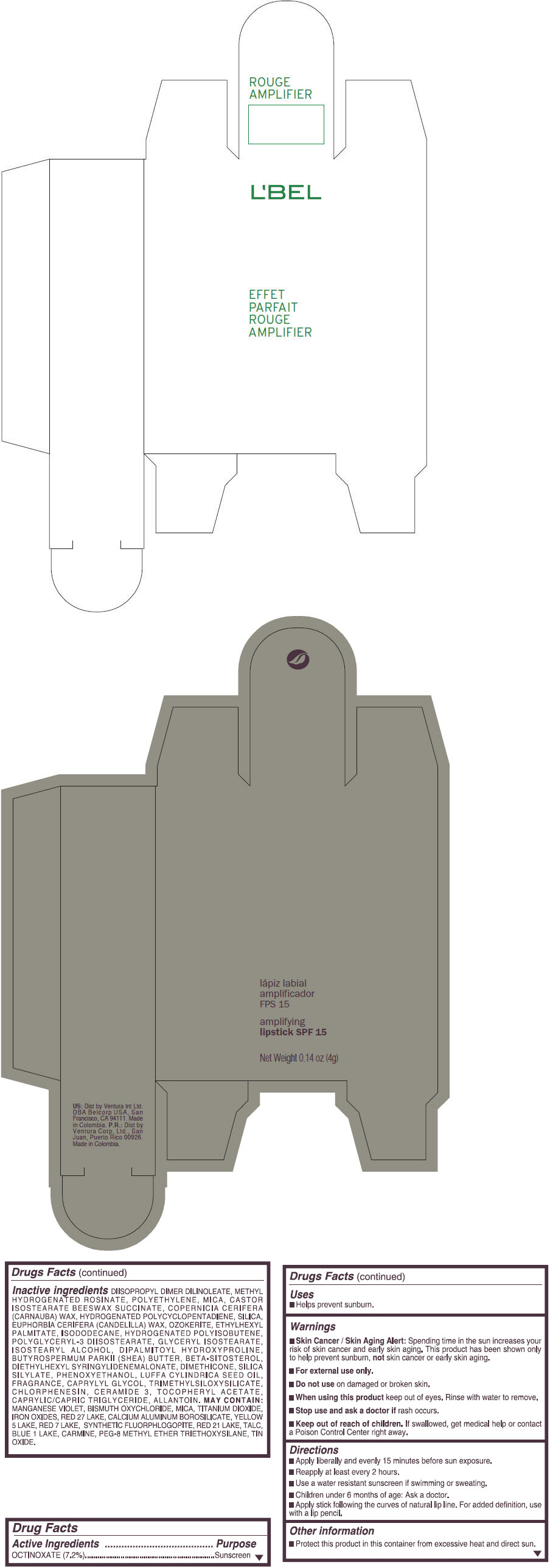
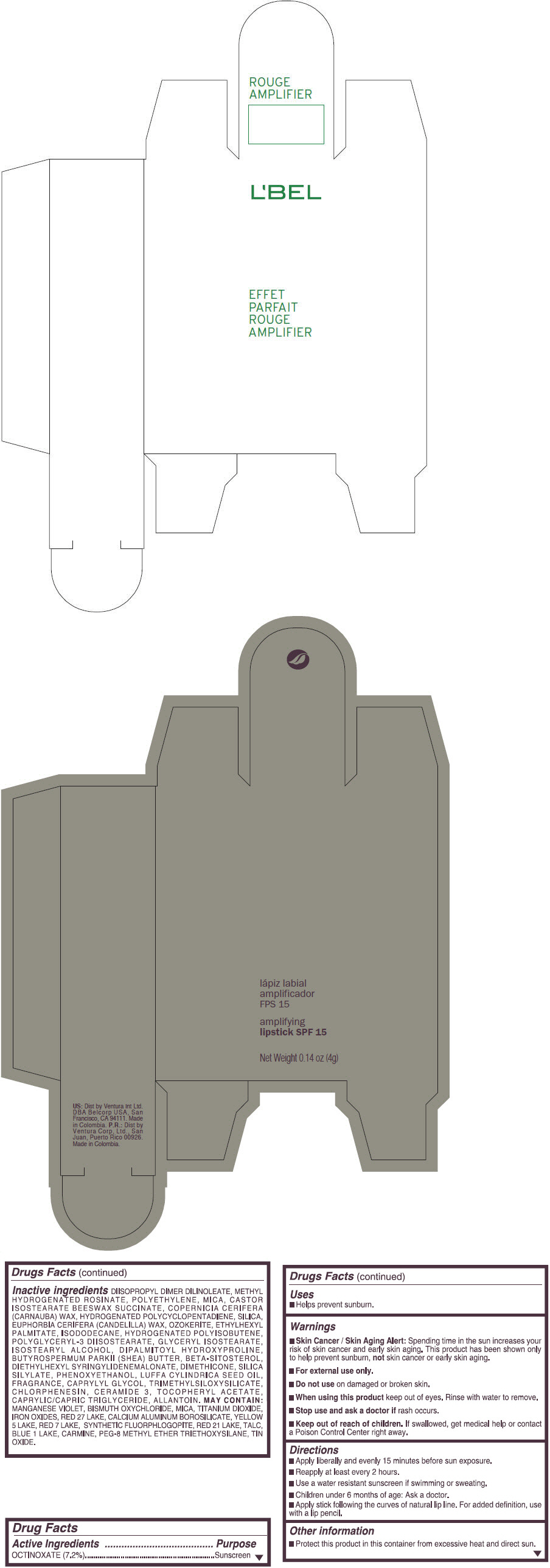
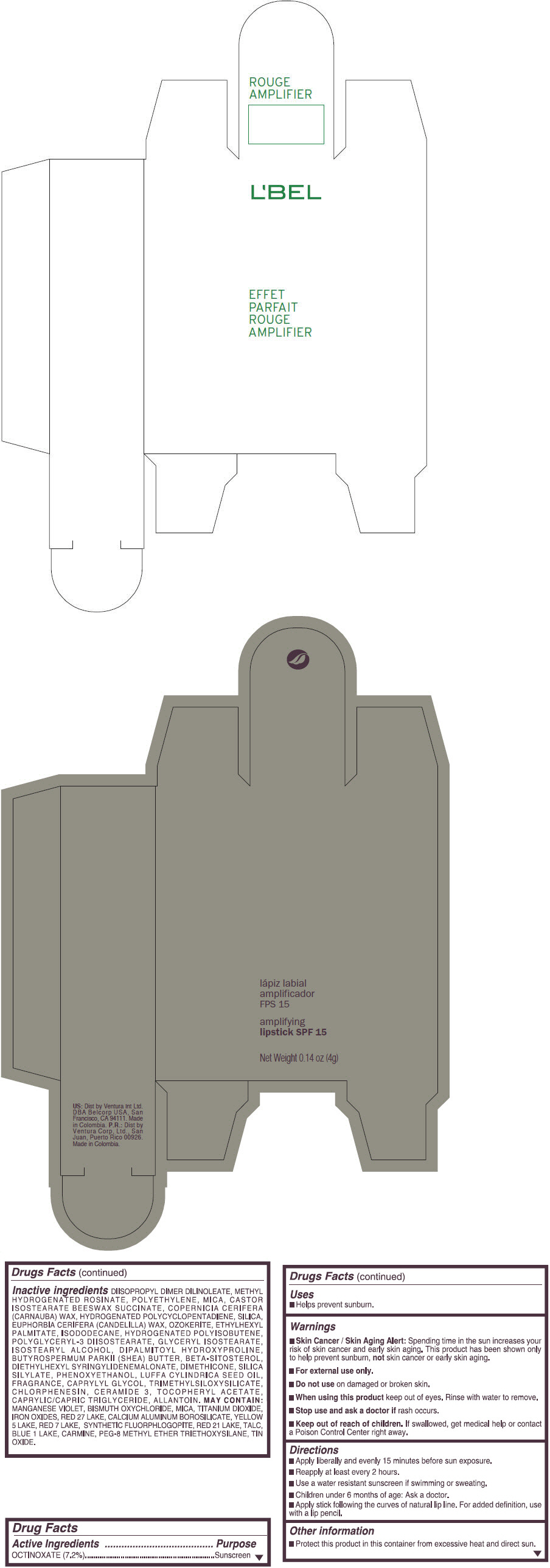
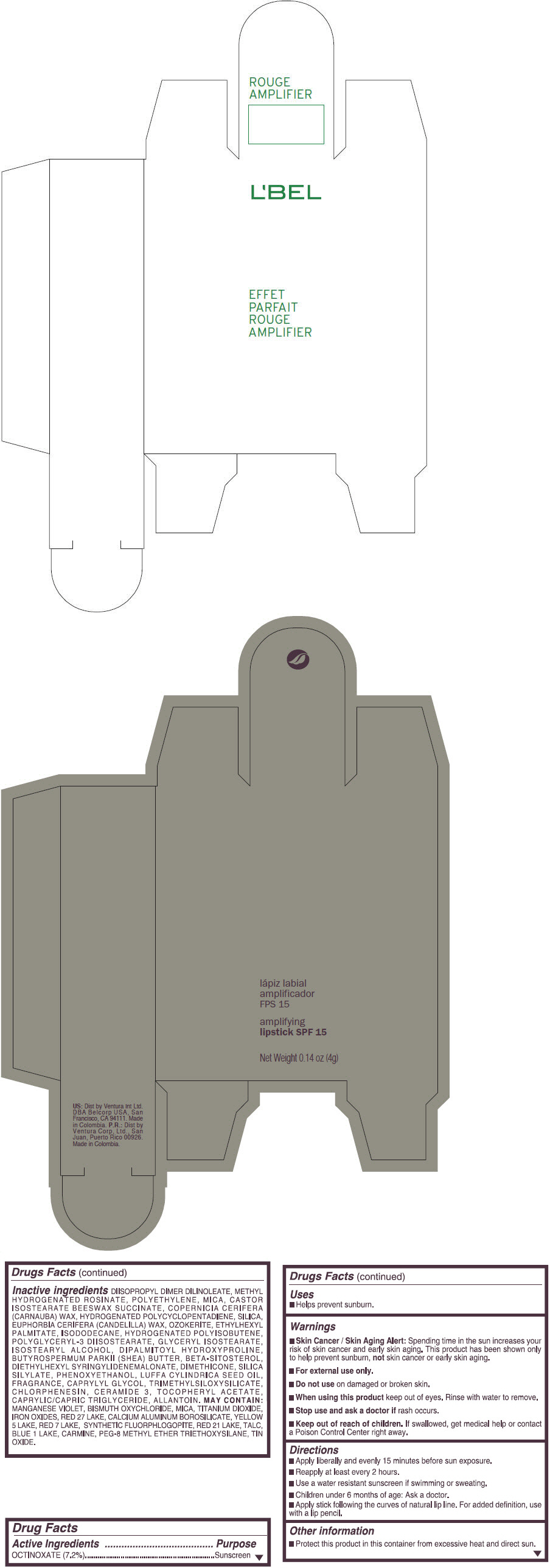
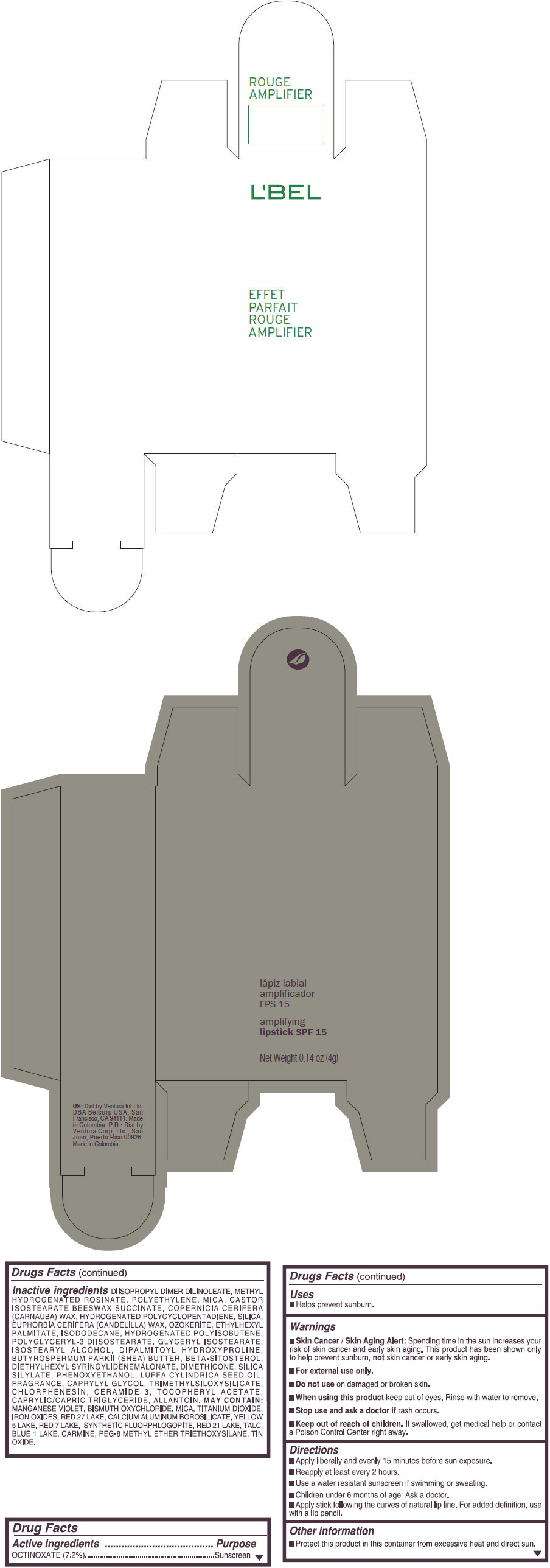
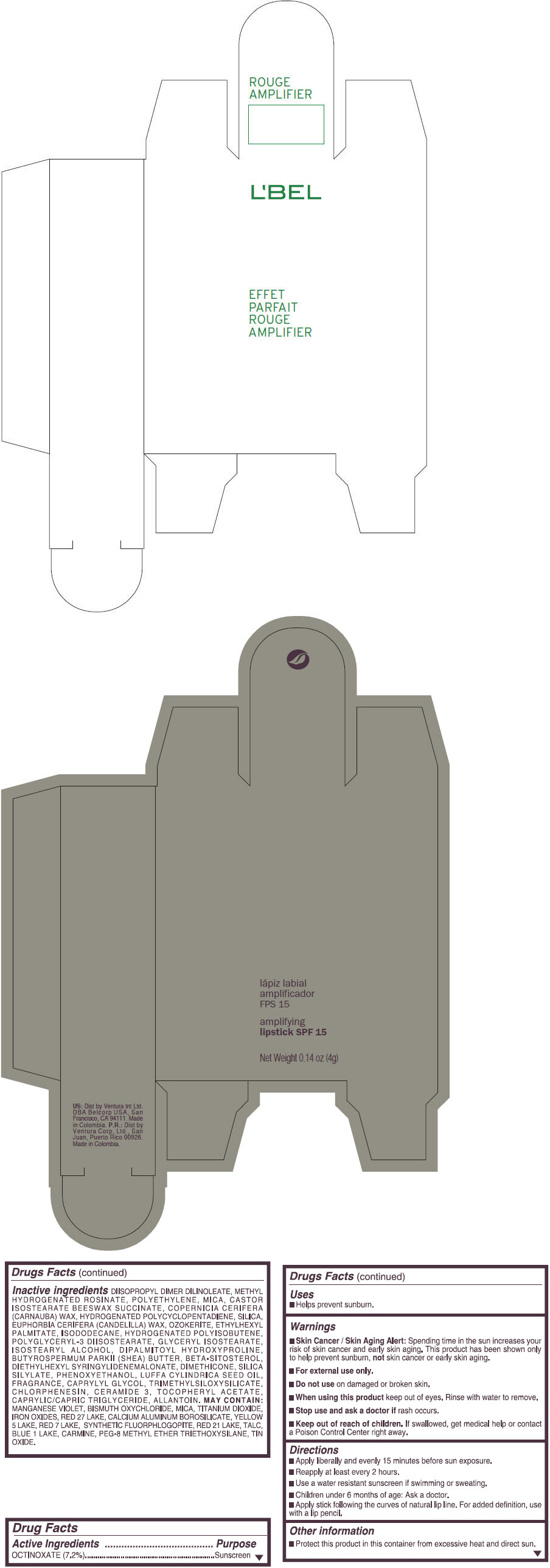
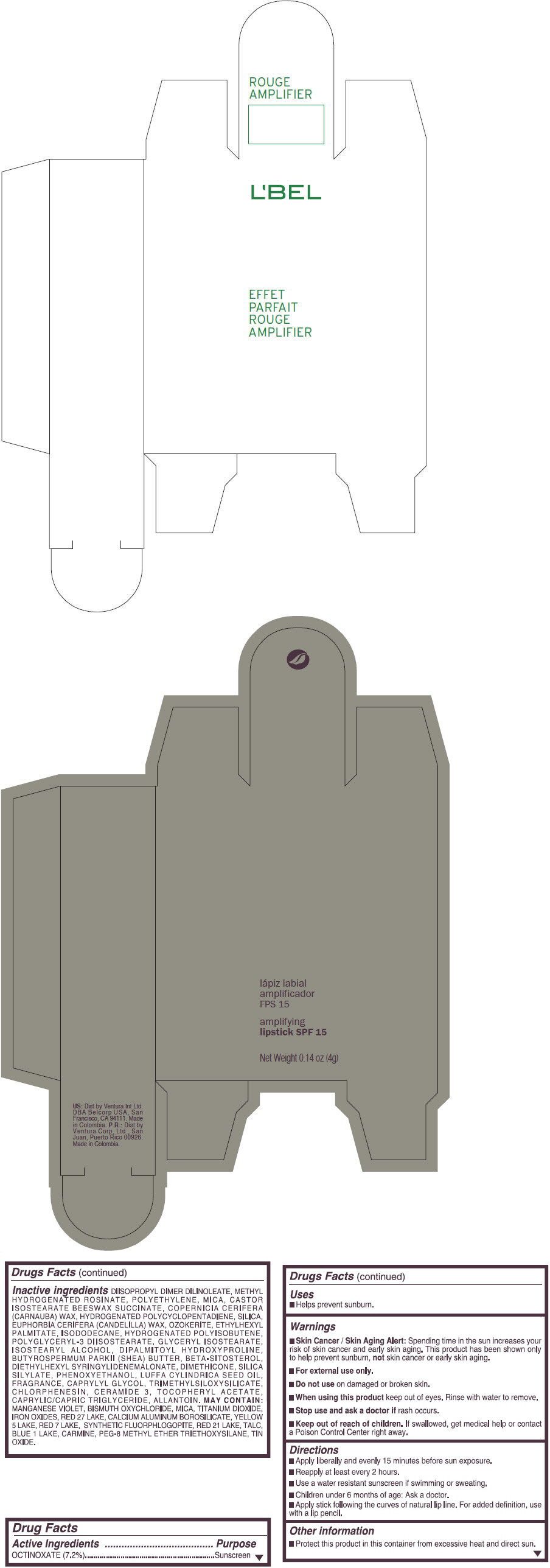
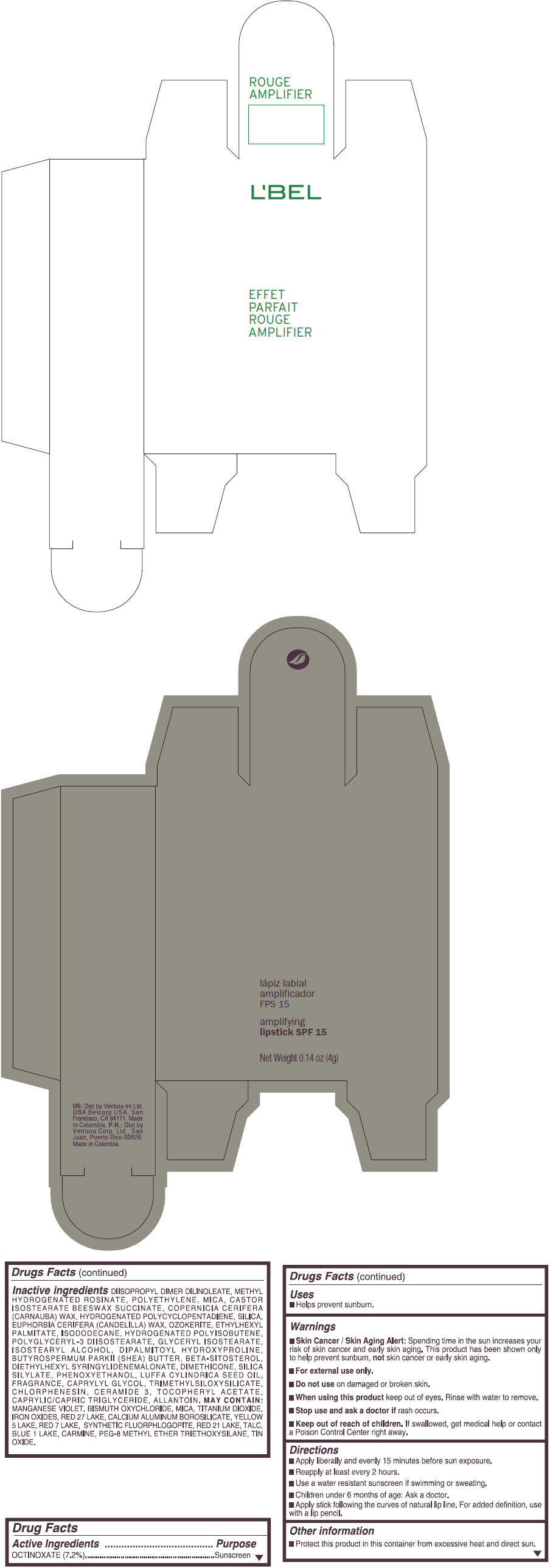
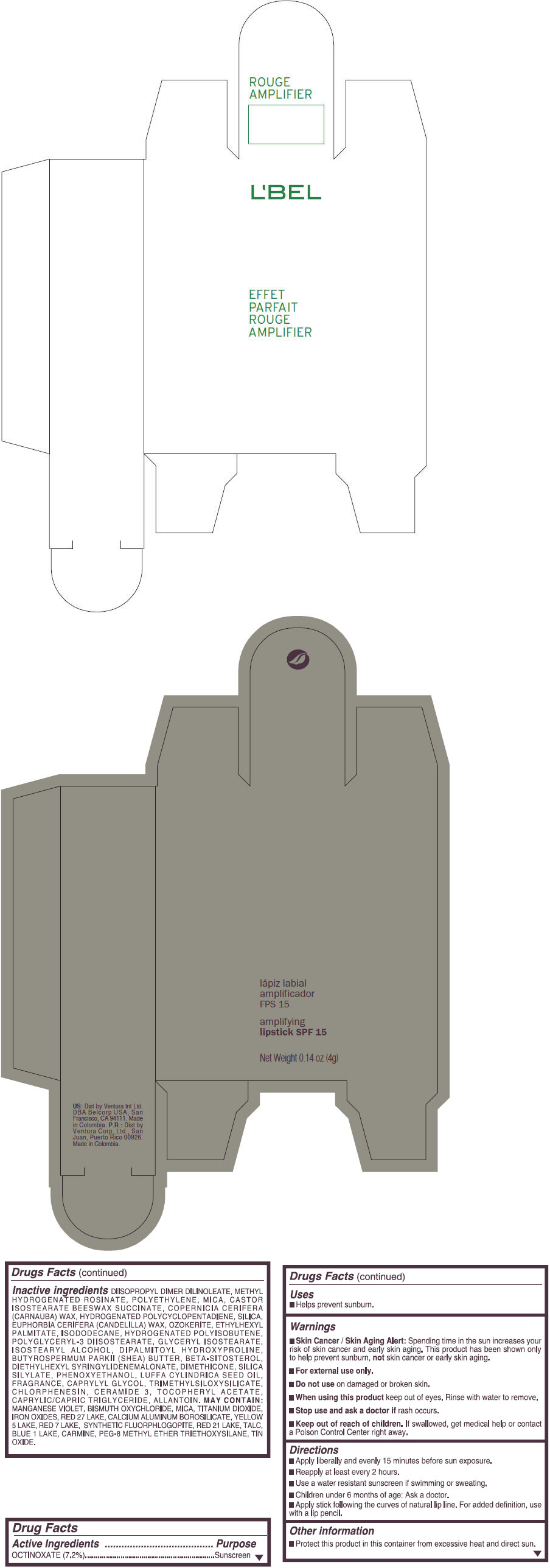
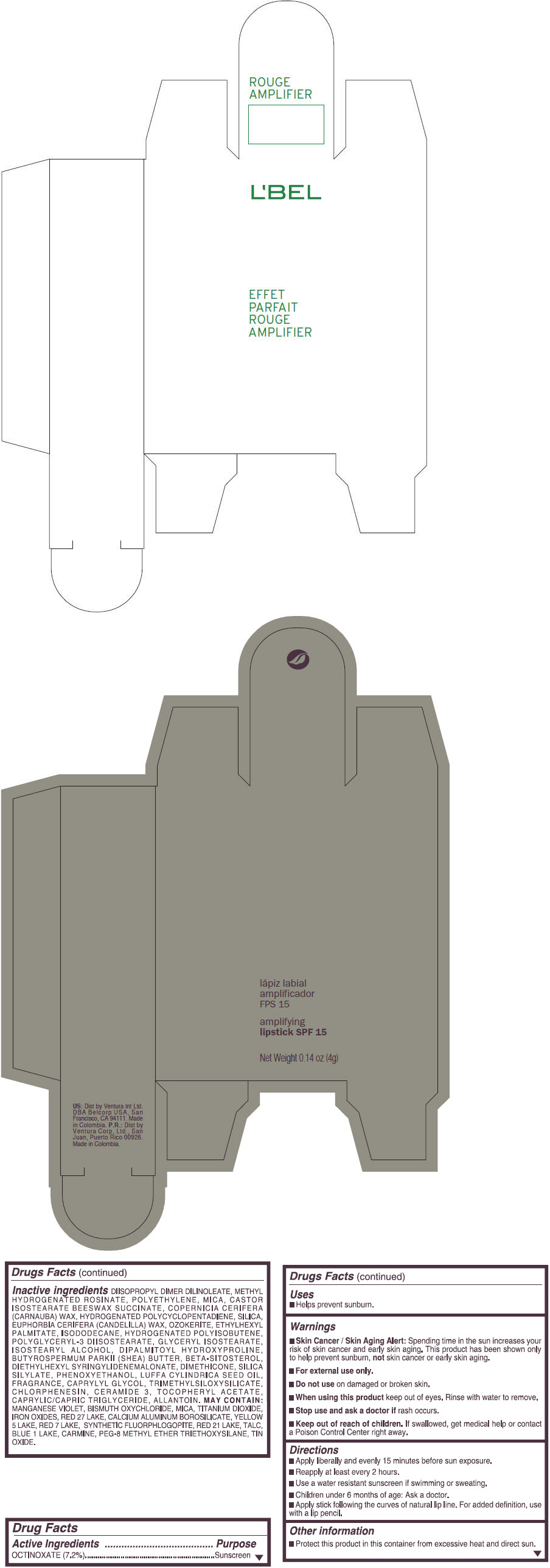
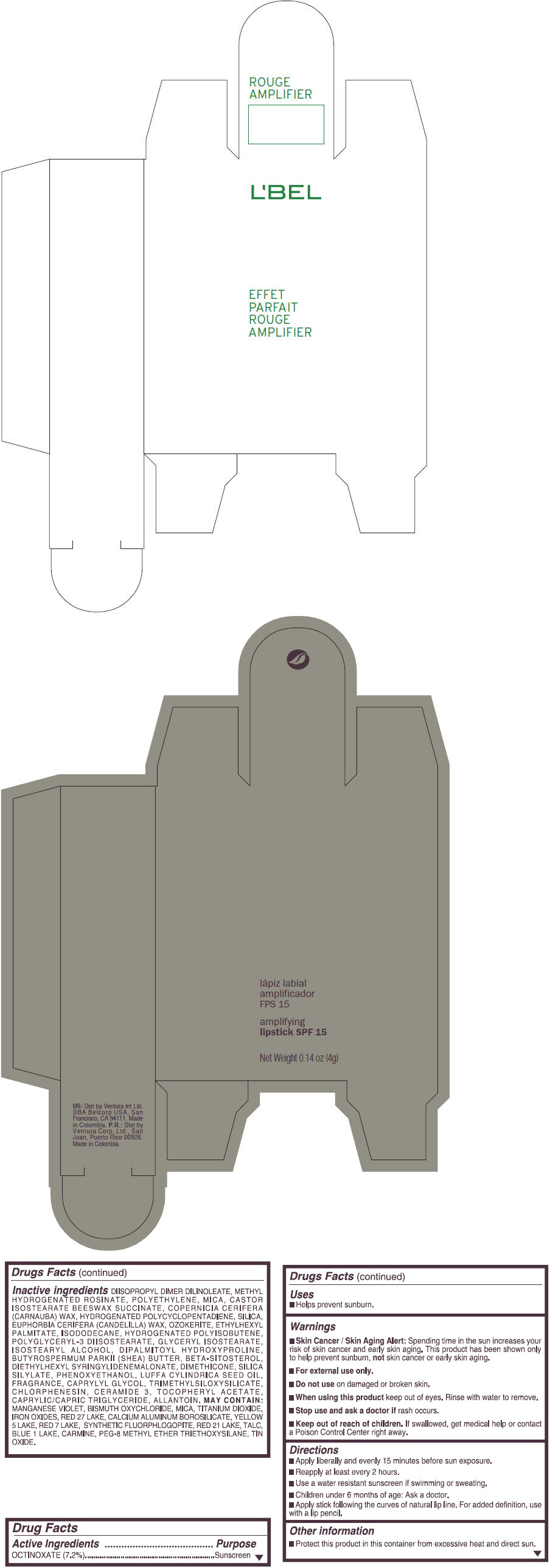
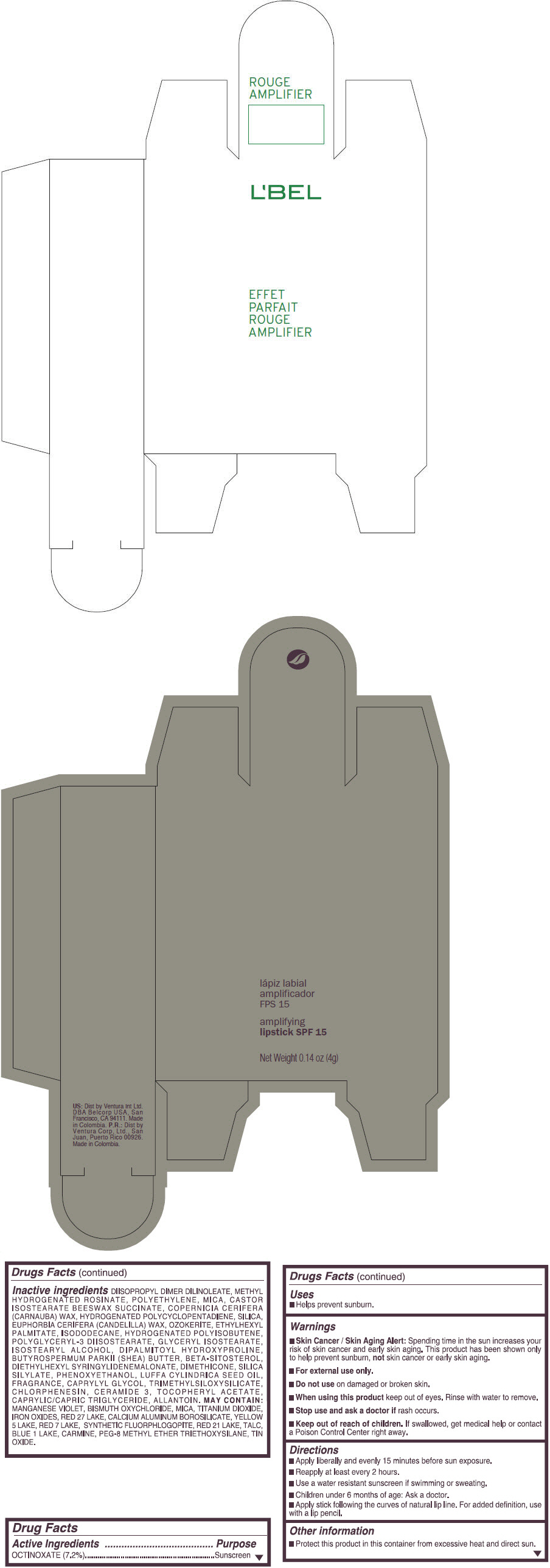
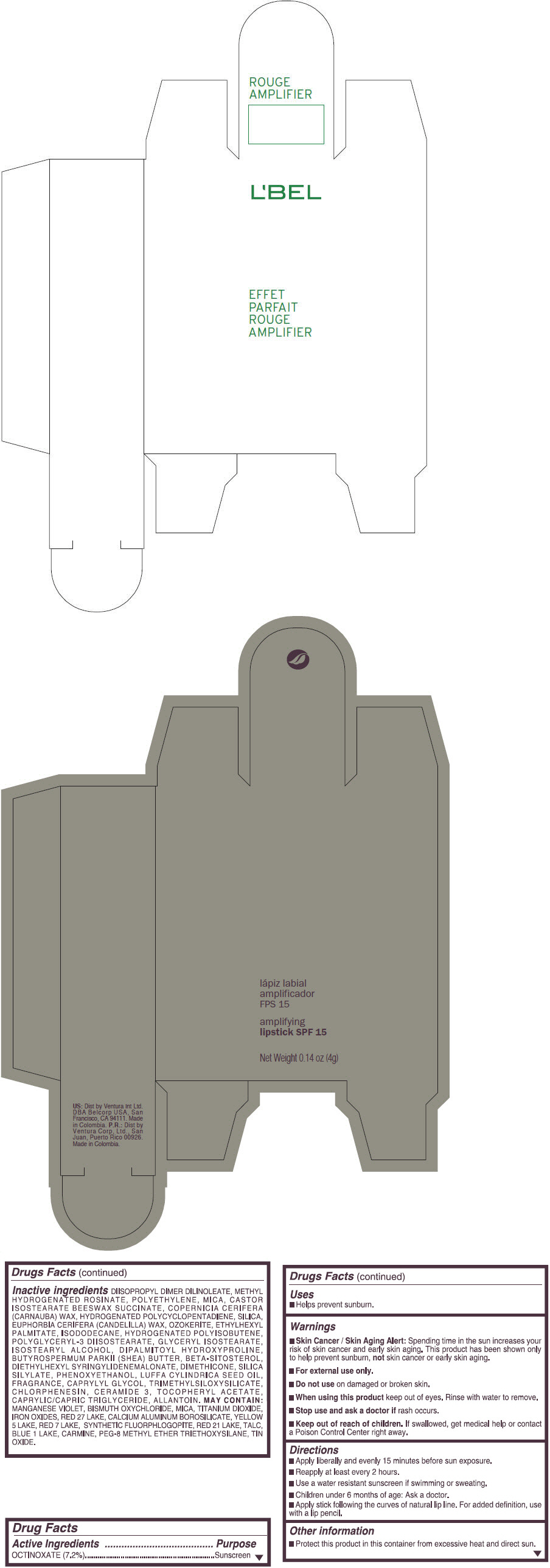
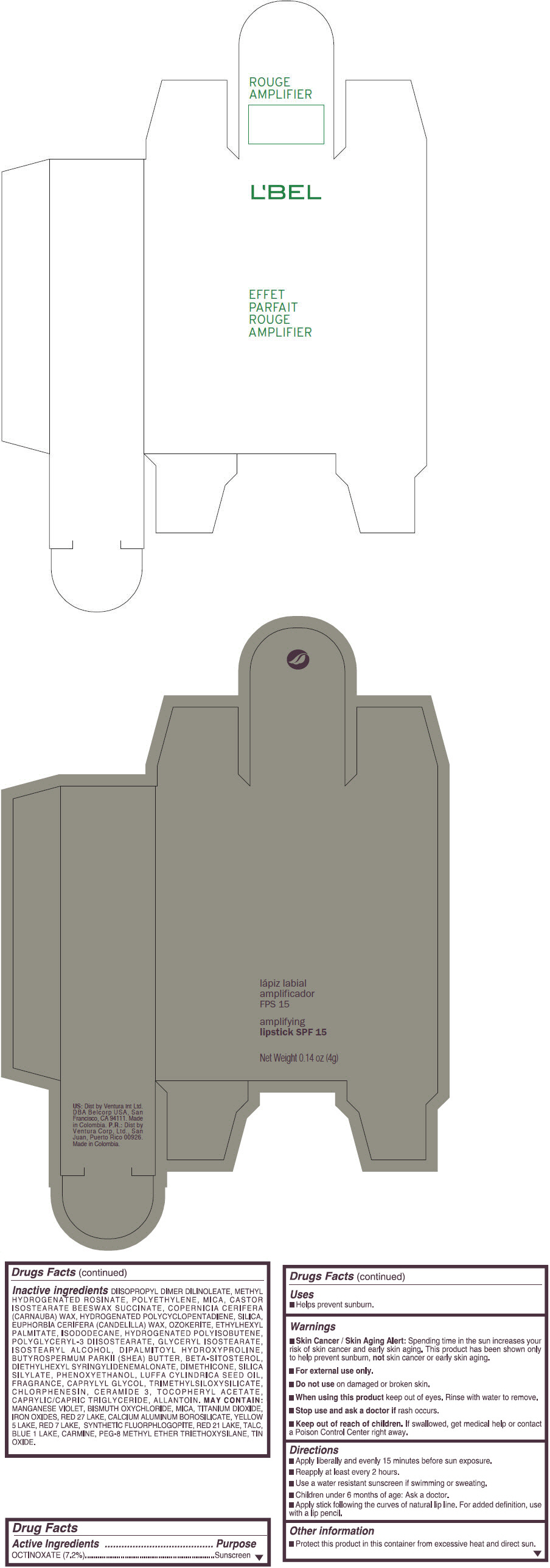
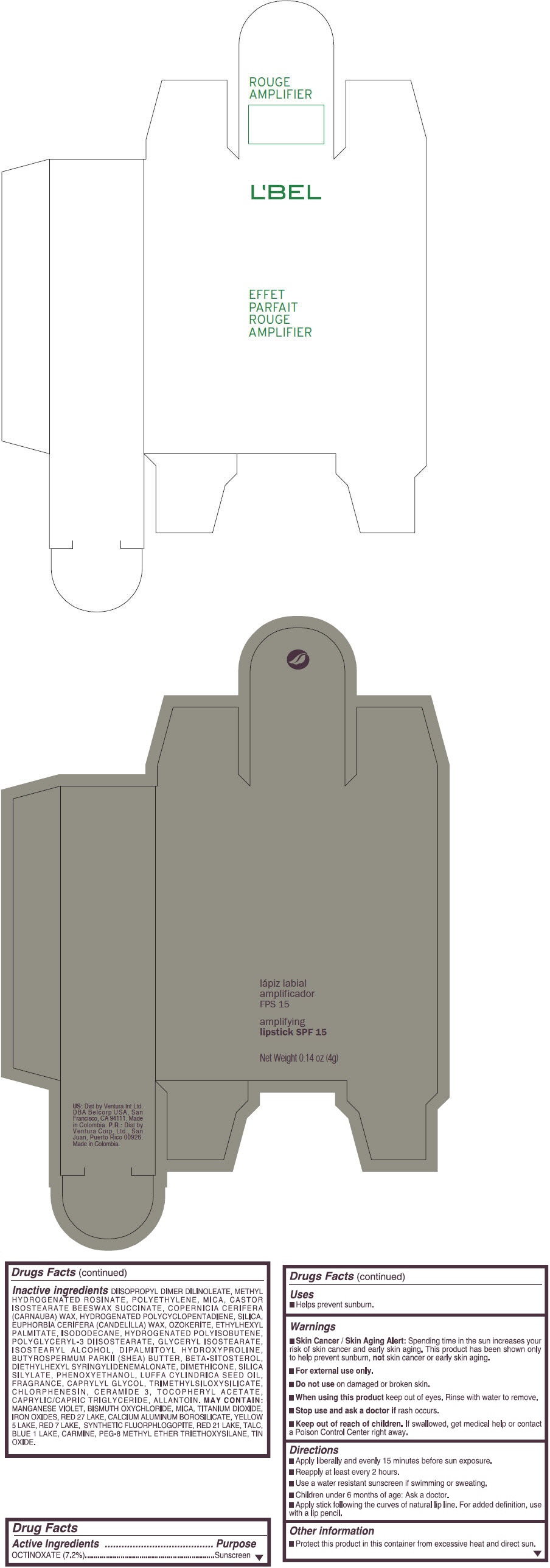
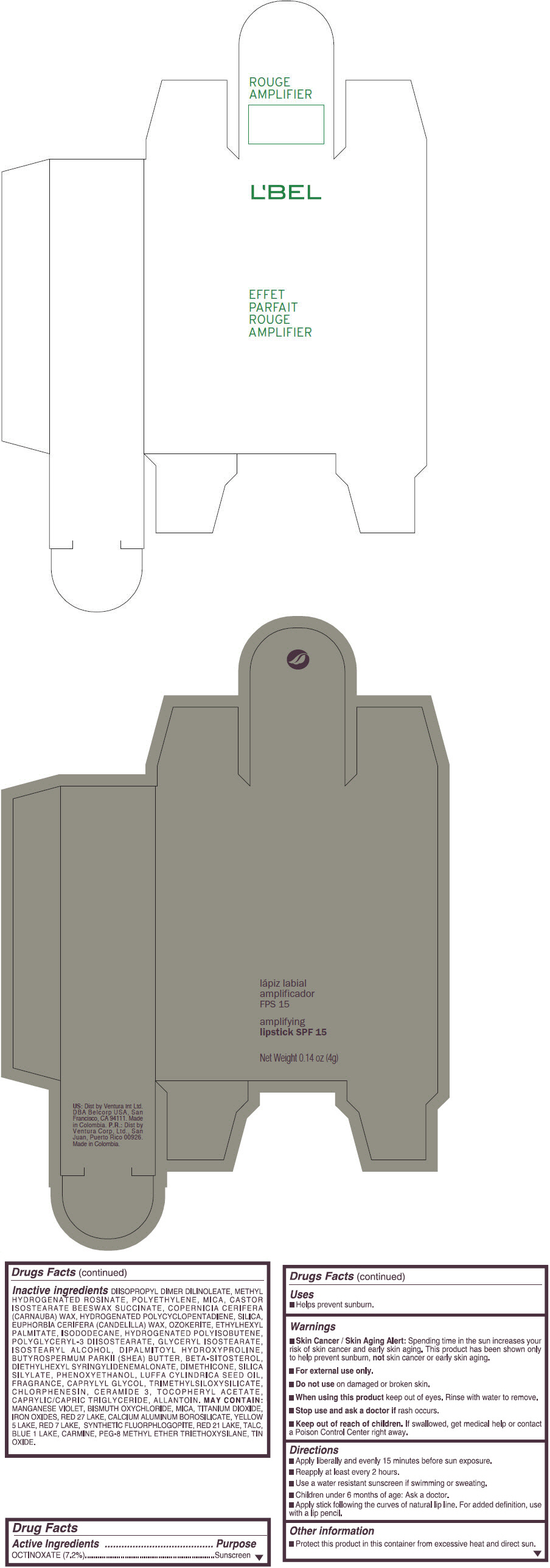
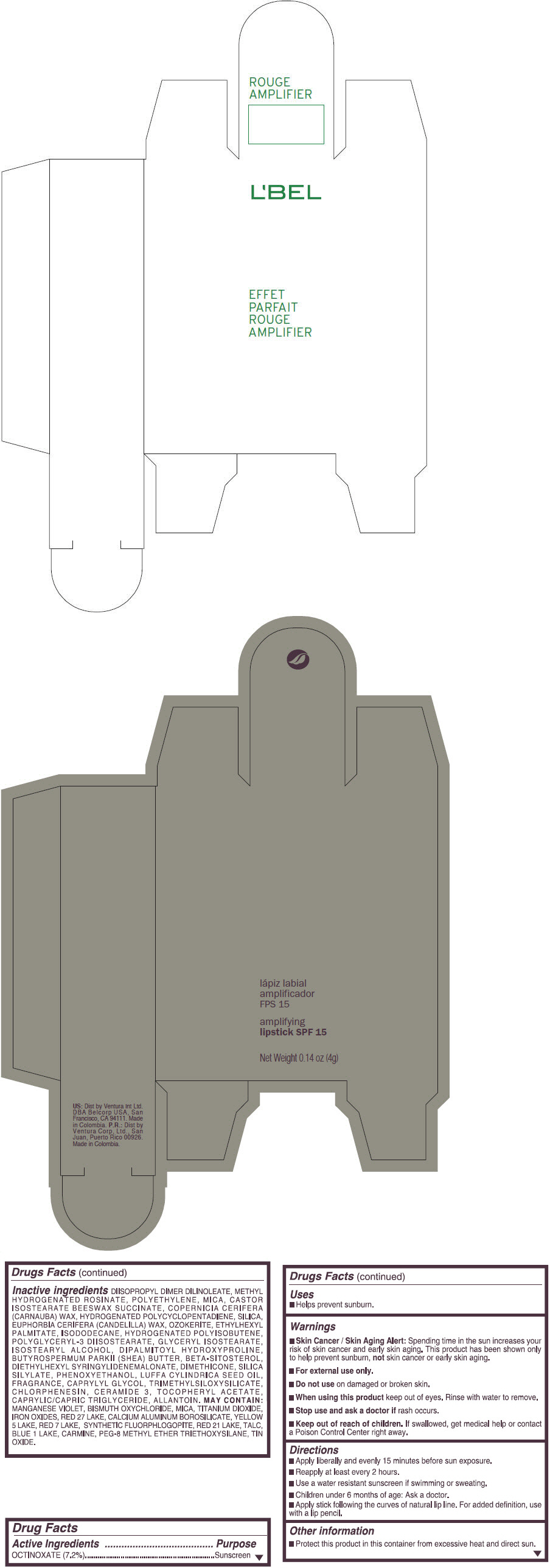
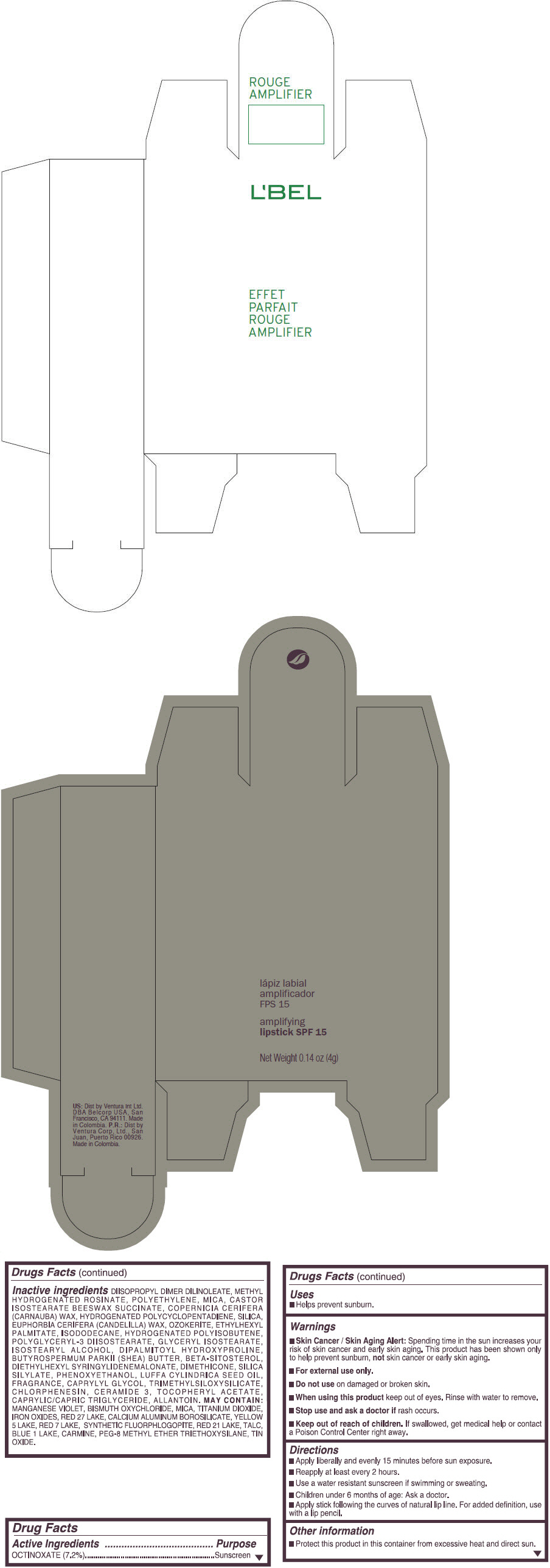
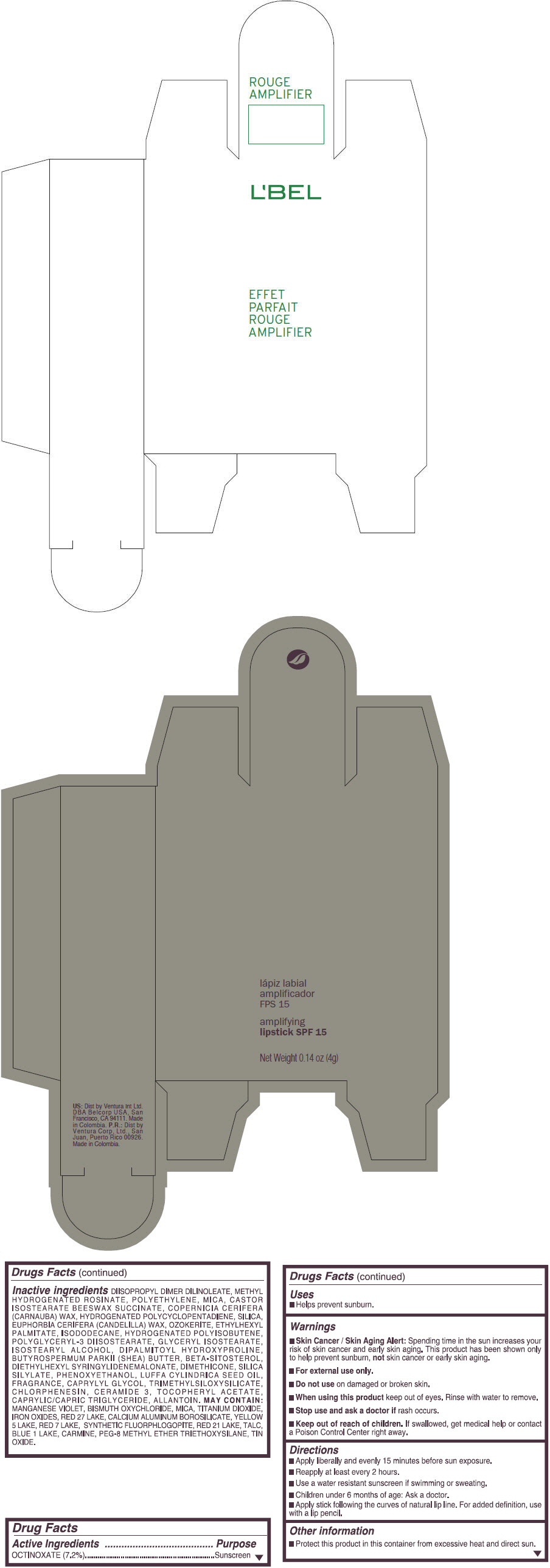
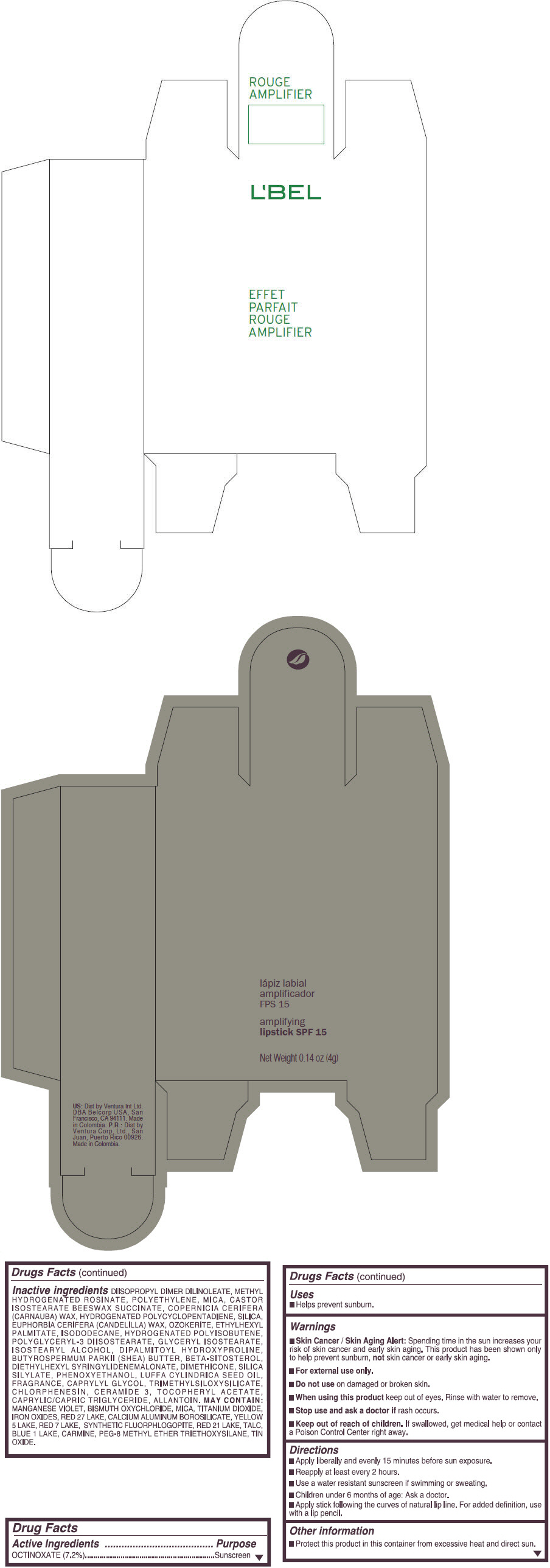
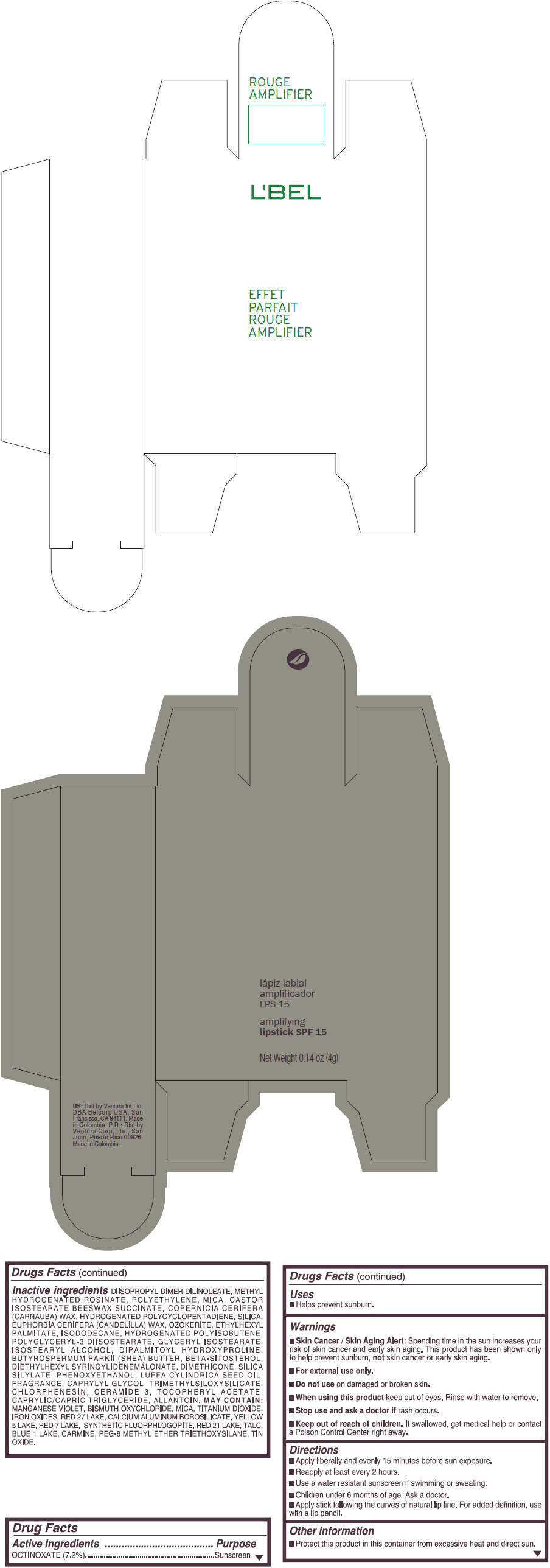
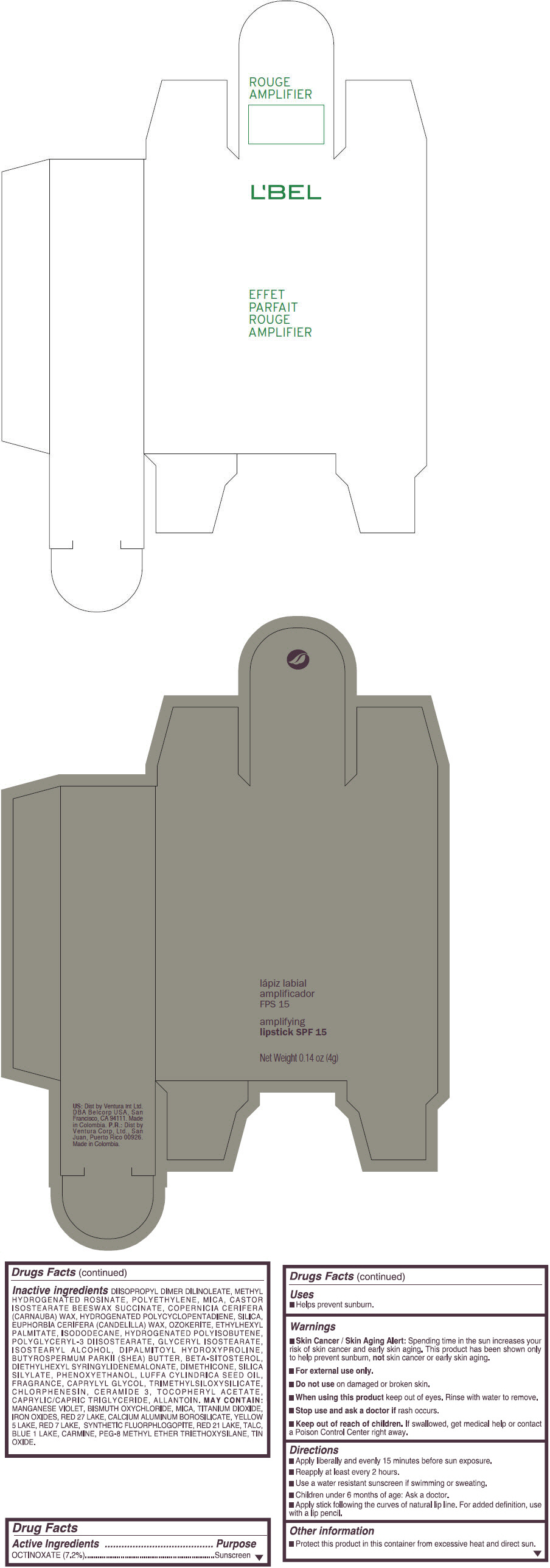
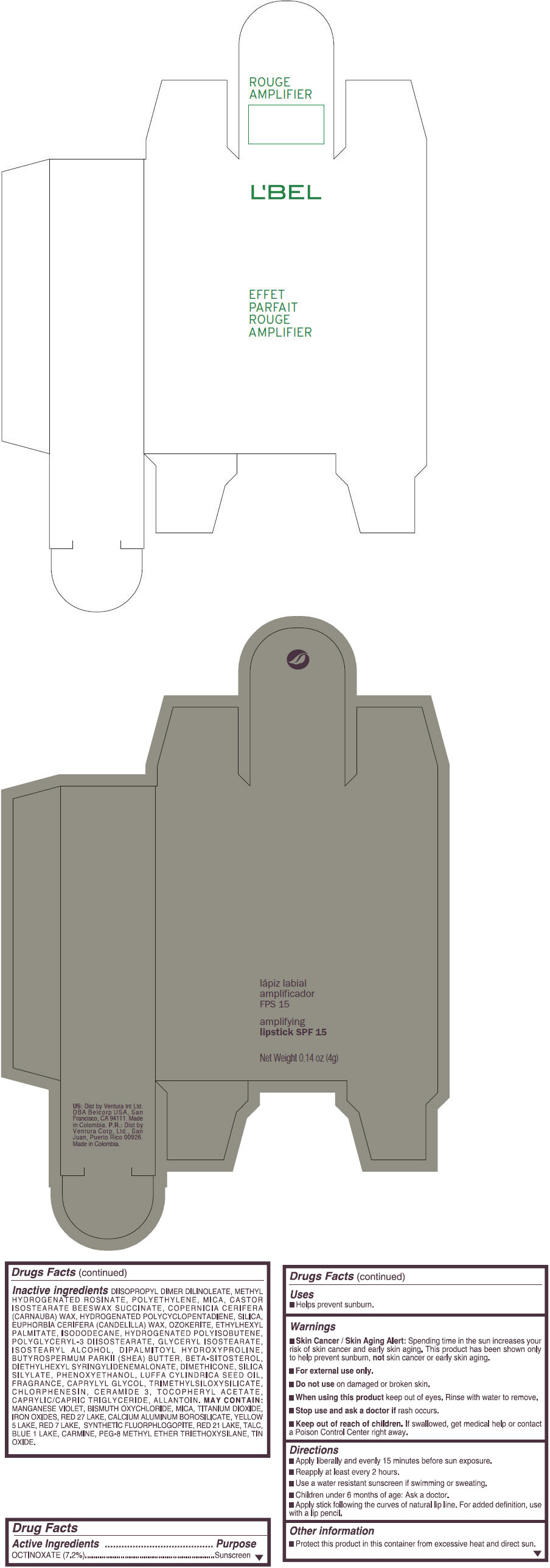
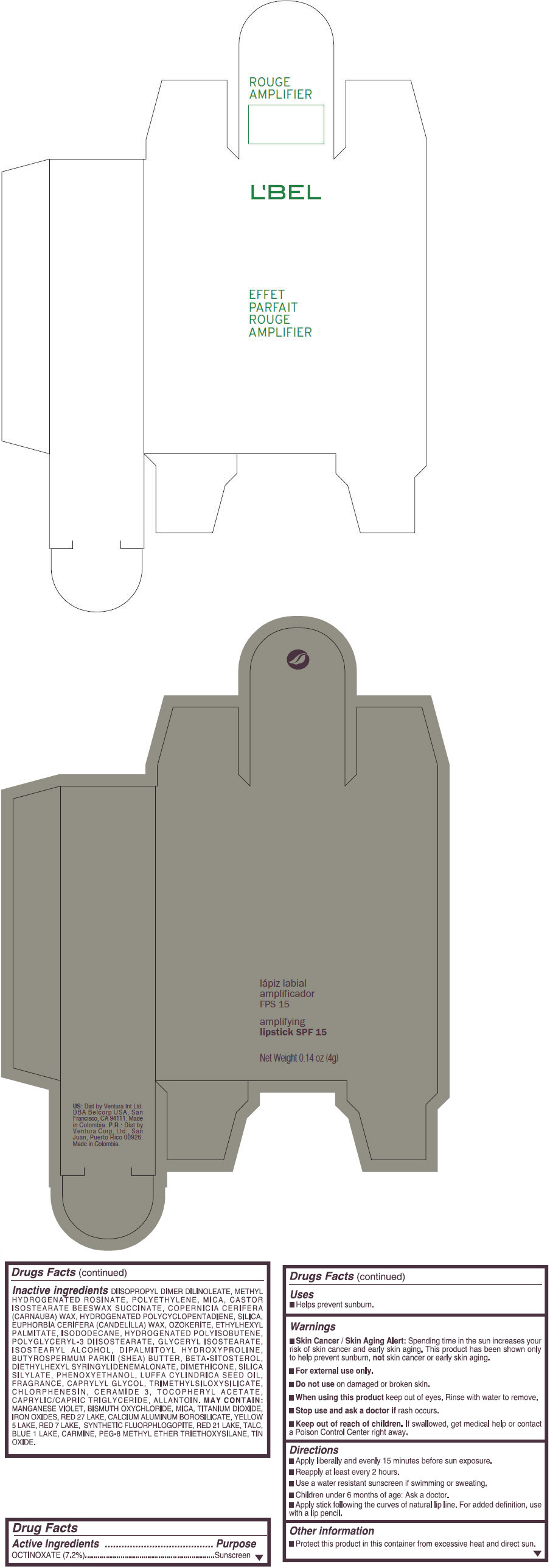
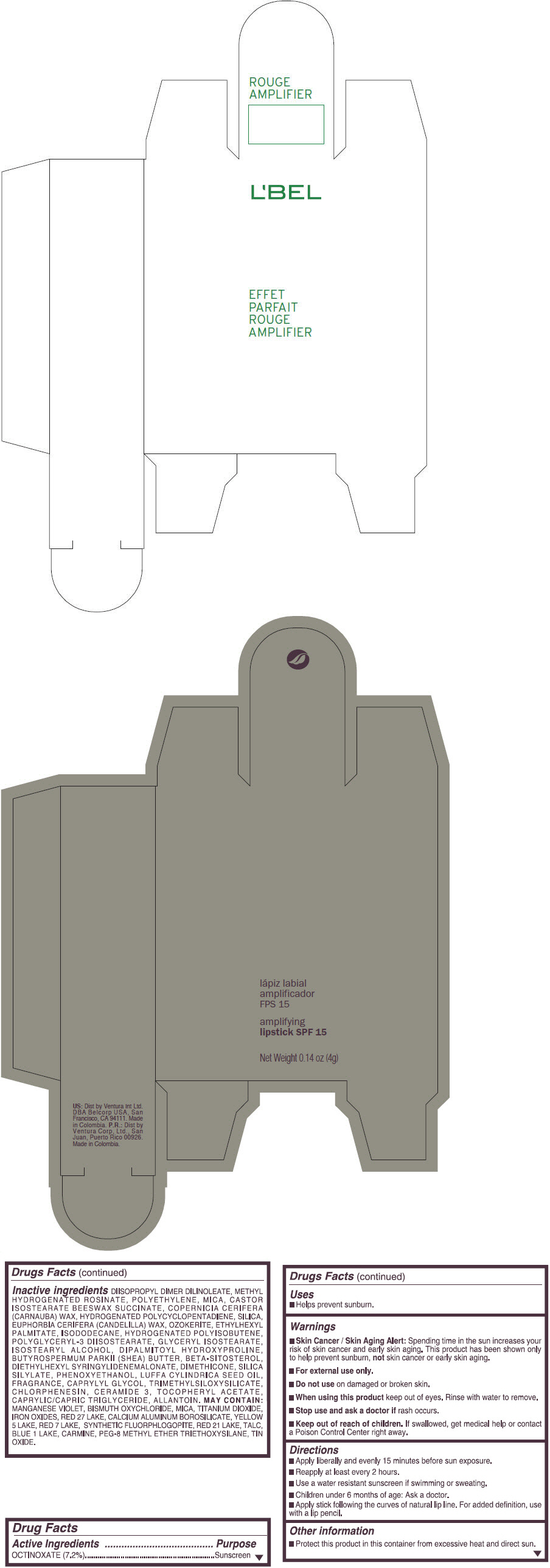
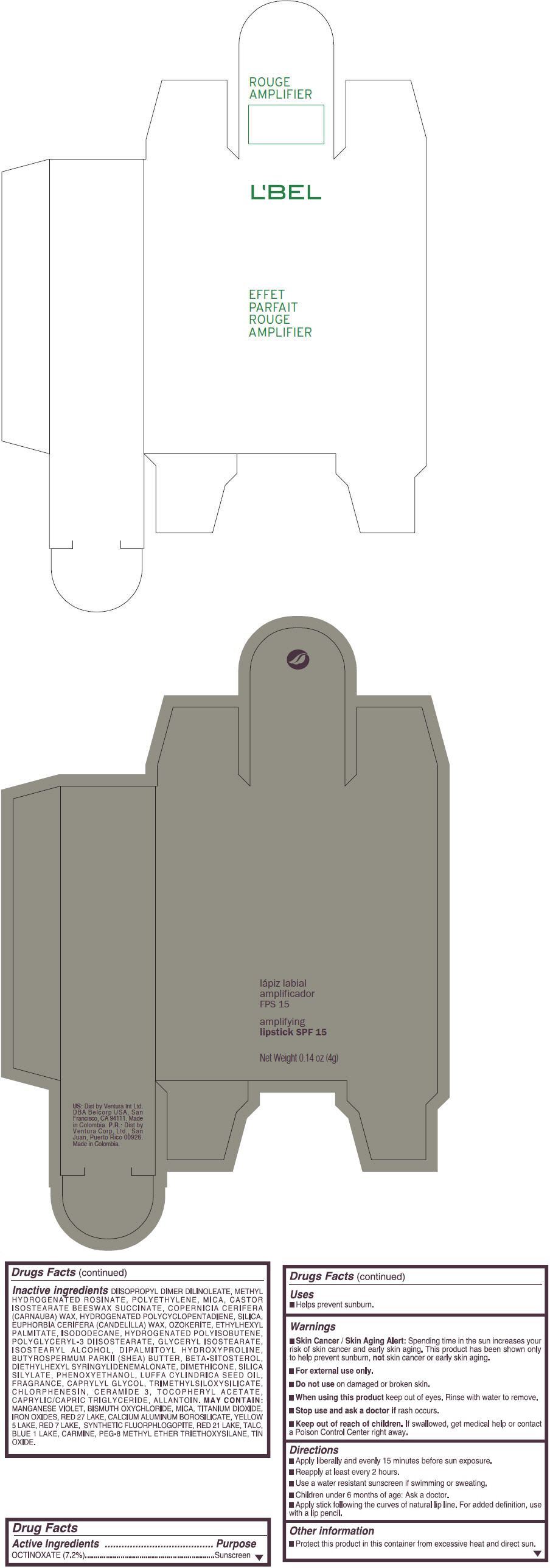
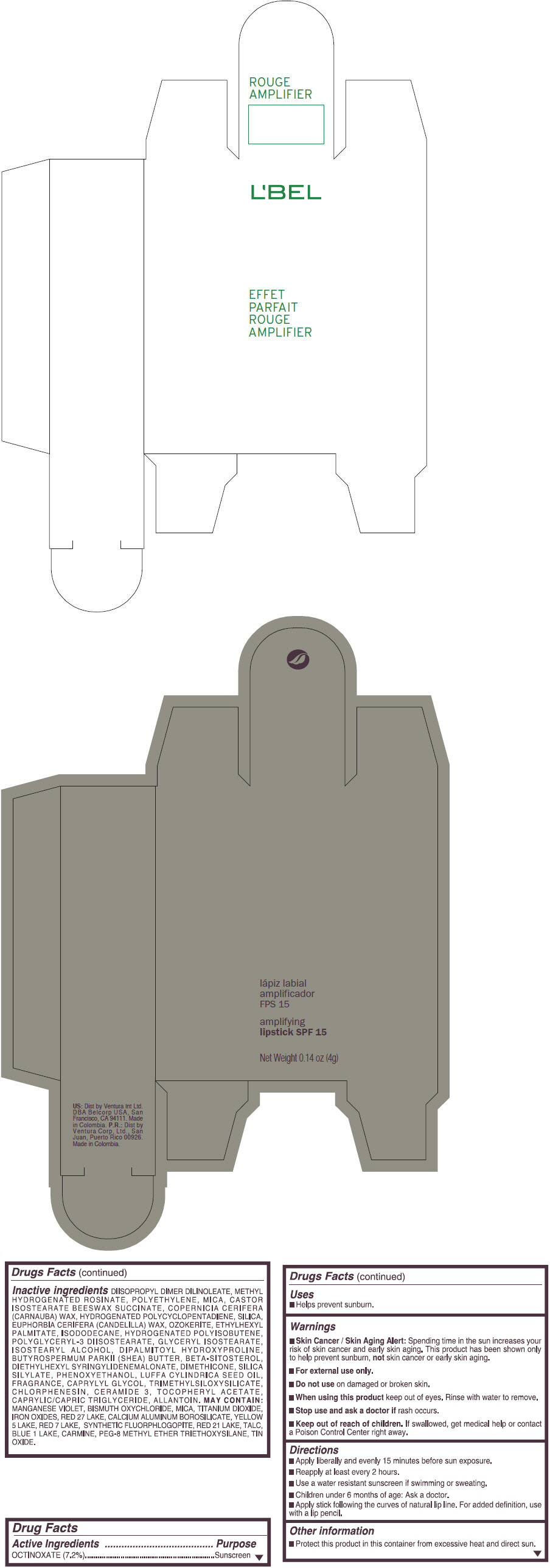
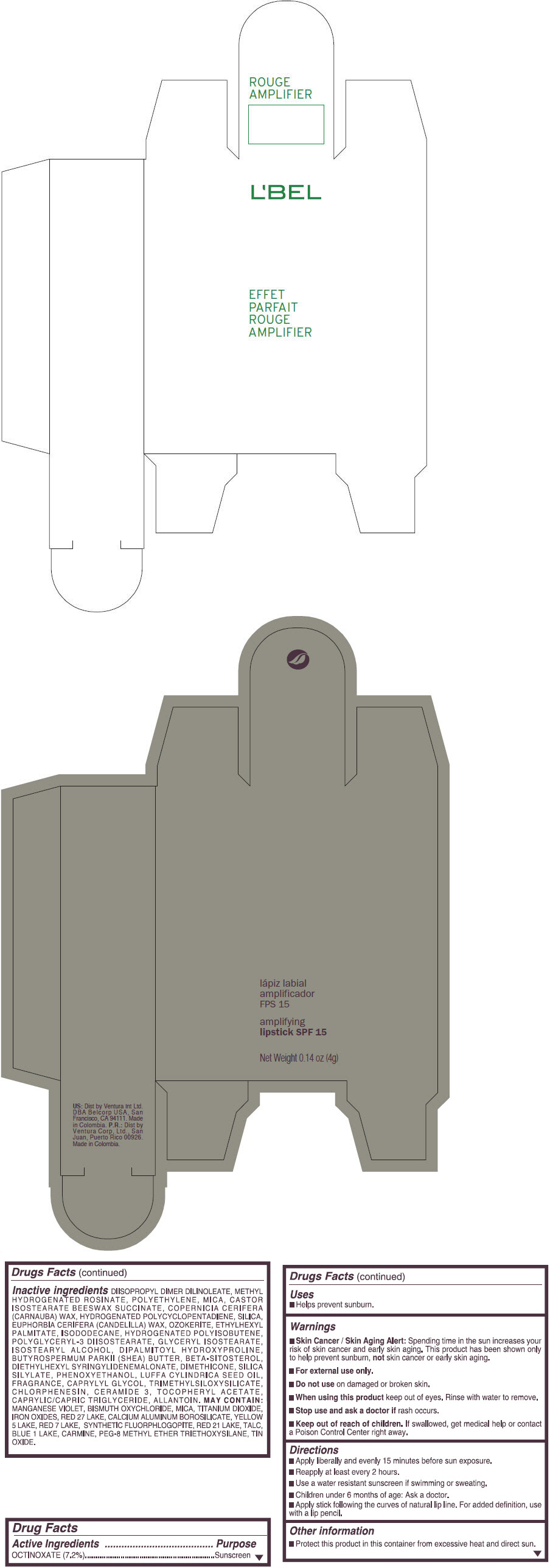
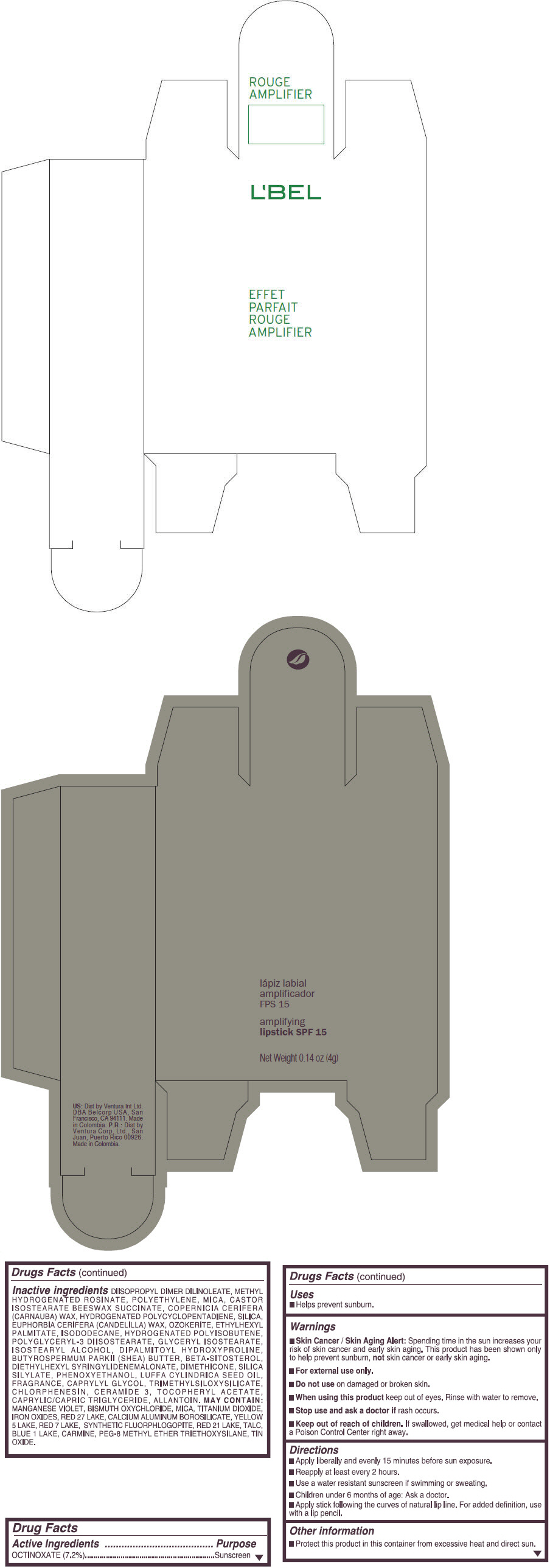
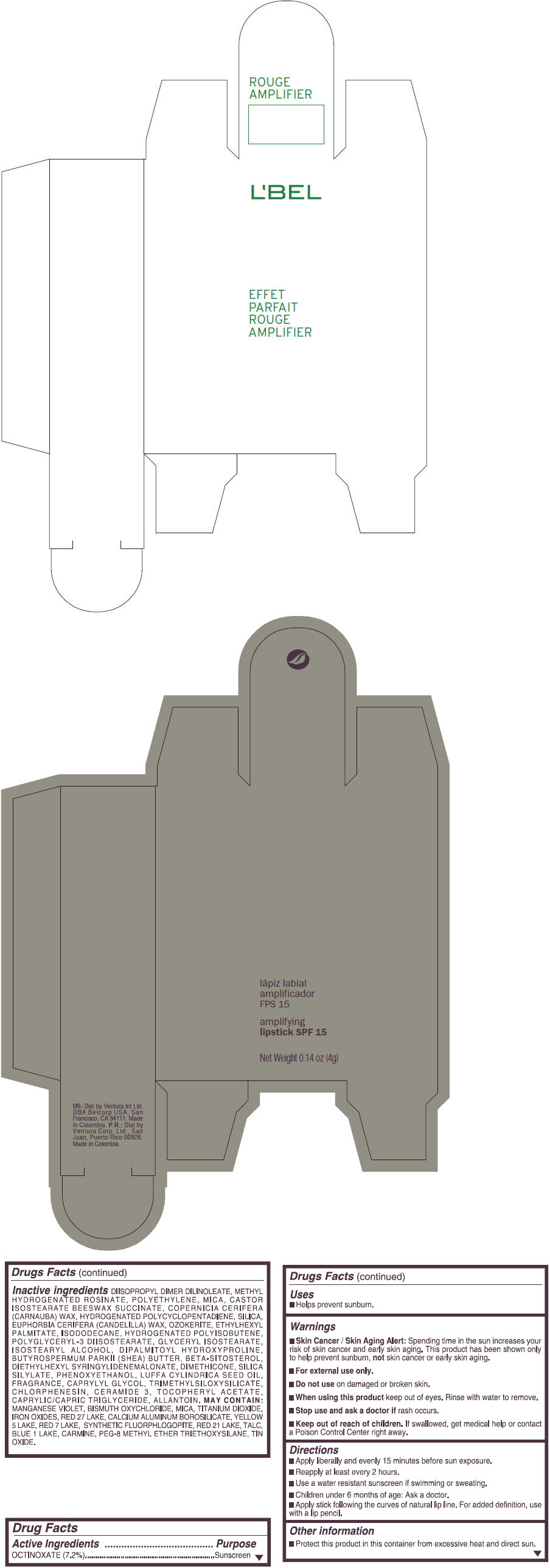
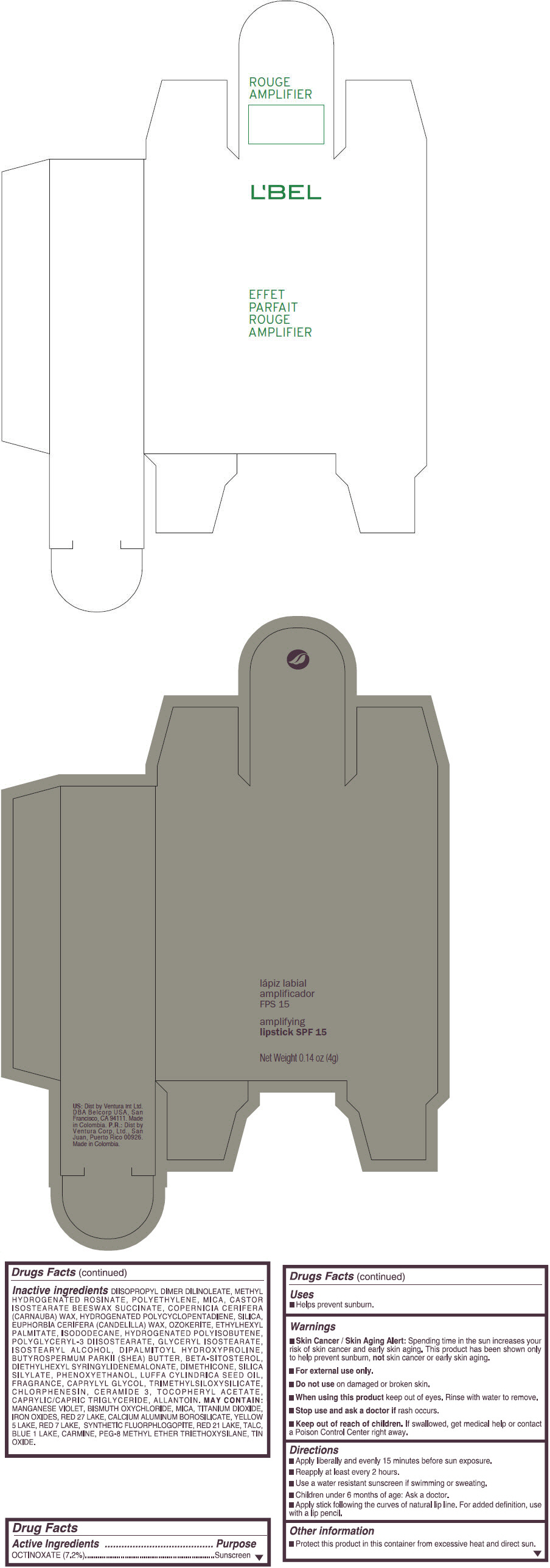
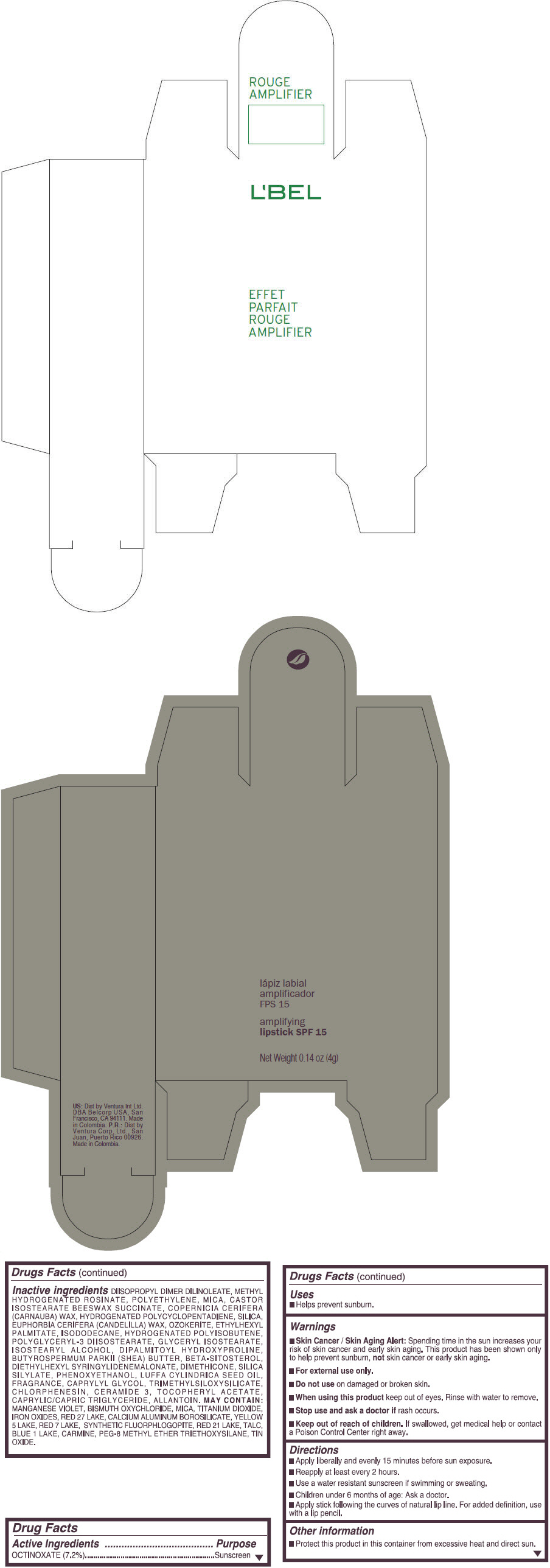
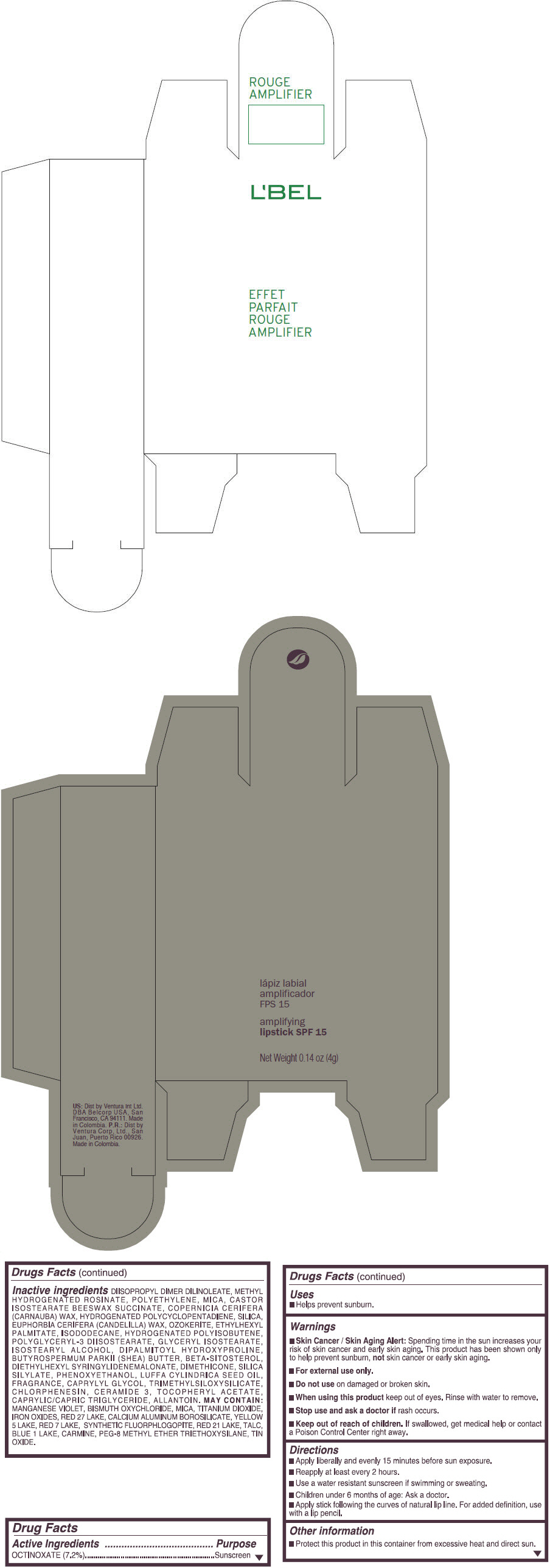
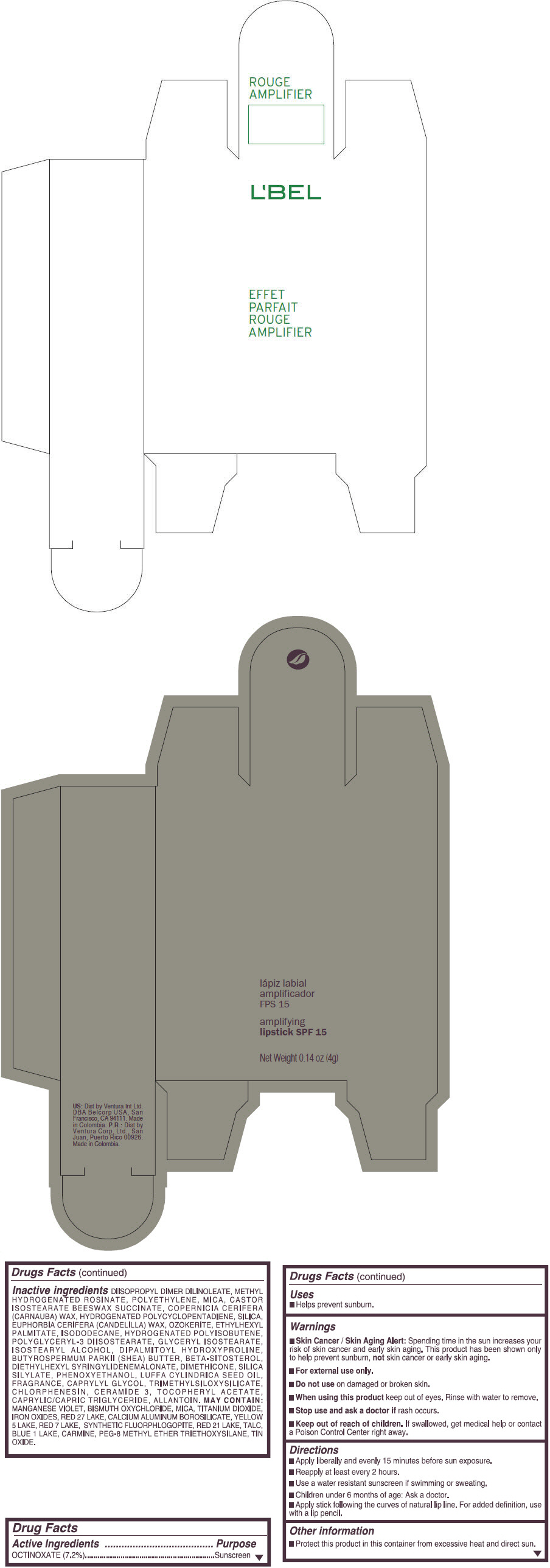
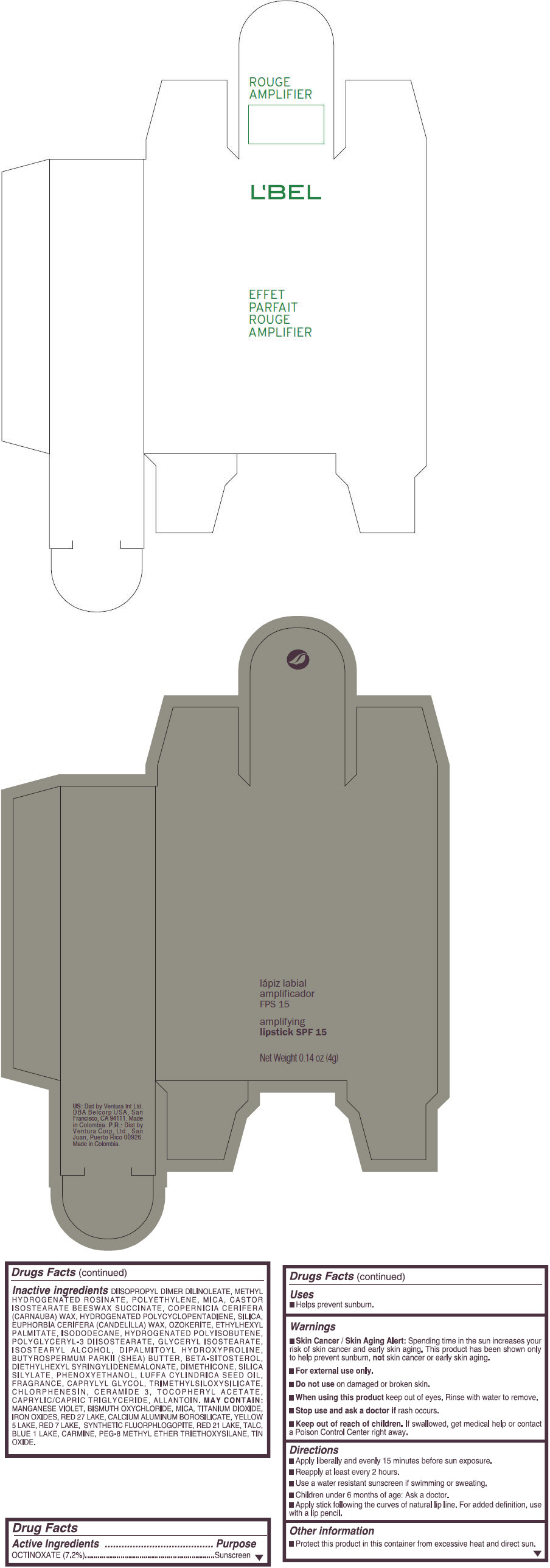
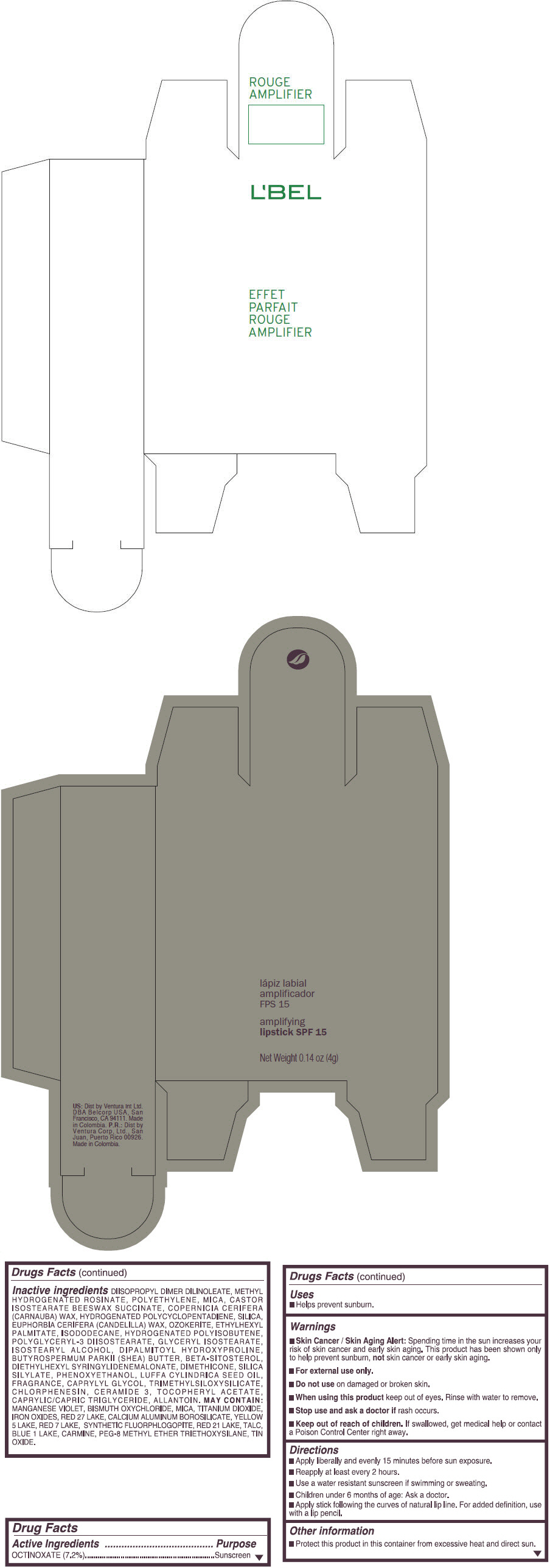
Inactive ingredients
DIISOPROPYL DIMER DILINOLEATE, METHYL HYDROGENATED ROSINATE, POLYETHYLENE, MICA, CASTOR ISOSTEARATE BEESWAX SUCCINATE, COPERNICIA CERIFERA (CARNAUBA) WAX, HYDROGENATED POLYCYCLOPENTADIENE, SILICA, EUPHORBIA CERIFERA (CANDELILLA) WAX, OZOKERITE, ETHYLHEXYL PALMITATE, ISODODECANE, HYDROGENATED POLYISOBUTENE, POLYGLYCERYL-3 DIISOSTEARATE, GLYCERYL ISOSTEARATE, ISOSTEARYL ALCOHOL, DIPALMITOYL HYDROXYPROLINE, BUTYROSPERMUM PARKII (SHEA) BUTTER, BETA-SITOSTEROL, DIETHYLHEXYL SYRINGYLIDENEMALONATE, DIMETHICONE, SILICA SILYLATE, PHENOXYETHANOL, LUFFA CYLINDRICA SEED OIL, FRAGRANCE, CAPRYLYL GLYCOL, TRIMETHYLSILOXYSILICATE, CHLORPHENESIN, CERAMIDE 3, TOCOPHERYL ACETATE, CAPRYLIC/CAPRIC TRIGLYCERIDE, ALLANTOIN.
MAY CONTAIN:
MANGANESE VIOLET, BISMUTH OXYCHLORIDE, MICA, TITANIUM DIOXIDE, IRON OXIDES, RED 27 LAKE, CALCIUM ALUMINUM BOROSILICATE, YELLOW 5 LAKE, RED 7 LAKE, SYNTHETIC FLUORPHLOGOPITE, RED 21 LAKE, TALC, BLUE 1 LAKE, CARMINE, PEG-8 METHYL ETHER TRIETHOXYSILANE, TIN OXIDE. - SPL UNCLASSIFIED SECTION
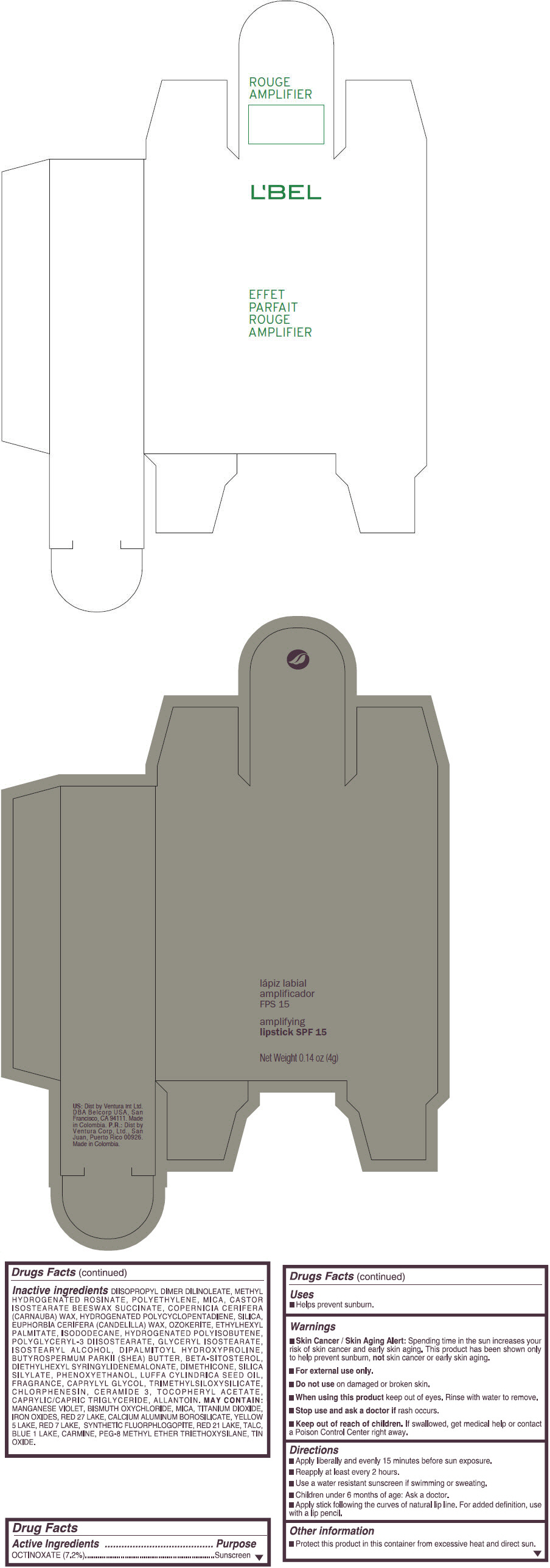
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROUGE GRANDIOSE) - RED
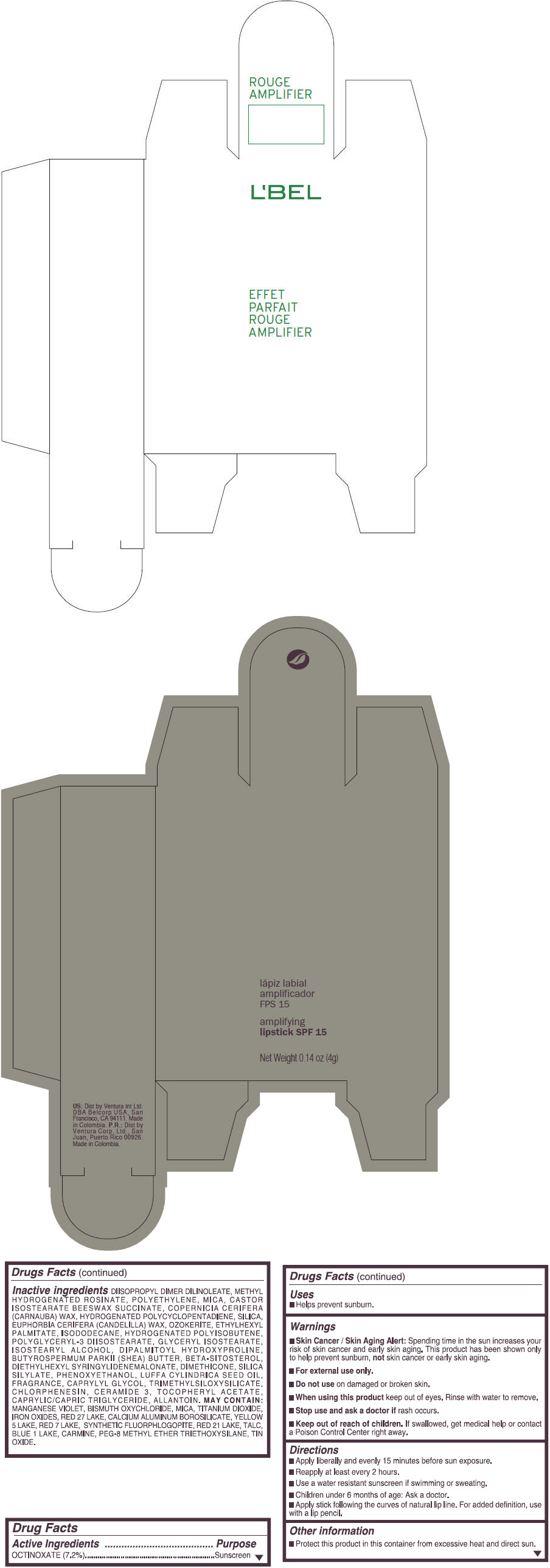
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROUGE RUBI) - RED
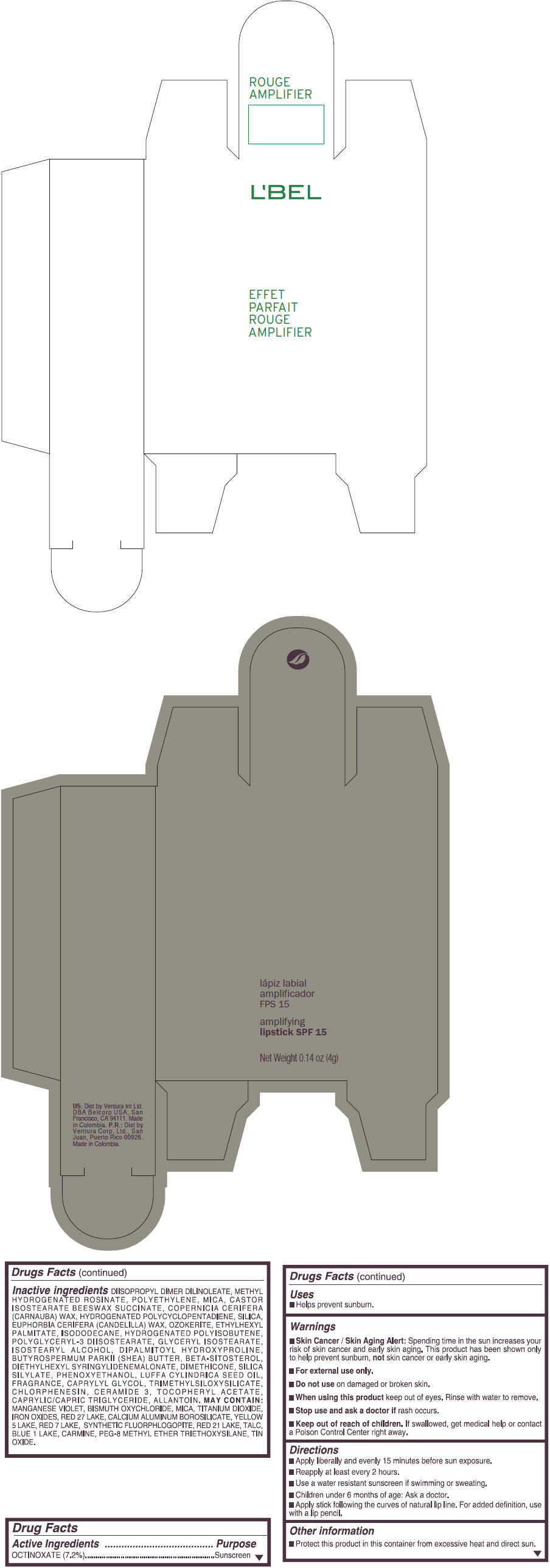
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (SOBRIETE) - BROWN
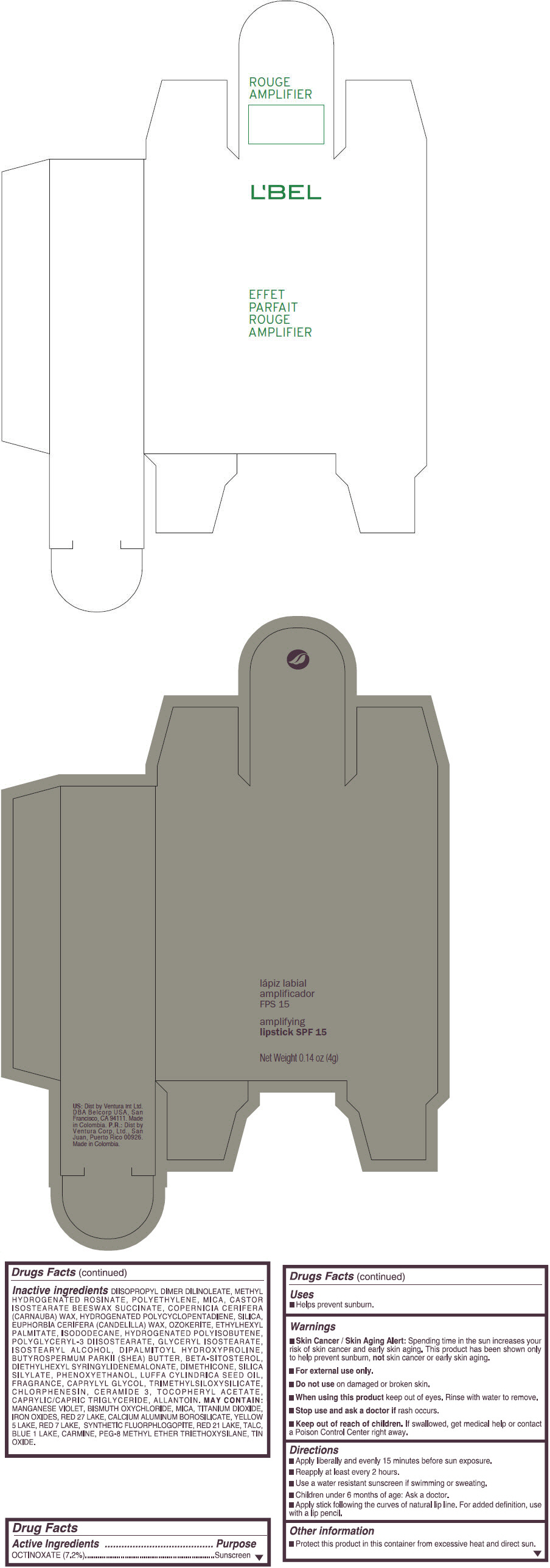
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (MARRON ILIMITEE) - BROWN
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (CORAL MAXIMUM) - ORANGE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROSE TENTATION) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROSE CHAMPAGNE) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROSE D'AMOUR) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROMANCE) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FUCSIA SPLENDIDE) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FUCSIA DESIR) - BROWN
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (ROUGE PROVOCATION) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (CARMINE) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (MALBEC) - BROWN
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (SALMON ESSENTIAL) - ORANGE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (BOURGOGNE VIF) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (GENUINE ROUGE) - RED
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (VIOLET VELVET) - PURPLE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (MYSTIC ROSE) - PINK
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (BELLE NUDE) - BEIGE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (FUCSIA PASSION) - PURPLE
- PRINCIPAL DISPLAY PANEL - 4 g Tube Box - (VIN CHIC) - BROWN
-
INGREDIENTS AND APPEARANCE
EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (ROUGE GRANDIOSE) - RED
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-870 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-870-02 1 in 1 BOX 1 NDC:13537-870-01 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (ROUGE RUBI) - RED
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-871 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-871-04 1 in 1 BOX 1 NDC:13537-871-03 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (SOBRIETE) - BROWN
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-872 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-872-06 1 in 1 BOX 1 NDC:13537-872-05 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (MARRON ILIMITEE) - BROWN
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-873 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-873-08 1 in 1 BOX 1 NDC:13537-873-07 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (CORAL MAXIMUM) - ORANGE
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-874 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-874-10 1 in 1 BOX 1 NDC:13537-874-09 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (ROSE TENTATION) - PINK
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-875 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-875-12 1 in 1 BOX 1 NDC:13537-875-11 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (ROSE CHAMPAGNE) - PINK
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-876 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-876-14 1 in 1 BOX 1 NDC:13537-876-13 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (ROSE DAMOUR) - PINK
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-877 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-877-16 1 in 1 BOX 1 NDC:13537-877-15 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (ROMANCE) - PINK
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-878 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-878-18 1 in 1 BOX 1 NDC:13537-878-17 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (FUCSIA SPLENDIDE) - PINK
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-879 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-879-20 1 in 1 BOX 1 NDC:13537-879-19 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (FUCSIA DESIR) - BROWN
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-881 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-881-22 1 in 1 BOX 1 NDC:13537-881-21 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (ROUGE PROVOCATION) - RED
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-882 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-882-24 1 in 1 BOX 1 NDC:13537-882-23 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (CARMINE) - RED
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-883 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-883-26 1 in 1 BOX 1 NDC:13537-883-25 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (MALBEC) - BROWN
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-884 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-884-28 1 in 1 BOX 1 NDC:13537-884-27 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (SALMON ESSENTIAL) - ORANGE
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-885 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-885-30 1 in 1 BOX 1 NDC:13537-885-29 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (BOURGOGNE VIF) - PINK
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-887 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-887-32 1 in 1 BOX 1 NDC:13537-887-31 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (GENUINE ROUGE) - RED
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-888 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-888-34 1 in 1 BOX 1 NDC:13537-888-33 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (VIOLET VELVET) - PURPLE
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-889 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-889-36 1 in 1 BOX 1 NDC:13537-889-35 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (MYSTIC ROSE) - PINK
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-890 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-890-38 1 in 1 BOX 1 NDC:13537-890-37 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (BELLE NUDE) - BEIGE
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-800 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-800-40 1 in 1 BOX 1 NDC:13537-800-39 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (FUCSIA PASSION) - PURPLE
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-801 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-801-42 1 in 1 BOX 1 NDC:13537-801-41 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 EFFET PARFAIT ROUGE AMPLIFIER AMPLIFYING SPF 15 (VIN CHIC) - BROWN
octinoxate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-802 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.072 g in 1 g Inactive Ingredients Ingredient Name Strength DIISOPROPYL DILINOLEATE (UNII: 5323S7S2LR) METHYL HYDROGENATED ROSINATE (UNII: 13DHA19W9N) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) MICA (UNII: V8A1AW0880) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CANDELILLA WAX (UNII: WL0328HX19) ETHYLHEXYL PALMITATE (UNII: 2865993309) ISODODECANE (UNII: A8289P68Y2) POLYGLYCERYL-3 DIISOSTEARATE (UNII: 46P231IQV8) GLYCERYL ISOSTEARATE (UNII: HYE7O27HAO) ISOSTEARYL ALCOHOL (UNII: Q613OCQ44Y) DIPALMITOYL HYDROXYPROLINE (UNII: E6AHA53N1H) SHEA BUTTER (UNII: K49155WL9Y) .BETA.-SITOSTEROL (UNII: S347WMO6M4) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) DIMETHICONE (UNII: 92RU3N3Y1O) PHENOXYETHANOL (UNII: HIE492ZZ3T) LUFFA AEGYPTIACA SEED OIL (UNII: 1A281RJ859) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHLORPHENESIN (UNII: I670DAL4SZ) CERAMIDE 3 (UNII: 4370DF050B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ALLANTOIN (UNII: 344S277G0Z) MANGANESE VIOLET (UNII: 72M48QQV8Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE RED (UNII: 1K09F3G675) D&C RED NO. 27 (UNII: 2LRS185U6K) ALUMINUM OXIDE (UNII: LMI26O6933) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) D&C RED NO. 7 (UNII: ECW0LZ41X8) D&C RED NO. 21 (UNII: 08744Z6JNY) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) STANNIC OXIDE (UNII: KM7N50LOS6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-802-44 1 in 1 BOX 1 NDC:13537-802-43 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 09/11/2014 Labeler - Ventura Corporation LTD (602751344) Establishment Name Address ID/FEI Business Operations Bel Star S.A. (Colombia) 880160197 MANUFACTURE(13537-870, 13537-871, 13537-872, 13537-873, 13537-874, 13537-875, 13537-876, 13537-877, 13537-878, 13537-879, 13537-881, 13537-882, 13537-883, 13537-884, 13537-885, 13537-887, 13537-888, 13537-889, 13537-890, 13537-800, 13537-801, 13537-802)