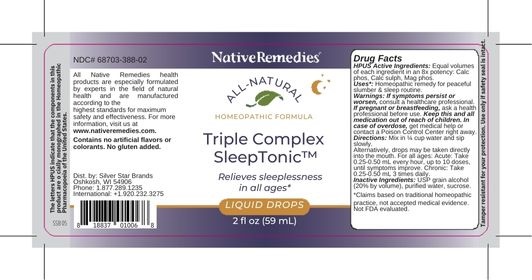
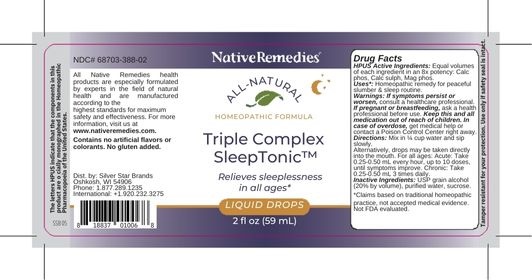
Label: TRIPLE COMPLEX SLEEP TONIC- calc phos, clac sulph, mag phos liquid
- NDC Code(s): 68703-388-02
- Packager: Silver Star Brands
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated December 25, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- HPUS Active Ingredients
- *Uses
- Warnings
- Directions
- OTHER SAFETY INFORMATION
- Inactive Ingredients
- KEEP OUT OF REACH OF CHILDREN
- KEEP OUT OF REACH OF CHILDREN
- PREGNANCY OR BREAST FEEDING
- PURPOSE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TRIPLE COMPLEX SLEEP TONIC
calc phos, clac sulph, mag phos liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68703-388 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAGNESIUM PHOSPHATE, DIBASIC TRIHYDRATE (UNII: HF539G9L3Q) (MAGNESIUM CATION - UNII:T6V3LHY838) MAGNESIUM PHOSPHATE, DIBASIC TRIHYDRATE 8 [hp_X] in 59 mL CALCIUM SULFIDE (UNII: 1MBW07J51Q) (CALCIUM SULFIDE - UNII:1MBW07J51Q) CALCIUM SULFIDE 8 [hp_X] in 59 mL CALCIUM PHOSPHATE (UNII: 97Z1WI3NDX) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM PHOSPHATE 8 [hp_X] in 59 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) SUCROSE (UNII: C151H8M554) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68703-388-02 59 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 03/09/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/09/2023 Labeler - Silver Star Brands (006070379) Registrant - Silver Star Brands (006070379) Establishment Name Address ID/FEI Business Operations OHM Pharma 030572478 manufacture(68703-388)