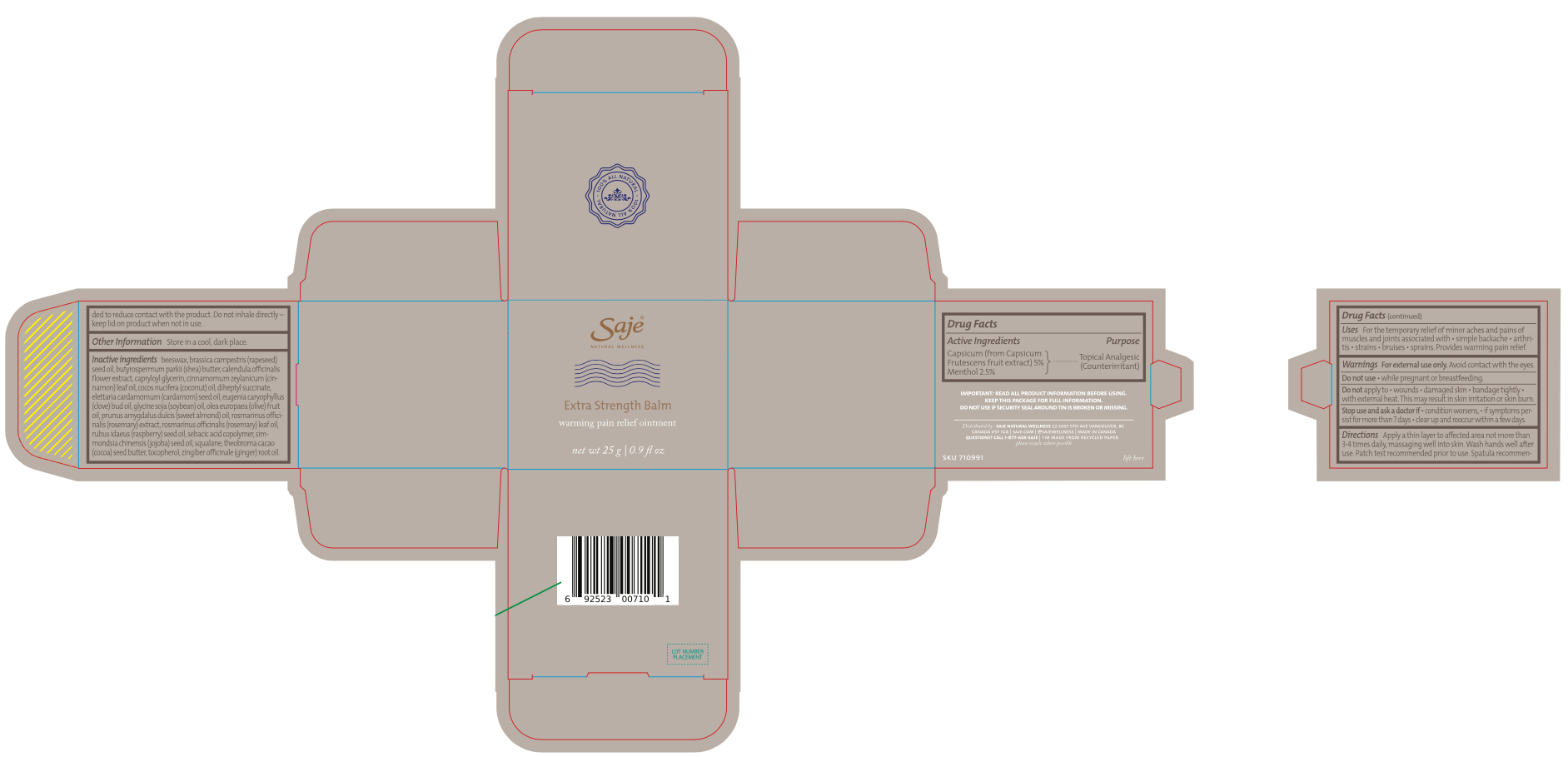
Label: EXTRA STRENGTH BALM- capsicum and menthol ointment
- NDC Code(s): 70983-027-01
- Packager: Saje Natural Business Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- WHEN USING
- PREGNANCY OR BREAST FEEDING
- DO NOT USE
- STOP USE
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
-
INACTIVE INGREDIENT
Beeswax, butyrospermum parkii (shea) butter, calendula officinalis flower extract, capryloyl glycerin, cinnamomum zeylanicum (cinnamon) leaf oil, cocos nucifera (coconut) oil, diheptyl succinate, elettaria cardamomum (cardamom) seed oil, eugenia caryophyllus (clove) bud oil, glycine soja (soybean) oil, olea europaea (olive) fruit oil, prunus amygdalus dulcis (sweet almond) oil, rosmarinus officinalis (rosemary) extract, rosmarinus officinalis (rosemary) leaf oil, rubus idaeus (raspberry) seed oil, sebacic acid copolymer, simmondsia chinensis (jojoba) seed oil, squalane, theobroma cacao (cocoa) seed butter, tocopherol, zingiber officinale (ginger) root oil
-
OTHER SAFETY INFORMATION
Important: Read all product information before using. Keep this package for full information.
Do not use if security seal is broken or missing.
distributed by: saje natural wellness 22 east 5th ave vancouver, bc canada v5t 1g8
saje.com | 1-877-ask-saje | @sajewellness
made in Canada
Questions? Call 1-877-ASK-SAJE
I'm made from recycled paper
please recycle where possible - DESCRIPTION
- KEEP OUT OF REACH OF CHILDREN
- Extra Strength Balm - outer label
-
INGREDIENTS AND APPEARANCE
EXTRA STRENGTH BALM
capsicum and menthol ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70983-027 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 2258.75 mg in 1 g CAPSICUM (UNII: 00UK7646FG) (CAPSICUM - UNII:00UK7646FG) CAPSICUM 5000 mg in 1 g Inactive Ingredients Ingredient Name Strength DIHEPTYL SUCCINATE (UNII: 057N7SS26Y) JOJOBA OIL (UNII: 724GKU717M) ALMOND OIL (UNII: 66YXD4DKO9) CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) SHEA BUTTER (UNII: K49155WL9Y) COCOA BUTTER (UNII: 512OYT1CRR) CAPRYLOYL GLYCERIN/SEBACIC ACID COPOLYMER (2000 MPA.S) (UNII: N7YC58165T) SQUALENE (UNII: 7QWM220FJH) TOCOPHEROL (UNII: R0ZB2556P8) ROSEMARY (UNII: IJ67X351P9) APIS MELLIFERA (UNII: 7S82P3R43Z) OLIVE OIL (UNII: 6UYK2W1W1E) CARDAMOM OIL (UNII: JM0KJ091HZ) COCONUT OIL (UNII: Q9L0O73W7L) RASPBERRY SEED OIL (UNII: 9S8867952A) CLOVE OIL (UNII: 578389D6D0) CINNAMON LEAF OIL (UNII: S92U8SQ71V) SOYBEAN OIL (UNII: 241ATL177A) GINGER OIL (UNII: SAS9Z1SVUK) ROSEMARY OIL (UNII: 8LGU7VM393) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70983-027-01 1 in 1 BOX 02/24/2023 1 25 g in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 02/18/2023 Labeler - Saje Natural Business Inc (080465432)