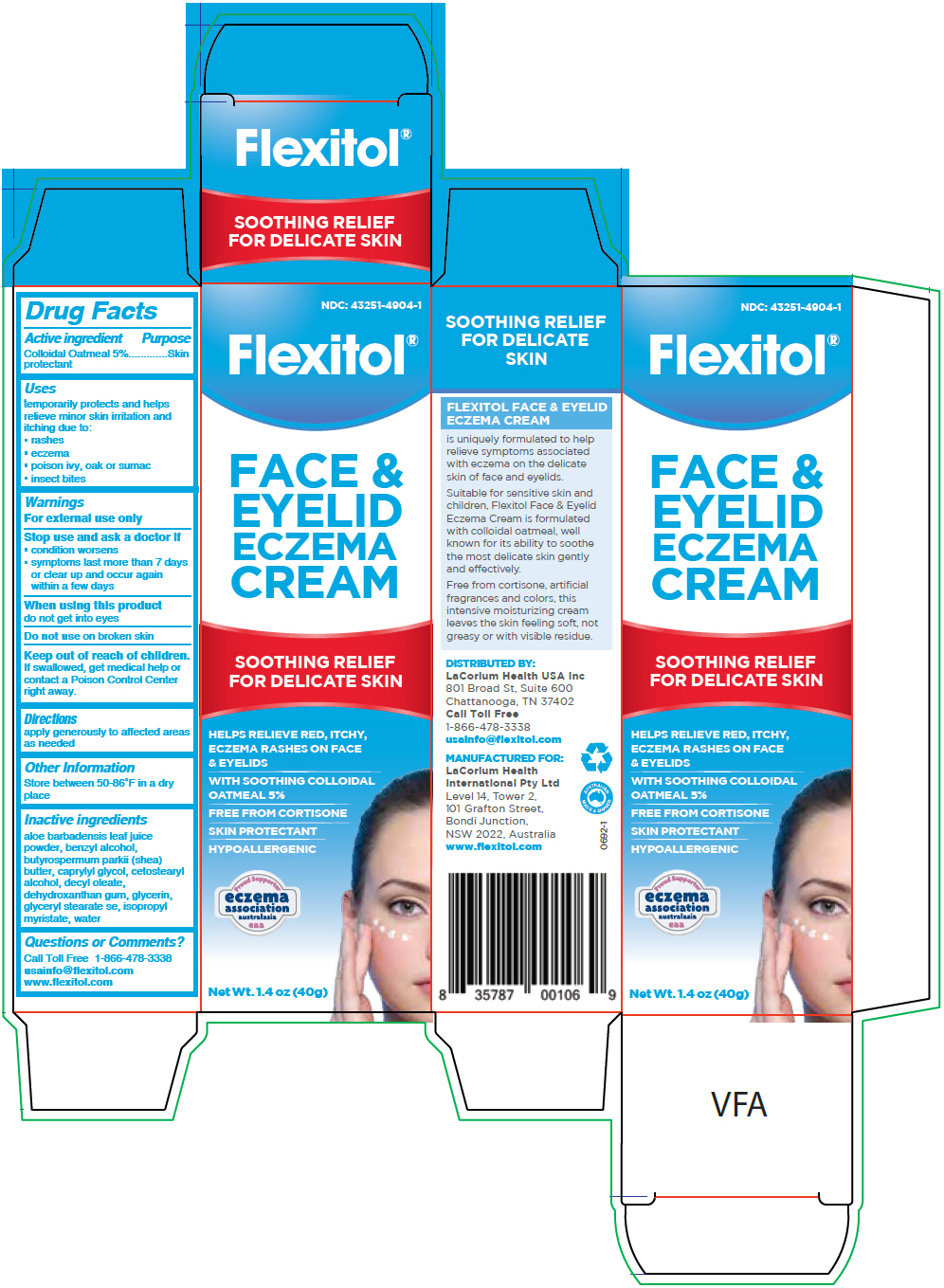
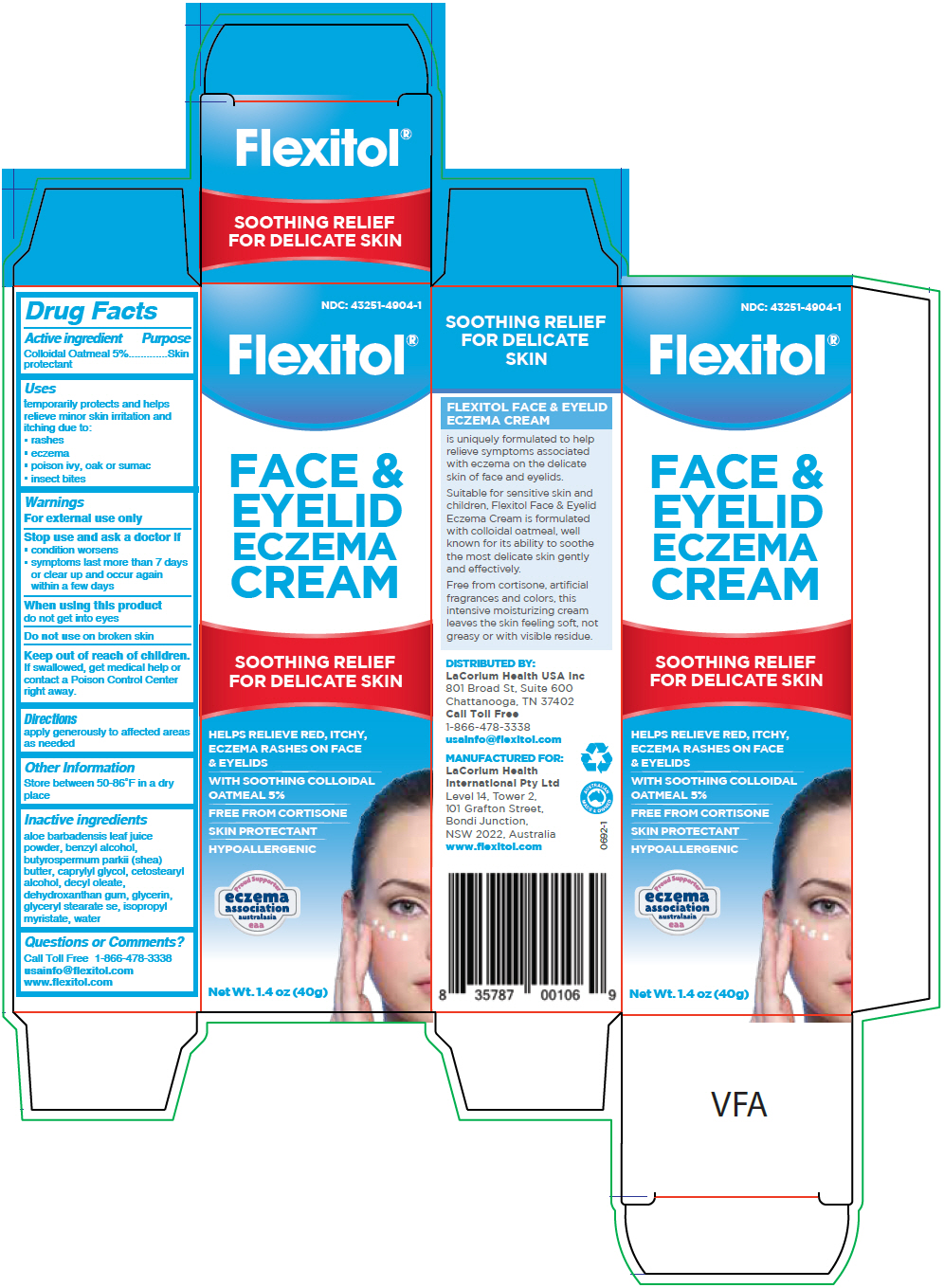
Label: FLEXITOL FACE AND EYELID ECZEMA- oatmeal cream
- NDC Code(s): 43251-4904-1
- Packager: LaCorium Health USA Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 10, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
- Warnings
- Directions
- Other Information
- Inactive ingredients
- Questions or Comments?
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 40 g Tube Carton
NDC: 43251-4904-1
Flexitol®
FACE &
EYELID
ECZEMA
CREAMSOOTHING RELIEF
FOR DELICATE SKINHELPS RELIEVE RED, ITCHY,
ECZEMA RASHES ON FACE
& EYELIDSWITH SOOTHING COLLOIDAL
OATMEAL 5%FREE FROM CORTISONE
SKIN PROTECTANT
HYPOALLERGENIC
Proud Supporter
eczema
association
australia
eaaNet Wt. 1.4 oz (40g)
-
INGREDIENTS AND APPEARANCE
FLEXITOL FACE AND EYELID ECZEMA
oatmeal creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:43251-4904 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength oatmeal (UNII: 8PI54V663Y) (oatmeal - UNII:8PI54V663Y) oatmeal 50 mg in 1 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Cetostearyl Alcohol (UNII: 2DMT128M1S) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) Isopropyl Myristate (UNII: 0RE8K4LNJS) DECYL OLEATE (UNII: ZGR06DO97T) Glycerin (UNII: PDC6A3C0OX) BENZYL ALCOHOL (UNII: LKG8494WBH) SHEA BUTTER (UNII: K49155WL9Y) Caprylyl glycol (UNII: 00YIU5438U) Dehydroxanthan gum (UNII: 63ZP7I1BQO) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:43251-4904-1 1 in 1 CARTON 01/01/2022 1 40 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 01/01/2022 Labeler - LaCorium Health USA Inc. (111254392) Establishment Name Address ID/FEI Business Operations Ross Laboratories Aust Pty Ltd 753529114 MANUFACTURE(43251-4904) , LABEL(43251-4904) , ANALYSIS(43251-4904) , PACK(43251-4904)