Label: BEAUELI- lidocaine cream
- NDC Code(s): 83462-001-01
- Packager: EUbizrival LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 18, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
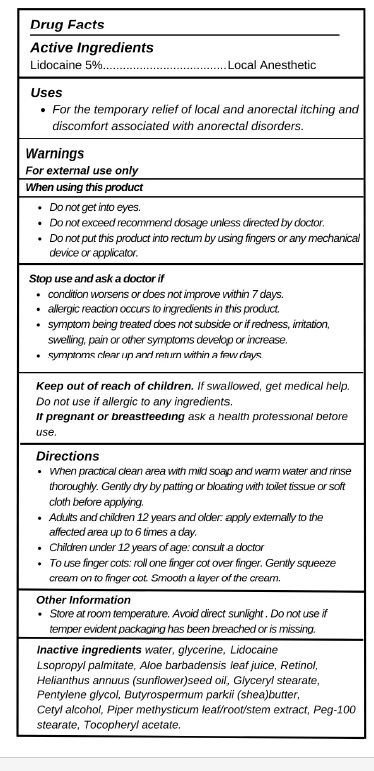
- Active ingredients
- Purpose
- Uses
-
Warnings
For external use only
When using this product
- Do not get into eyes.
- Do not exceed recommend dosage unless directed by doctor.
- Do not put this product into rectum by using fingers or any mechanical device or applicator.
Stop use and ask a doctor if
- condition worsens or does not improve within 7 days.
- allergic reaction occurs to ingredients in this product.
- symptom being treated does not subside or if redness, irritation, swelling, pain or other symptoms develop or increase.
- symptoms clear up and return within a few days.
Keep out of reach of children.If swallowed, get medical help. Do not use if allergic to any ingredients.
If pregnant or breastfeedingask a health professional before use.
- KEEP OUT OF REACH OF CHILDREN
-
Directions
- When practical clean area with mild soap and warm water and rinse thoroughly. Gently dry by patting or bloating with toilet tissue or soft cloth before applying.
- Adults and children 12 years and older: apply externally to the affected area up to 6 times a day.
- Children under 12 years of age: consult a doctor
- To use finger cots: roll one finger cot over finger. Gently squeeze cream on to finger cot. Smooth a layer of the cream.
- Other information
- INACTIVE INGREDIENT
- Product label
-
INGREDIENTS AND APPEARANCE
BEAUELI
lidocaine creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83462-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 5 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) ALOE (UNII: V5VD430YW9) SHEA BUTTER (UNII: K49155WL9Y) RETINOL (UNII: G2SH0XKK91) SUNFLOWER OIL (UNII: 3W1JG795YI) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) PENTYLENE GLYCOL (UNII: 50C1307PZG) CETYL ALCOHOL (UNII: 936JST6JCN) PIPER METHYSTICUM WHOLE (UNII: 3P306S300W) PEG-100 STEARATE (UNII: YD01N1999R) .ALPHA.-TOCOPHEROL ACETATE, D- (UNII: A7E6112E4N) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83462-001-01 50 g in 1 BOTTLE; Type 0: Not a Combination Product 07/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 07/01/2023 Labeler - EUbizrival LLC (036572203) Establishment Name Address ID/FEI Business Operations Guangzhou Pallas Cosmetics Co.,Ltd. 419564106 manufacture(83462-001)