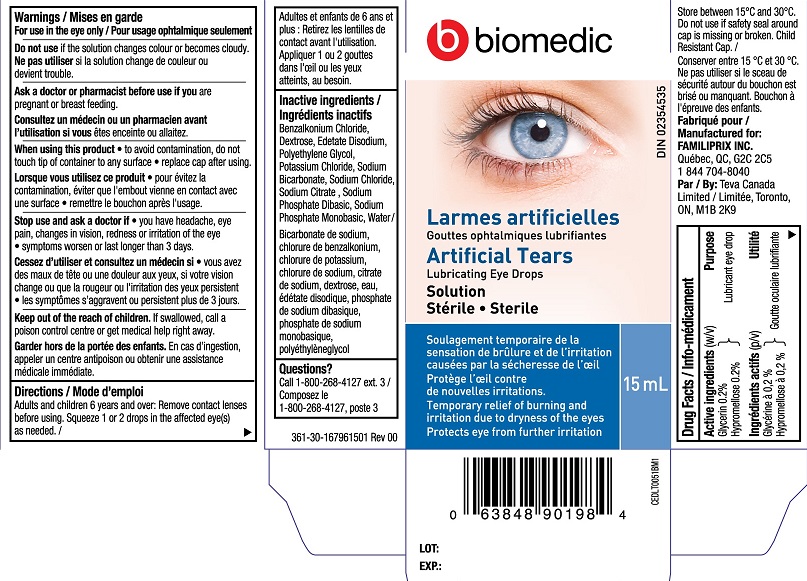
Label: BIOMEDIC ARTIFICIAL TEARS- glycerin, hypromellose liquid
- NDC Code(s): 55651-031-01
- Packager: KC Pharmaceuticals, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Export only
Drug Label Information
Updated March 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BIOMEDIC ARTIFICIAL TEARS
glycerin, hypromellose liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55651-031 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 0.2 g in 100 mL HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) (HYPROMELLOSE, UNSPECIFIED - UNII:3NXW29V3WO) HYPROMELLOSE, UNSPECIFIED 0.2 g in 100 mL Inactive Ingredients Ingredient Name Strength POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) DEXTROSE (UNII: IY9XDZ35W2) EDETATE DISODIUM (UNII: 7FLD91C86K) POTASSIUM CHLORIDE (UNII: 660YQ98I10) WATER (UNII: 059QF0KO0R) SODIUM BICARBONATE (UNII: 8MDF5V39QO) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CITRATE (UNII: 1Q73Q2JULR) SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) SODIUM PHOSPHATE, MONOBASIC (UNII: 3980JIH2SW) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55651-031-01 1 in 1 CARTON 04/30/2019 1 15 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Export only 04/30/2019 Labeler - KC Pharmaceuticals, Inc. (174450460) Establishment Name Address ID/FEI Business Operations KC Pharmaceuticals, Inc. 174450460 manufacture(55651-031) , pack(55651-031) , label(55651-031)