Label: EQUATE HYDROCORTISONE- hydrocortisone cream
- NDC Code(s): 79903-092-01
- Packager: WalMart
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
-
Uses
- temporarily relieves itching associated with minor skin irritations, inflammation, and rashes due to:
- eczema
- psoriasis
- poison ivy, oak, sumac
- insect bites
- detergents
- jewelry
- cosmetics
- soaps
- seborrheic dermatitis
- temporarily relieves external anal and genital itching
- other uses of this product should only be under the advice and supervision of a doctor
- temporarily relieves itching associated with minor skin irritations, inflammation, and rashes due to:
-
Warnings
For external use only.
Do not use
- in the genital area if you have a vaginal discharge. Consult a doctor.
- for the treatment of diaper rash. Consult a doctor.
When using this product
- avoid contact with eyes
- do not use more than directed unless told to do so by a doctor
- do not put directly into the rectum by using fingers or any mechanical device or applicator
-
Directions
-
for itching of skin irritation, inflammation, and rashes:
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
-
for external anal and genital itching, adults:
- when practical, cleanse the affected area with mild soap and warm water and rinse thoroughly
- gently dry by patting or blotting with toilet tissue or a soft cloth before applying
- apply to affected area not more than 3 to 4 times daily
- children under 12 years of age: ask a doctor
-
for itching of skin irritation, inflammation, and rashes:
- Other information
-
Inactive ingredients
aloe barbadensis leaf juice, ammonium acryloyldimethyltaurate/VP copolymer, bees wax, carbomer, ceteareth-6, cetyl alcohol, citric acid, coconut oil, corn starch, dimethicone, disodium EDTA, glycerin, isopropyl palmitate, methylparaben, mineral oil, propylene glycol, propylparaben, shea butter, sodium citrate, sodium lauryl sulfate, tocopheryl (vitamin E) acetate, white petrolatum, USP purified water
- Questions or comments?
- SPL UNCLASSIFIED SECTION
-
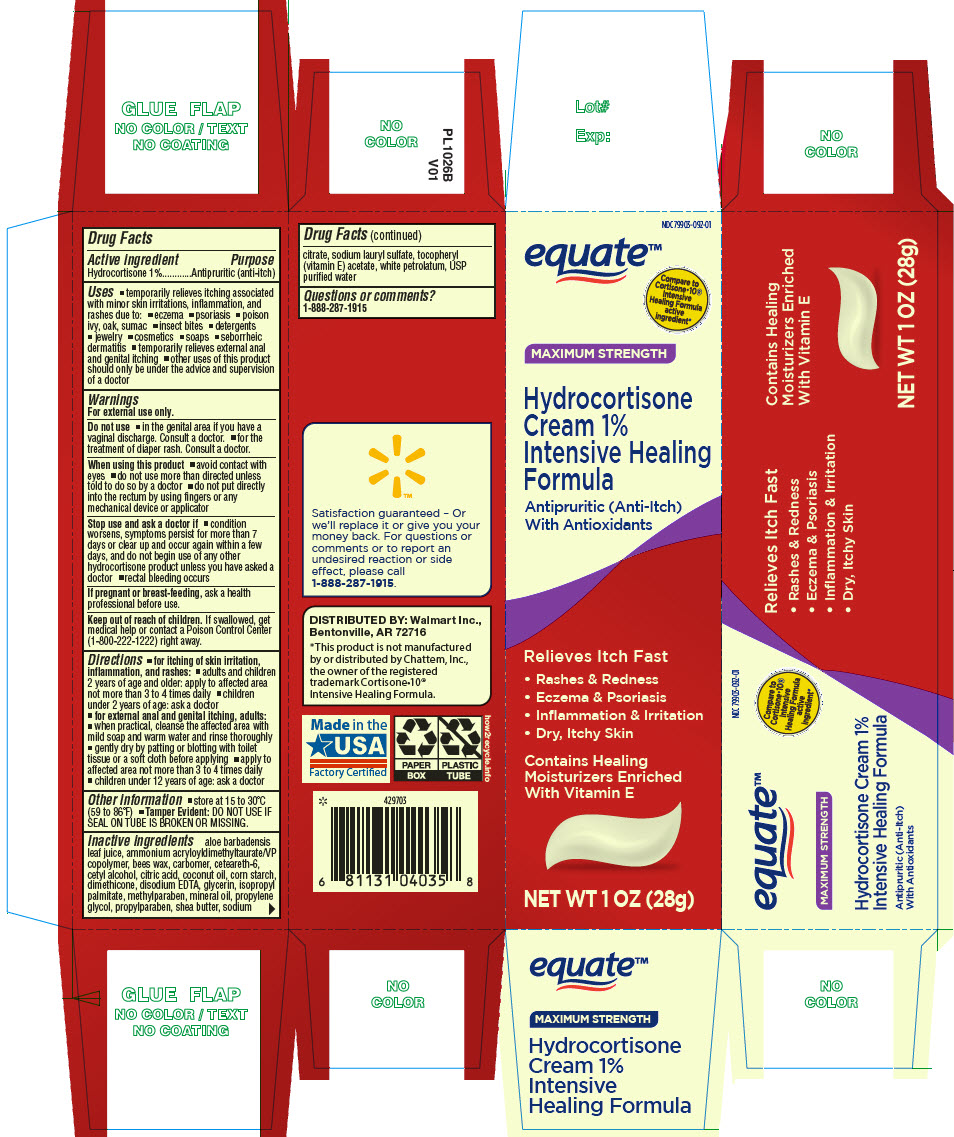
PRINCIPAL DISPLAY PANEL - 28 g Tube Carton
NDC 79903-092-01
equate™
Compare to
Cortisone•10®
Intensive
Healing Formula
active
ingredient*MAXIMUM STRENGTH
Hydrocortisone
Cream 1%
Intensive Healing
FormulaAntipruritic (Anti-Itch)
With AntioxidantsRelieves Itch Fast
- Rashes & Redness
- Eczema & Psoriasis
- Inflammation & Irritation
- Dry, Itchy Skin
Contains Healing
Moisturizers Enriched
With Vitamin ENET WT 1 OZ (28g)
-
INGREDIENTS AND APPEARANCE
EQUATE HYDROCORTISONE
hydrocortisone creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79903-092 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Hydrocortisone (UNII: WI4X0X7BPJ) (Hydrocortisone - UNII:WI4X0X7BPJ) Hydrocortisone 10 mg in 1 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) ammonium acryloyldimethyltaurate/VP copolymer (UNII: W59H9296ZG) YELLOW WAX (UNII: 2ZA36H0S2V) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) ceteareth-6 (UNII: 2RJS3559D3) cetyl alcohol (UNII: 936JST6JCN) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) coconut oil (UNII: Q9L0O73W7L) STARCH, CORN (UNII: O8232NY3SJ) dimethicone (UNII: 92RU3N3Y1O) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) glycerin (UNII: PDC6A3C0OX) isopropyl palmitate (UNII: 8CRQ2TH63M) methylparaben (UNII: A2I8C7HI9T) mineral oil (UNII: T5L8T28FGP) propylene glycol (UNII: 6DC9Q167V3) propylparaben (UNII: Z8IX2SC1OH) shea butter (UNII: K49155WL9Y) sodium (UNII: 9NEZ333N27) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79903-092-01 1 in 1 CARTON 11/18/2021 1 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 11/18/2021 Labeler - WalMart (051957769) Establishment Name Address ID/FEI Business Operations Natureplex LLC 062808196 MANUFACTURE(79903-092)

