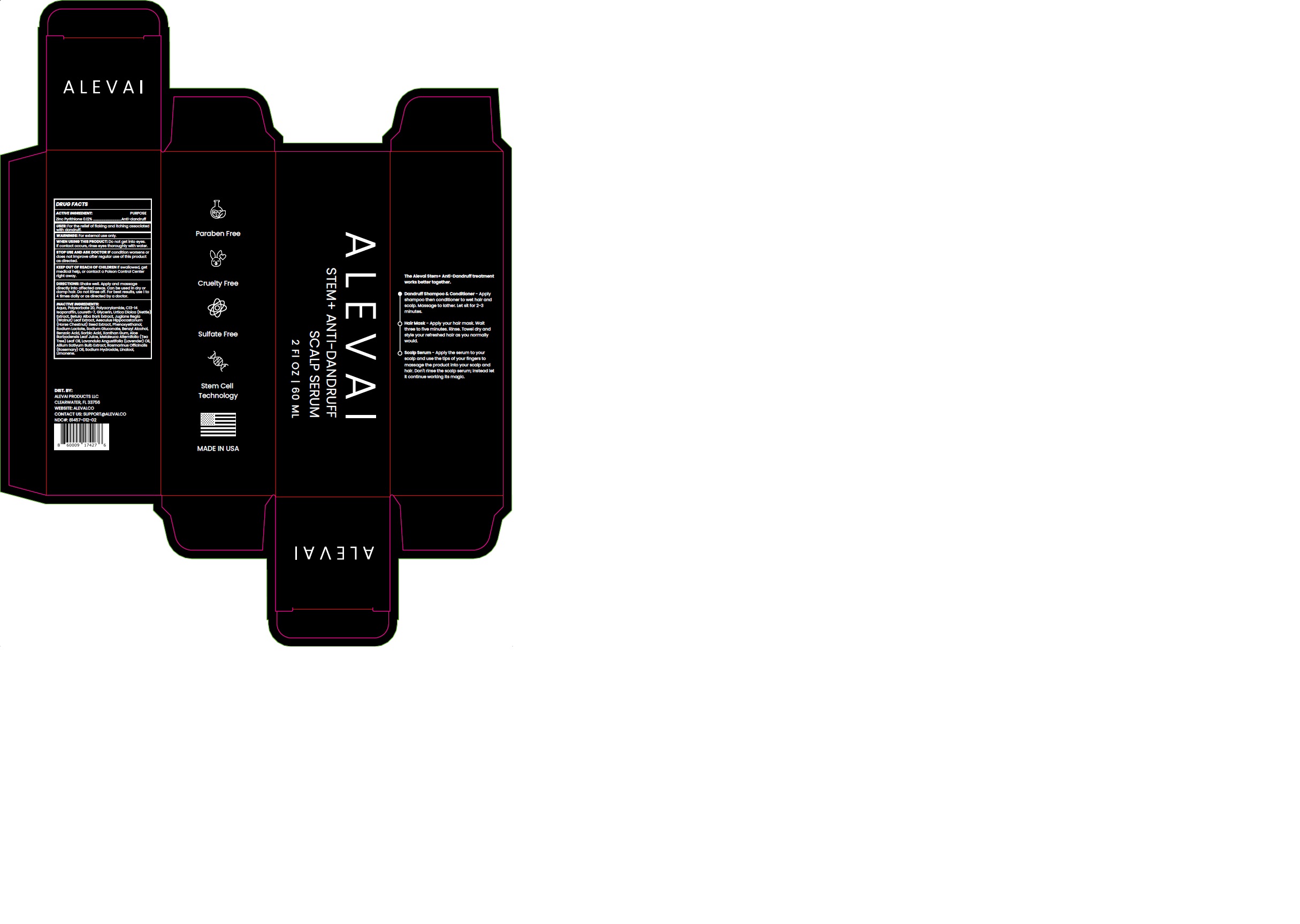
Label: ALEVAI - ANTI DANDRUFF SCALP SERUM- zinc pyrithione cream
- NDC Code(s): 81457-012-01, 81457-012-02
- Packager: ALEVAI PRODUCTS LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 7, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- USES:
- WARNINGS
- WHEN USING
- STOP USE AND ASK DOCTOR IF
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS:
-
INACTIVE INGREDIENT
Aqua, Polysorbate 20, Polyacrylamide, C13-14 Isoparaffin, Laureth-7, Glycerin, Urtica Dioica (Nettle) Extract, Betula Alba Bark Extract, Juglans Regia (Walnut) Leaf Extract, Aesculus Hippocastanum (Horse Chestnut) Seed Extract, Phenoxyethanol, Sodium Lactate, Sodium Gluconate, Benzyl Alcohol, Benzoic Acid, Sorbic Acid, Xanthan Gum, Aloe Barbadensis Leaf Juice, Melaleuca Alternifolia (Tea Tree) Leaf Oil, Lavandula Angustifolia (Lavender) Oil, Allium Sativum Bulb Extract, Rosmarinus Officinalis (Rosemary) Oil, Sodium Hydroxide, Linalool, Limonene.
- ALEVAI STEM + ANTI-DANDRUFF SCALP SERUM 1 FL OZ
- ALEVAI STEM + ANTI-DANDRUFF SCALP SERUM 2 FL OZ
-
INGREDIENTS AND APPEARANCE
ALEVAI - ANTI DANDRUFF SCALP SERUM
zinc pyrithione creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81457-012 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PYRITHIONE ZINC (UNII: R953O2RHZ5) (PYRITHIONE ZINC - UNII:R953O2RHZ5) PYRITHIONE ZINC 0.12 g in 100 mL Inactive Ingredients Ingredient Name Strength SORBIC ACID (UNII: X045WJ989B) URTICA DIOICA LEAF (UNII: X6M0DRN46Q) BETULA PUBESCENS BARK (UNII: 3R504894L9) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) POLYSORBATE 20 (UNII: 7T1F30V5YH) C13-14 ISOPARAFFIN (UNII: E4F12ROE70) MELALEUCA ALTERNIFOLIA LEAF (UNII: G43C57162K) ALLIUM SATIVUM WHOLE (UNII: IIF21WT8O3) PHENOXYETHANOL (UNII: HIE492ZZ3T) ROSEMARY OIL (UNII: 8LGU7VM393) LINALOOL, (+/-)- (UNII: D81QY6I88E) LIMONENE, (+)- (UNII: GFD7C86Q1W) HORSE CHESTNUT (UNII: 3C18L6RJAZ) SODIUM LACTATE (UNII: TU7HW0W0QT) LAURETH-7 (UNII: Z95S6G8201) XANTHAN GUM (UNII: TTV12P4NEE) SODIUM HYDROXIDE (UNII: 55X04QC32I) SODIUM GLUCONATE (UNII: R6Q3791S76) BENZYL ALCOHOL (UNII: LKG8494WBH) BENZOIC ACID (UNII: 8SKN0B0MIM) ALOE VERA LEAF (UNII: ZY81Z83H0X) JUGLANS REGIA LEAF (UNII: 85HKB87105) LAVENDER OIL (UNII: ZBP1YXW0H8) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81457-012-01 1 in 1 BOX 04/17/2023 1 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 2 NDC:81457-012-02 1 in 1 BOX 04/17/2023 2 60 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final M032 04/17/2023 Labeler - ALEVAI PRODUCTS LLC (117842557)