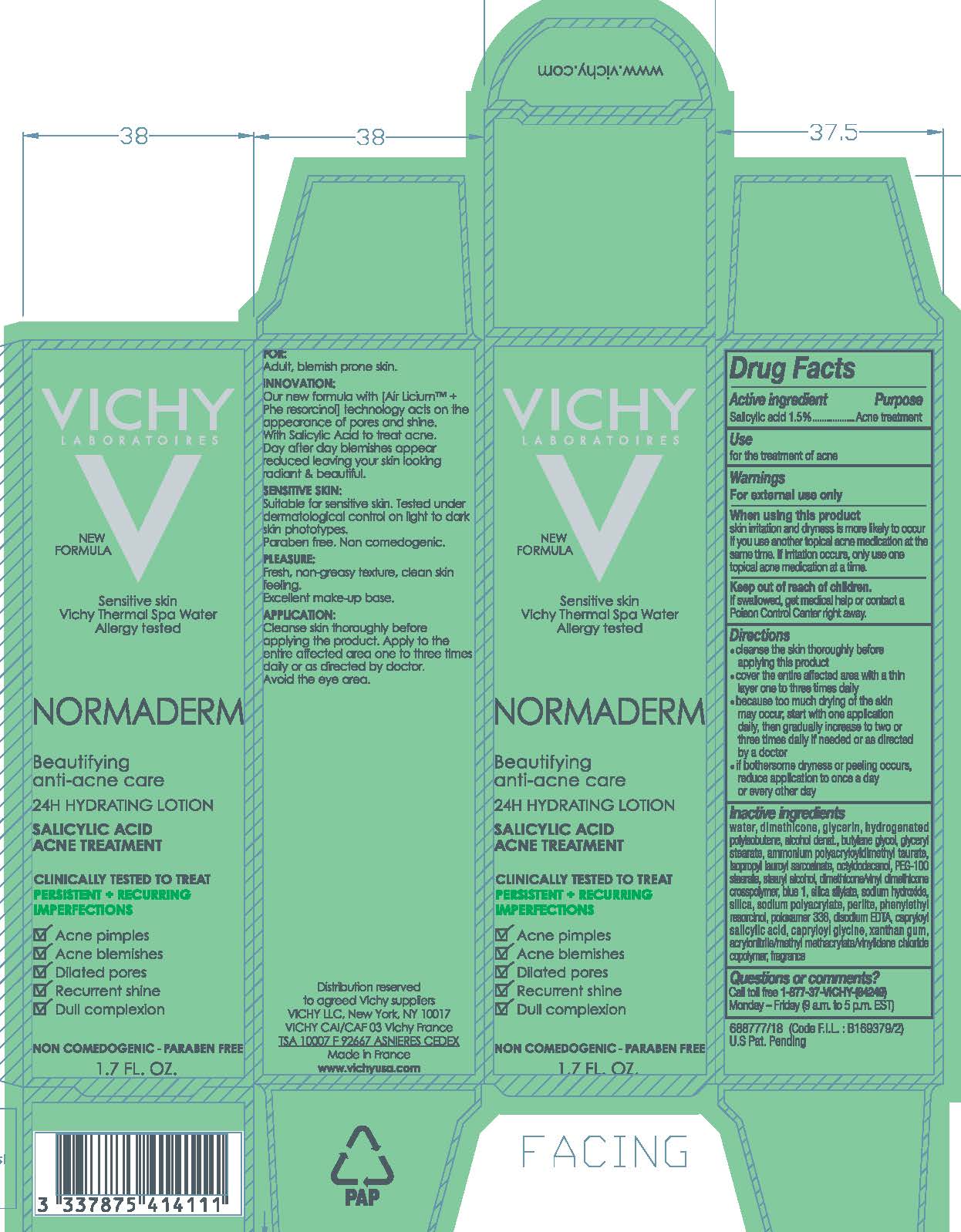
Label: VICHY LABORATOIRES NORMADERM BEAUTIFYING ANTI ACNE CARE 24 H HYDRATING ACNE TREATMENT- salicylic acid lotion
- NDC Code(s): 49967-411-01
- Packager: L'Oreal USA Products Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 31, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Use
- Warnings
- When using this product
- Keep out of reach of children.
-
Directions
- cleanse the skin thoroughly before applying this product
- cover the entire affected area with a thin layer one to three times daily
- because too much drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day
-
Inactive ingredients
water, dimethicone, glycerin, hydrogenated polyisobutene, alcohol denat., butylene glycol, glyceryl stearate, ammonium polyacryloyldimethyl taurate, isopropyl lauroyl sarcosinate, octyldodecanol, PEG-100 stearate, stearyl alcohol, dimethicone/vinyl dimethicone crosspolymer blue 1, silica silylate, sodium hydroxide, silica, sodium polyacrylate, perlite, phenylethyl resorcinol, poloxamer 338, disodium EDTA, capryloyl salicylic acid, capryloyl glycine, xanthan gum, acrylonitrile/methyl methacrylate/vinylidene chloride copolymer, fragrance
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
VICHY LABORATOIRES NORMADERM BEAUTIFYING ANTI ACNE CARE 24 H HYDRATING ACNE TREATMENT
salicylic acid lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49967-411 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 15 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCERIN (UNII: PDC6A3C0OX) HYDROGENATED POLYBUTENE (1300 MW) (UNII: 7D1YQ9Y5EZ) ALCOHOL (UNII: 3K9958V90M) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) ISOPROPYL LAUROYL SARCOSINATE (UNII: LYR06W430J) OCTYLDODECANOL (UNII: 461N1O614Y) PEG-100 STEARATE (UNII: YD01N1999R) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (HARD PARTICLE) (UNII: H895X08VNQ) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) SODIUM HYDROXIDE (UNII: 55X04QC32I) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM POLYACRYLATE (8000 MW) (UNII: 285CYO341L) PERLITE (UNII: 0SG101ZGK9) PHENYLETHYL RESORCINOL (UNII: G37UFG162O) POLOXAMER 338 (UNII: F75JV2T505) EDETATE DISODIUM (UNII: 7FLD91C86K) CAPRYLOYL SALICYLIC ACID (UNII: 5F7PJF6AA4) CAPRYLOYL GLYCINE (UNII: 8TY5YO42NJ) XANTHAN GUM (UNII: TTV12P4NEE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49967-411-01 1 in 1 CARTON 01/01/2016 06/10/2025 1 50 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 01/01/2016 06/10/2025 Labeler - L'Oreal USA Products Inc (002136794) Establishment Name Address ID/FEI Business Operations COSMETIQUE ACTIVE PRODUCTION 282658798 manufacture(49967-411) , pack(49967-411)