Label: ARTEMISIA ARGYI LEAF- arm slim sticker patch
- NDC Code(s): 52784-010-01
- Packager: ZHENGZHOU GIANT BIOCHEMISTRY GROUP CO.,LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated February 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
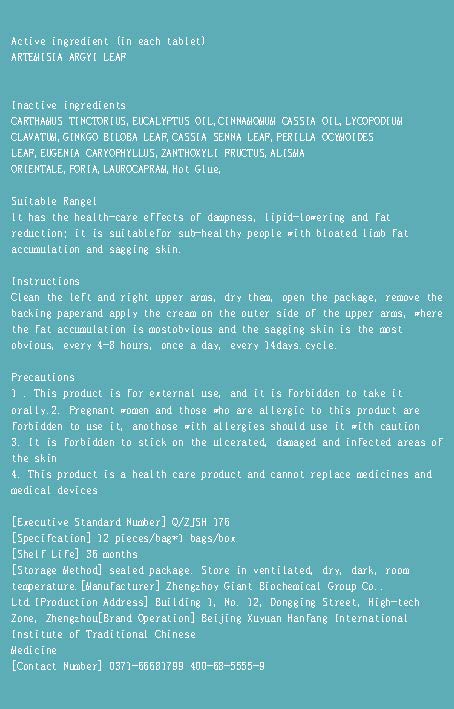
- Active ingredient
- Suitable Rangel
- KEEP OUT OF REACH OF CHILDREN
- Instructions
-
Warning
1 . This product is for external use, and it is forbidden to take itorally.
2. Pregnant women and those who are allergic to this product areforbidden to use it, anothose with allergies should use it with caution
3. It is forbidden to stick on the ulcerated,damaged and infected areas ofthe skin
4. This product is a health care product and cannot replace medicines andmedical devices - DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ARTEMISIA ARGYI LEAF
arm slim sticker patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:52784-010 Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ARTEMISIA ARGYI LEAF (UNII: 2JYC99Q0WZ) (ARTEMISIA ARGYI LEAF - UNII:2JYC99Q0WZ) ARTEMISIA ARGYI LEAF 16 g in 100 g Inactive Ingredients Ingredient Name Strength EUCALYPTUS OIL (UNII: 2R04ONI662) PERILLA FRUTESCENS LEAF (UNII: T4L5881Y68) CLOVE (UNII: K48IKT5321) ZANTHOXYLUM BUNGEANUM FRUIT (UNII: 3CIP16A418) ALISMA PLANTAGO-AQUATICA SUBSP. ORIENTALE ROOT (UNII: 555C7I140L) LAUROCAPRAM (UNII: 1F3X9DRV9X) SAFFLOWER (UNII: 4VBL71TY4Y) CHINESE CINNAMON OIL (UNII: A4WO0626T5) LYCOPODIUM CLAVATUM WHOLE (UNII: 005ICF6L27) GINKGO (UNII: 19FUJ2C58T) SENNA LEAF (UNII: AK7JF626KX) FU LING (UNII: XH37TWY5O4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52784-010-01 12 in 1 BOX 03/22/2023 1 24 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/22/2023 Labeler - ZHENGZHOU GIANT BIOCHEMISTRY GROUP CO.,LTD (527840360) Establishment Name Address ID/FEI Business Operations ZHENGZHOU GIANT BIOCHEMISTRY GROUP CO.,LTD 527840360 manufacture(52784-010)