Label: KIDS PAIN AND FEVER- acetaminophen liquid
- NDC Code(s): 69676-0084-9
- Packager: Genexa Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 31, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if your child takes
- more than 5 doses in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
Allergy alert: acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: if sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
-
DOSAGE & ADMINISTRATION
Directions
- this product does not contain directions or complete warnings for adult use.
- do not give more than directed (see Overdose warning)
- SHAKE WELL BEFORE USING
- find right dose on chart below
- for accurate dosing, use measuring cup provided. Do not use with any other dosing device.
- give dose every 4 hours while symptoms last
- if possible, use weight to dose; otherwise, use age
- do not give more than 5 times in 24 hours
Weight Age Dose under 24 lbs. under 2 years ask a doctor 24-35 lbs. 2-3 years 5 mL 36-47 lbs. 4-5 years 7.5 mL 48-59 lbs. 6-8 years 10 mL 60-71 lbs. 9-10 years 12.5 mL 72-95 lbs. 11 years 15 mL - STORAGE AND HANDLING
- INACTIVE INGREDIENT
- QUESTIONS
-
SPL UNCLASSIFIED SECTION
Bottle is safety sealed with a perforated band around bottle. DO NOT use if bottle seal is disturbed or missing.
CERTIFIED B CORPORATION
CERTIFIED GLUTEN FREE GFCO.ORG
CERTIFIED VEGAN VEGAN.ORG
Patent Pending | Distributed by: Genexa Inc.
Atlanta, GA 30324 | genexa.com
* This product is not manufactured or distributed by Johnson & Johnson Corp., owner of the registered trademark Children's Tylenol® Oral Suspension Cherry.
- SPL UNCLASSIFIED SECTION
-
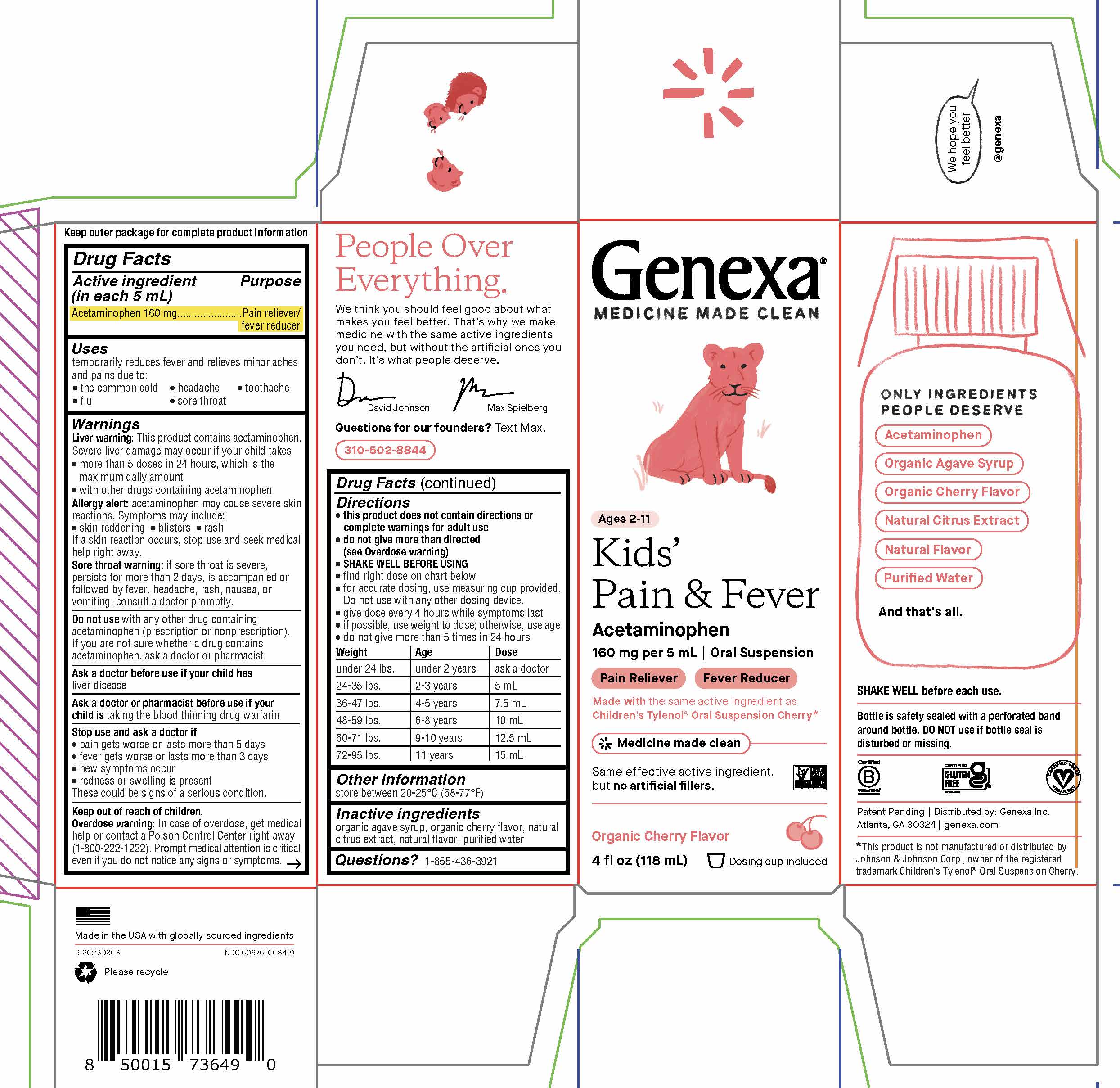
PRINCIPAL DISPLAY PANEL
Genexa®
MEDICINE MADE CLEAN
Ages 2-11
Kids' Pain & Fever
Acetaminophen
160 mg per 5 mL | Oral Suspension
Pain Reliever Fever ReducerMade with the same active ingredient as
Children’s Tylenol® Oral Suspension Cherry*
Medicene made clean
Same effective active ingredient, but no artificial fillers.
Organic Cherry Flavor
4 FL OZ (118 mL) Liquid Dosing cup included

-
INGREDIENTS AND APPEARANCE
KIDS PAIN AND FEVER
acetaminophen liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69676-0084 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 160 mg in 5 mL Inactive Ingredients Ingredient Name Strength CHERRY (UNII: BUC5I9595W) AGAVE TEQUILANA JUICE (UNII: GVG8G0207O) WATER (UNII: 059QF0KO0R) CITRUS FRUIT (UNII: XDK00Z8012) Product Characteristics Color brown Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69676-0084-9 1 in 1 CARTON 05/30/2023 1 118 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part343 05/30/2023 Labeler - Genexa Inc. (079751024)

