Label: DILTIAZEM HYDROCHLORIDE tablet
DILTIAZEM HYDROCHLORIDE tablet
-
NDC Code(s):
68682-006-10,
68682-006-50,
68682-007-10,
68682-007-50, view more68682-008-10, 68682-008-50, 68682-009-10
- Packager: Oceanside Pharmaceuticals
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application Authorized Generic
Drug Label Information
Updated June 2, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
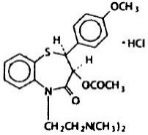
Diltiazem hydrochloride is a calcium ion cellular influx inhibitor (slow channel blocker or calcium antagonist). Chemically, diltiazem hydrochloride is 1,5-Benzothiazepin-4(5H)-one,3-(acetyloxy)-5-[2-(dimethylamino)ethyl]-2, 3-dihydro-2-(4-methoxyphenyl)-, monohydrochloride,(+)-cis-. The chemical structure is:
Diltiazem hydrochloride is a white to off-white crystalline powder with a bitter taste. It is soluble in water, methanol, and chloroform. It has a molecular weight of 450.98. Each tablet contains 30 mg, 60 mg, 90 mg, or 120 mg diltiazem hydrochloride.
Also contains: colloidal silicon dioxide, D&C Yellow #10 Aluminum Lake, FD&C Blue #1 Aluminum Lake (30 mg and 90 mg), FD&C Yellow #6 Aluminum Lake (60 mg and 120 mg), hydroxypropyl cellulose, hypromellose, lactose, magnesium stearate, methylparaben, microcrystalline cellulose, and polyethylene glycol.
For oral administration.
-
CLINICAL PHARMACOLOGY
The therapeutic benefits achieved with diltiazem hydrochloride are believed to be related to its ability to inhibit the influx of calcium ions during membrane depolarization of cardiac and vascular smooth muscle.
Mechanisms of Action
Although precise mechanisms of its antianginal actions are still being delineated, diltiazem hydrochloride is believed to act in the following ways:
- •
- Angina Due to Coronary Artery Spasm: Diltiazem hydrochloride has been shown to be a potent dilator of coronary arteries both epicardial and subendocardial. Spontaneous and ergonovine-induced coronary artery spasms are inhibited by diltiazem hydrochloride.
- •
- Exertional Angina: Diltiazem hydrochloride has been shown to produce increases in exercise tolerance, probably due to its ability to reduce myocardial oxygen demand. This is accomplished via reductions in heart rate and systemic blood pressure at submaximal and maximal exercise workloads.
In animal models, diltiazem interferes with the slow inward (depolarizing) current in excitable tissue. It causes excitation-contraction uncoupling in various myocardial tissues without changes in the configuration of the action potential. Diltiazem produces relaxation of coronary vascular smooth muscle and dilation of both large and small coronary arteries at drug levels which cause little or no negative inotropic effect. The resultant increases in coronary blood flow (epicardial and subendocardial) occur in ischemic and nonischemic models and are accompanied by dose-dependent decreases in systemic blood pressure and decreases in peripheral resistance.
Hemodynamic and Electrophysiologic Effects
Like other calcium antagonists, diltiazem decreases sinoatrial and atrioventricular conduction in isolated tissues and has a negative inotropic effect in isolated preparations. In the intact animal, prolongation of the AH interval can be seen at higher doses.
In man, diltiazem prevents spontaneous and ergonovine-provoked coronary artery spasm. It causes a decrease in peripheral vascular resistance and a modest fall in blood pressure, and in exercise tolerance studies in patients with ischemic heart disease, reduces the heart rate-blood pressure product for any given workload. Studies to date, primarily in patients with good ventricular function, have not revealed evidence of a negative inotropic effect; cardiac output, ejection fraction, and left ventricular end-diastolic pressure have not been affected. There are as yet few data on the interaction of diltiazem and beta-blockers. Resting heart rate is usually unchanged or slightly reduced by diltiazem.
Intravenous diltiazem in doses of 20 mg prolongs AH conduction time and AV node functional and effective refractory periods approximately 20%. In a study involving single oral doses of 300 mg of diltiazem hydrochloride in six normal volunteers, the average maximum PR prolongation was 14% with no instances of greater than first-degree AV block. Diltiazem-associated prolongation of the AH interval is not more pronounced in patients with first-degree heart block. In patients with sick sinus syndrome, diltiazem significantly prolongs sinus cycle length (up to 50% in some cases).
Chronic oral administration of diltiazem hydrochloride in doses of up to 240 mg/day has resulted in small increases in PR interval but has not usually produced abnormal prolongation.
Pharmacokinetics and Metabolism
Diltiazem is well absorbed from the gastrointestinal tract and is subject to an extensive first-pass effect, giving an absolute bioavailability (compared to intravenous dosing) of about 40%. Diltiazem hydrochloride undergoes extensive metabolism in which 2% to 4% of the unchanged drug appears in the urine. In vitrobinding studies show diltiazem hydrochloride is 70% to 80% bound to plasma proteins. Competitive in vitroligand binding studies have also shown diltiazem hydrochloride binding is not altered by therapeutic concentrations of digoxin, hydrochlorothiazide, phenylbutazone, propranolol, salicylic acid, or warfarin. The plasma elimination half-life following single or multiple drug administration is approximately 3.0 to 4.5 hours. Desacetyl diltiazem is also present in the plasma at levels of 10% to 20% of the parent drug and is 25% to 50% as potent as a coronary vasodilator as diltiazem. Minimum therapeutic plasma levels of diltiazem hydrochloride appear to be in the range of 50 to 200 ng/mL. There is a departure from linearity when dose strengths are increased. A study that compared patients with normal hepatic function to patients with cirrhosis found an increase in half-life and a 69% increase in AUC (area-under-the-plasma concentration vs. time curve) in the hepatically impaired patients. A single study in nine patients with severely impaired renal functions showed no difference in the pharmacokinetic profile of diltiazem as compared to patients with normal renal function.
Diltiazem Hydrochloride Tablets: Diltiazem is absorbed from the tablet formulation to about 98% of a reference solution. Single oral doses of 30 to 120 mg of diltiazem hydrochloride tablets result in detectable plasma levels within 30 to 60 minutes and peak plasma levels 2 to 4 hours after drug administration. As the dose of diltiazem hydrochloride tablets is increased from a daily dose of 120 mg (30 mg qid) to 240 mg (60 mg qid) daily, there is an increase in area-under-the-curve of 2.3 times. When the dose is increased from 240 mg to 360 mg daily, there is an increase in area-under-the-curve of 1.8 times.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
Diltiazem hydrochloride tablets are contraindicated in:
- •
- Patients with sick sinus syndrome except in the presence of a functioning ventricular pacemaker
- •
- Patients with second- or third-degree AV block except in the presence of a functioning ventricular pacemaker
- •
- Patients with hypotension (less than 90 mm Hg systolic)
- •
- Patients who have demonstrated hypersensitivity to the drug
- •
- Patients with acute myocardial infarction and pulmonary congestion documented by x-ray on admission
-
WARNINGS
1. Cardiac Conduction: Diltiazem hydrochloride prolongs AV node refractory periods without significantly prolonging sinus node recovery time, except in patients with sick sinus syndrome. This effect may rarely result in abnormally slow heart rates (particularly in patients with sick sinus syndrome) or second- or third-degree AV block (six of 1243 patients for 0.48%). Concomitant use of diltiazem with beta-blockers or digitalis may result in additive effects on cardiac conduction. A patient with Prinzmetal’s angina developed periods of asystole (2 to 5 seconds) after a single dose of 60 mg of diltiazem (see ADVERSE REACTIONS).
2. Congestive Heart Failure: Although diltiazem has a negative inotropic effect in isolated animal tissue preparations, hemodynamic studies in humans with normal ventricular function have not shown a reduction in cardiac index nor consistent negative effects on contractility (dP/dt). Experience with the use of diltiazem hydrochloride alone or in combination with beta-blockers in patients with impaired ventricular function is very limited. Caution should be exercised when using the drug in such patients.
3. Hypotension: Decreases in blood pressure associated with diltiazem hydrochloride therapy may occasionally result in symptomatic hypotension.
4. Acute Hepatic Injury: In rare instances, significant elevations in enzymes such as alkaline phosphatase, LDH, SGOT, SGPT, and other phenomena consistent with acute hepatic injury have been noted. These reactions have been reversible upon discontinuation of drug therapy. The relationship to diltiazem hydrochloride is uncertain in most cases, but probable in some (see PRECAUTIONS).
-
PRECAUTIONS
General
Diltiazem hydrochloride is extensively metabolized by the liver and excreted by the kidneys and in bile. As with any drug given over prolonged periods, laboratory parameters of renal and hepatic function should be monitored at regular intervals. The drug should be used with caution in patients with impaired renal or hepatic function. In subacute and chronic dog and rat studies designed to produce toxicity, high doses of diltiazem were associated with hepatic damage. In special subacute hepatic studies, oral doses of 125 mg/kg and higher in rats were associated with histological changes in the liver, which were reversible when the drug was discontinued. In dogs, doses of 20 mg/kg were also associated with hepatic changes; however, these changes were reversible with continued dosing.
Dermatological events (see ADVERSE REACTIONS) may be transient and may disappear despite continued use of diltiazem hydrochloride. However, skin eruptions progressing to erythema multiforme and/or exfoliative dermatitis have also been infrequently reported. Should a dermatologic reaction persist, the drug should be discontinued.
Drug Interactions
Due to the potential for additive effects, caution and careful titration are warranted in patients receiving diltiazem hydrochloride concomitantly with any agents known to affect cardiac contractility and/or conduction (see WARNINGS). Pharmacologic studies indicate that there may be additive effects in prolonging AV conduction when using beta-blockers or digitalis concomitantly with diltiazem hydrochloride (see WARNINGS).
As with all drugs, care should be exercised when treating patients with multiple medications. Diltiazem is both a substrate and an inhibitor of the cytochrome P450 3A4 enzyme system. Other drugs that are specific substrates, inhibitors, or inducers of this enzyme system may have a significant impact on the efficacy and side effect profile of diltiazem. Patients taking other drugs that are substrates of CYP450 3A4, especially patients with renal and/or hepatic impairment, may require dosage adjustment when starting or stopping concomitantly administered diltiazem in order to maintain optimum therapeutic blood levels.
Anesthetics: The depression of cardiac contractility, conductivity, and automaticity, as well as the vascular dilation associated with anesthetics, may be potentiated by calcium channel blockers. When used concomitantly, anesthetics and calcium blockers should be titrated carefully.
Benzodiazepines: Studies showed that diltiazem increased the AUC of midazolam and triazolam by 3- to 4-fold and the Cmax by 2-fold, compared to placebo. The elimination half-life of midazolam and triazolam also increased (1.5- to 2.5-fold) during coadministration with diltiazem. These pharmacokinetic effects seen during diltiazem coadministration can result in increased clinical effects (e.g., prolonged sedation) of both midazolam and triazolam.
Beta-blockers: Controlled and uncontrolled domestic studies suggest that concomitant use of diltiazem hydrochloride and beta-blockers is usually well tolerated. Available data are not sufficient, however, to predict the effects of concomitant treatment, particularly in patients with left ventricular dysfunction or cardiac conduction abnormalities.
Administration of diltiazem hydrochloride concomitantly with propranolol in five normal volunteers resulted in increased propranolol levels in all subjects, and bioavailability of propranolol was increased approximately 50%. In vitro, propranolol appears to be displaced from its binding sites by diltiazem. If combination therapy is initiated or withdrawn in conjunction with propranolol, an adjustment in the propranolol dose may be warranted (see WARNINGS).
Buspirone: In nine healthy subjects, diltiazem significantly increased the mean buspirone AUC 5.5-fold and Cmax 4.1-fold compared to placebo. The T1/2 and Tmax of buspirone were not significantly affected by diltiazem. Enhanced effects and increased toxicity of buspirone may be possible during concomitant administration with diltiazem. Subsequent dose adjustments may be necessary during coadministration, and should be based on clinical assessment.
Carbamazepine: Concomitant administration of diltiazem with carbamazepine has been reported to result in elevated serum levels of carbamazepine (40% to 72% increase) resulting in toxicity in some cases. Patients receiving these drugs concurrently should be monitored for a potential drug interaction.
Cimetidine: A study in six healthy volunteers has shown a significant increase in peak diltiazem plasma levels (58%) and area-under-the-curve (53%) after a 1-week course of cimetidine at 1200 mg per day and a single dose of diltiazem 60 mg. Ranitidine produced smaller, nonsignificant increases. The effect may be mediated by cimetidine’s known inhibition of hepatic cytochrome P450, the enzyme system responsible for the first-pass metabolism of diltiazem. Patients currently receiving diltiazem therapy should be carefully monitored for a change in pharmacological effect when initiating and discontinuing therapy with cimetidine. An adjustment in the diltiazem dose may be warranted.
Clonidine: Sinus bradycardia resulting in hospitalization and pacemaker insertion has been reported in association with the use of clonidine concurrently with diltiazem. Monitor heart rate in patients receiving concomitant diltiazem and clonidine.
Cyclosporine: A pharmacokinetic interaction between diltiazem and cyclosporine has been observed during studies involving renal and cardiac transplant patients. In renal and cardiac transplant recipients, a reduction of cyclosporine trough dose ranging from 15% to 48% was necessary to maintain concentrations similar to those seen prior to the addition of diltiazem. If these agents are to be administered concurrently, cyclosporine concentrations should be monitored, especially when diltiazem therapy is initiated, adjusted, or discontinued. The effect of cyclosporine on diltiazem plasma concentrations has not been evaluated.
Digitalis: Administration of diltiazem hydrochloride with digoxin in 24 healthy male subjects increased plasma digoxin concentrations approximately 20%. Another investigator found no increase in digoxin levels in 12 patients with coronary artery disease. Since there have been conflicting results regarding the effect of digoxin levels, it is recommended that digoxin levels be monitored when initiating, adjusting, and discontinuing diltiazem hydrochloride therapy to avoid possible over- or under-digitalization (see WARNINGS).
Ivabradine: Concurrent use of diltiazem increases exposure to ivabradine and may exacerbate bradycardia and conduction disturbances. Avoid concomitant use of ivabradine and diltiazem.
Quinidine: Diltiazem significantly increases the AUC(0 → ∞) of quinidine by 51%, T1/2 by 36%, and decreases its CLoral by 33%. Monitoring for quinidine adverse effects may be warranted and the dose adjusted accordingly.
Rifampin: Coadministration of rifampin with diltiazem lowered the diltiazem plasma concentrations to undetectable levels. Coadministration of diltiazem with rifampin or any known CYP3A4 inducer should be avoided when possible, and alternative therapy considered.
Statins: Diltiazem is an inhibitor of CYP3A4 and has been shown to increase significantly the AUC of some statins. The risk of myopathy and rhabdomyolysis with statins metabolized by CYP3A4 may be increased with concomitant use of diltiazem. When possible, use a non-CYP3A4-metabolized statin together with diltiazem; otherwise, dose adjustments for both diltiazem and the statin should be considered along with close monitoring for signs and symptoms of any statin-related adverse events.
In a healthy volunteer crossover study (N=10), coadministration of a single 20 mg dose of simvastatin at the end of a 14-day regimen with 120 mg BID diltiazem SR resulted in a 5-fold increase in mean simvastatin AUC versus simvastatin alone. Subjects with increased average steady-state exposures of diltiazem showed a greater fold increase in simvastatin exposure. Computer-based simulations showed that at a daily dose of 480 mg of diltiazem, an 8- to 9-fold mean increase in simvastatin AUC can be expected. If coadministration of simvastatin with diltiazem is required, limit the daily doses of simvastatin to 10 mg and diltiazem to 240 mg.
In a ten-subject randomized, open label, 4-way crossover study, coadministration of diltiazem (120 mg BID diltiazem SR for 2 weeks) with a single 20 mg dose of lovastatin resulted in 3- to 4-fold increase in mean lovastatin AUC and Cmax versus lovastatin alone. In the same study, there was no significant change in 20 mg single dose pravastatin AUC and Cmax during diltiazem coadministration. Diltiazem plasma levels were not significantly affected by lovastatin or pravastatin.
Carcinogenesis, Mutagenesis, Impairment of Fertility
A 24-month study in rats and a 21-month study in mice showed no evidence of carcinogenicity. There was also no mutagenic response in in vitrobacterial tests. No intrinsic effect on fertility was observed in rats.
Pregnancy
Reproduction studies have been conducted in mice, rats, and rabbits. Administration of doses ranging from five to ten times greater (on a mg/kg basis) than the daily recommended therapeutic dose has resulted in embryo and fetal lethality. These doses, in some studies, have been reported to cause skeletal abnormalities. In the perinatal/postnatal studies, there was some reduction in early individual pup weights and survival rates. There was an increased incidence of stillbirths at doses of 20 times the human dose or greater.
There are no well-controlled studies in pregnant women; therefore, use diltiazem hydrochloride in pregnant women only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
Diltiazem is excreted in human milk. One report suggests that concentrations in breast milk may approximate serum levels. If use of diltiazem hydrochloride is deemed essential, an alternative method of infant feeding should be instituted.
Geriatric Use
Clinical studies of diltiazem did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Information for Patients
30 mg – Diltiazem Hydrochloride tablets may be swallowed whole, crushed, or chewed. Do not split Diltiazem Hydrochloride tablets.
60 mg, 90 mg, and 120 mg – Diltiazem Hydrochloride tablets may be swallowed whole, crushed, or chewed.
-
ADVERSE REACTIONS
Serious adverse reactions have been rare in studies carried out to date, but it should be recognized that patients with impaired ventricular function and cardiac conduction abnormalities usually have been excluded.
In domestic placebo-controlled angina trials, the incidence of adverse reactions reported during diltiazem hydrochloride therapy was not greater than that reported during placebo therapy.
The following represent occurrences observed in clinical studies of angina patients. In many cases, the relationship to diltiazem hydrochloride has not been established. The most common occurrences from these studies, as well as their frequency of presentation, are edema (2.4%), headache (2.1%), nausea (1.9%), dizziness (1.5%), rash (1.3%), and asthenia (1.2%). In addition, the following events were reported infrequently (less than 1%):
Cardiovascular: Angina, arrhythmia, AV block (first-degree), AV block (second- or third-degree – see WARNINGS, Cardiac Conduction), bradycardia, bundle branch block, congestive heart failure, ECG abnormality, flushing, hypotension, palpitations, syncope, tachycardia, ventricular extrasystoles
Nervous System: Abnormal dreams, amnesia, depression, gait abnormality, hallucinations, insomnia, nervousness, paresthesia, personality change, somnolence, tremor
Gastrointestinal: Anorexia, constipation, diarrhea, dysgeusia, dyspepsia, mild elevations of alkaline phosphatase, SGOT, SGPT, and LDH (see WARNINGS, Acute Hepatic Injury), thirst, vomiting, weight increase.
Dermatological: Petechiae, photosensitivity, pruritus, urticaria
Other: Amblyopia, CPK elevation, dry mouth, dyspnea, epistaxis, eye irritation, hyperglycemia, hyperuricemia, impotence, muscle cramps, nasal congestion, nocturia, osteoarticular pain, polyuria, sexual difficulties, tinnitus
The following postmarketing events have been reported infrequently in patients receiving diltiazem hydrochloride tablets: acute generalized exanthematous pustulosis, allergic reactions, alopecia, angioedema (including facial or periorbital edema), asystole, erythema multiforme (including Stevens-Johnson syndrome, toxic epidermal necrolysis), extrapyramidal symptoms, gingival hyperplasia, hemolytic anemia, increased bleeding time, leukopenia, photosensitivity (including lichenoid keratosis and hyperpigmentation at sun-exposed skin areas), purpura, retinopathy, myopathy, and thrombocytopenia. There have been observed cases of a generalized rash, some characterized as leukocytoclastic vasculitis. In addition, events such as myocardial infarction have been observed, which are not readily distinguishable from the natural history of the disease in these patients. A definitive cause and effect relationship between these events and diltiazem hydrochloride therapy cannot yet be established. Exfoliative dermatitis (proven by rechallenge) has also been reported.
To report SUSPECTED ADVERSE REACTIONS, contact Oceanside Pharmaceuticals at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
OVERDOSAGE
The oral LD50s in mice and rats range from 415 to 740 mg/kg and from 560 to 810 mg/kg, respectively. The intravenous LD50s in these species were 60 and 38 mg/kg, respectively. The oral LD50 in dogs is considered to be in excess of 50 mg/kg, while lethality was seen in monkeys at 360 mg/kg.
The toxic dose in man is not known. Due to extensive metabolism, blood levels after a standard dose of diltiazem can vary over tenfold, limiting the usefulness of blood levels in overdose cases.
There have been reports of diltiazem overdose in amounts ranging from <1 g to 18 g. Of cases with known outcome, most patients recovered and in cases with a fatal outcome, the majority involved multiple drug ingestion.
Events observed following diltiazem overdose included bradycardia, hypotension, heart block, and cardiac failure. Most reports of overdose described some supportive medical measure and/or drug treatment. Bradycardia frequently responded favorably to atropine, as did heart block, although cardiac pacing was also frequently utilized to treat heart block. Fluids and vasopressors were used to maintain blood pressure, and in cases of cardiac failure, inotropic agents were administered. In addition, some patients received treatment with ventilatory support, gastric lavage, activated charcoal, and/or intravenous calcium.
The effectiveness of intravenous calcium administration to reverse the pharmacological effects of diltiazem overdose has been inconsistent. In a few reported cases, overdose with calcium channel blockers associated with hypotension and bradycardia that was initially refractory to atropine became more responsive to atropine after the patients received intravenous calcium. In some cases intravenous calcium has been administered (1 g calcium chloride or 3 g calcium gluconate) over 5 minutes and repeated every 10 to 20 minutes as necessary. Calcium gluconate has also been administered as a continuous infusion at a rate of 2 g per hour for 10 hours. Infusions of calcium for 24 hours or more may be required. Patients should be monitored for signs of hypercalcemia.
In the event of overdose or exaggerated response, appropriate supportive measures should be employed in addition to gastrointestinal decontamination. Diltiazem does not appear to be removed by peritoneal or hemodialysis. Limited data suggest that plasmapheresis or charcoal hemoperfusion may hasten diltiazem elimination following overdose. Based on the known pharmacological effects of diltiazem and/or reported clinical experiences, the following measures may be considered:
Bradycardia: Administer atropine (0.60 to 1.0 mg). If there is no response to vagal blockade, administer isoproterenol cautiously.
High-Degree AV Block: Treat as for bradycardia above. Fixed high-degree AV block should be treated with cardiac pacing.
Cardiac Failure: Administer inotropic agents (isoproterenol, dopamine, or dobutamine) and diuretics.
Hypotension: Vasopressors (e.g., dopamine or norepinephrine).
Actual treatment and dosage should depend on the severity of the clinical situation and the judgment and experience of the treating physician.
-
DOSAGE AND ADMINISTRATION
Exertional Angina Pectoris Due to Atherosclerotic Coronary Artery Disease or Angina Pectoris at Rest Due to Coronary Artery Spasm: Dosage must be adjusted to each patient’s needs. Starting with 30 mg four times daily, before meals and at bedtime, dosage should be increased gradually (given in divided doses three or four times daily) at 1- to 2-day intervals until optimum response is obtained. Although individual patients may respond to any dosage level, the average optimum dosage range appears to be 180 to 360 mg/day. There are no available data concerning dosage requirements in patients with impaired renal or hepatic function. If the drug must be used in such patients, titration should be carried out with particular caution.
Concomitant Use with Other Cardiovascular Agents
- •
- Sublingual NTG may be taken as required to abort acute anginal attacks during diltiazem hydrochloride therapy.
- •
- Prophylactic Nitrate Therapy. Diltiazem hydrochloride may be safely coadministered with short- and long-acting nitrates, but there have been no controlled studies to evaluate the antianginal effectiveness of this combination.
- •
- Beta-blockers. (See WARNINGS and PRECAUTIONS.)
- 30 mg – Diltiazem Hydrochloride tablets may be swallowed whole, crushed, or chewed. Do not split Diltiazem Hydrochloride tablets.
- 60 mg, 90 mg, and 120 mg – Diltiazem Hydrochloride tablets may be swallowed whole, crushed, or chewed.
-
HOW SUPPLIED
Diltiazem hydrochloride 30 mg tablets are supplied in bottles of 100 (NDC 68682-006-10) and 500 (NDC 68682-006-50). Each light green, round tablet is engraved with MARION on one side and 1771 on the other.
Diltiazem hydrochloride 60 mg scored tablets are supplied in bottles of 100 (NDC 68682-007-10) and 500 (NDC 68682-007-50). Each light yellow, round tablet is engraved with MARION on one side and 1772 on the other.
Diltiazem hydrochloride 90 mg scored tablets are supplied in bottles of 100 (NDC 68682-008-10) and 500 (NDC 68682-008-50). Each light green, capsule-shaped tablet is engraved with CARDIZEM on one side and 90 mg on the other.
Diltiazem hydrochloride 120 mg scored tablets are supplied in bottles of 100 (NDC 68682-009-10). Each yellow, capsule-shaped tablet is engraved with CARDIZEM on one side and 120 mg on the other.
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Avoid excessive humidity.
Distributed by:
Oceanside Pharmaceuticals, a division of
Bausch Health US, LLC
Bridgewater, NJ 08807 USAManufactured by:
Bausch Health Companies Inc.
Laval, Quebec H7L 4A8, CanadaCARDIZEM is a trademark of Bausch Health Companies Inc. or its affiliates.
© 2020 Bausch Health Companies Inc. or its affiliates
Rev. 06/2020
9467803
- PRINCIPAL DISPLAY PANEL - 30 mg Capsules
- PRINCIPAL DISPLAY PANEL - 60 mg Capsules
- PRINCIPAL DISPLAY PANEL - 90 mg Capsules
- PRINCIPAL DISPLAY PANEL - 120 mg Capsules
-
INGREDIENTS AND APPEARANCE
DILTIAZEM HYDROCHLORIDE
diltiazem hydrochloride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68682-006 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Diltiazem hydrochloride (UNII: OLH94387TE) (DILTIAZEM - UNII:EE92BBP03H) Diltiazem hydrochloride 30 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C Yellow no. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) LACTOSE, UNSPECIFIED FORM (UNII: J2B2A4N98G) Magnesium Stearate (UNII: 70097M6I30) METHYLPARABEN (UNII: A2I8C7HI9T) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) Product Characteristics Color GREEN (light) Score no score Shape ROUND Size 8mm Flavor Imprint Code MARION;1771 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68682-006-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 12/25/2010 2 NDC:68682-006-50 500 in 1 BOTTLE; Type 0: Not a Combination Product 12/25/2010 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA authorized generic NDA018602 12/25/2010 DILTIAZEM HYDROCHLORIDE
diltiazem hydrochloride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68682-007 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Diltiazem Hydrochloride (UNII: OLH94387TE) (DILTIAZEM - UNII:EE92BBP03H) Diltiazem Hydrochloride 60 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C Yellow no. 10 (UNII: 35SW5USQ3G) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) LACTOSE, UNSPECIFIED FORM (UNII: J2B2A4N98G) Magnesium Stearate (UNII: 70097M6I30) METHYLPARABEN (UNII: A2I8C7HI9T) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) Product Characteristics Color YELLOW (light) Score 2 pieces Shape ROUND Size 10mm Flavor Imprint Code MARION;1772 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68682-007-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 12/25/2010 2 NDC:68682-007-50 500 in 1 BOTTLE; Type 0: Not a Combination Product 12/25/2010 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA authorized generic NDA018602 12/25/2010 DILTIAZEM HYDROCHLORIDE
diltiazem hydrochloride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68682-008 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Diltiazem Hydrochloride (UNII: OLH94387TE) (DILTIAZEM - UNII:EE92BBP03H) Diltiazem Hydrochloride 90 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C Yellow no. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) LACTOSE, UNSPECIFIED FORM (UNII: J2B2A4N98G) Magnesium Stearate (UNII: 70097M6I30) METHYLPARABEN (UNII: A2I8C7HI9T) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) Product Characteristics Color GREEN (light) Score 2 pieces Shape BULLET (oblong) Size 17mm Flavor Imprint Code CARDIZEM;90mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68682-008-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 12/25/2010 2 NDC:68682-008-50 500 in 1 BOTTLE; Type 0: Not a Combination Product 12/25/2010 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA authorized generic NDA018602 12/25/2010 DILTIAZEM HYDROCHLORIDE
diltiazem hydrochloride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68682-009 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Diltiazem Hydrochloride (UNII: OLH94387TE) (DILTIAZEM - UNII:EE92BBP03H) Diltiazem Hydrochloride 120 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C Yellow no. 10 (UNII: 35SW5USQ3G) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) LACTOSE, UNSPECIFIED FORM (UNII: J2B2A4N98G) Magnesium Stearate (UNII: 70097M6I30) METHYLPARABEN (UNII: A2I8C7HI9T) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) Product Characteristics Color YELLOW Score 2 pieces Shape CAPSULE (oblong) Size 19mm Flavor Imprint Code CARDIZEM;120mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68682-009-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 12/25/2010 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA authorized generic NDA018602 12/25/2010 Labeler - Oceanside Pharmaceuticals (832011691) Establishment Name Address ID/FEI Business Operations Bausch Health Companies Inc. 245141858 MANUFACTURE(68682-006, 68682-007, 68682-008, 68682-009) , PACK(68682-006, 68682-007, 68682-008, 68682-009) , LABEL(68682-006, 68682-007, 68682-008, 68682-009)