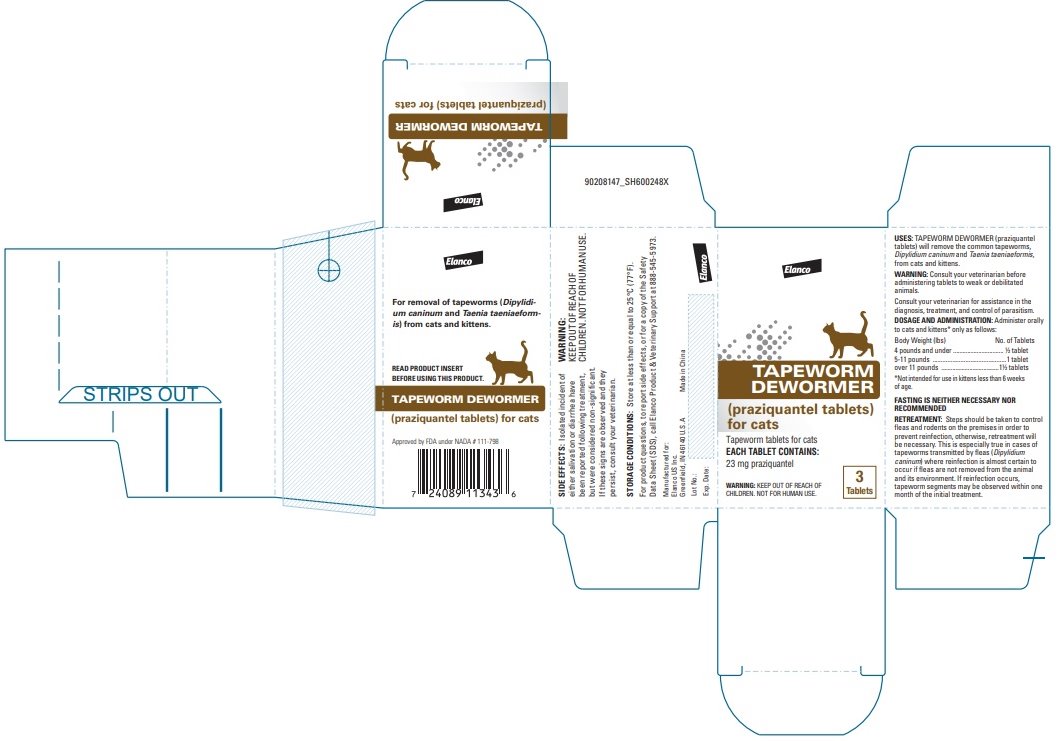
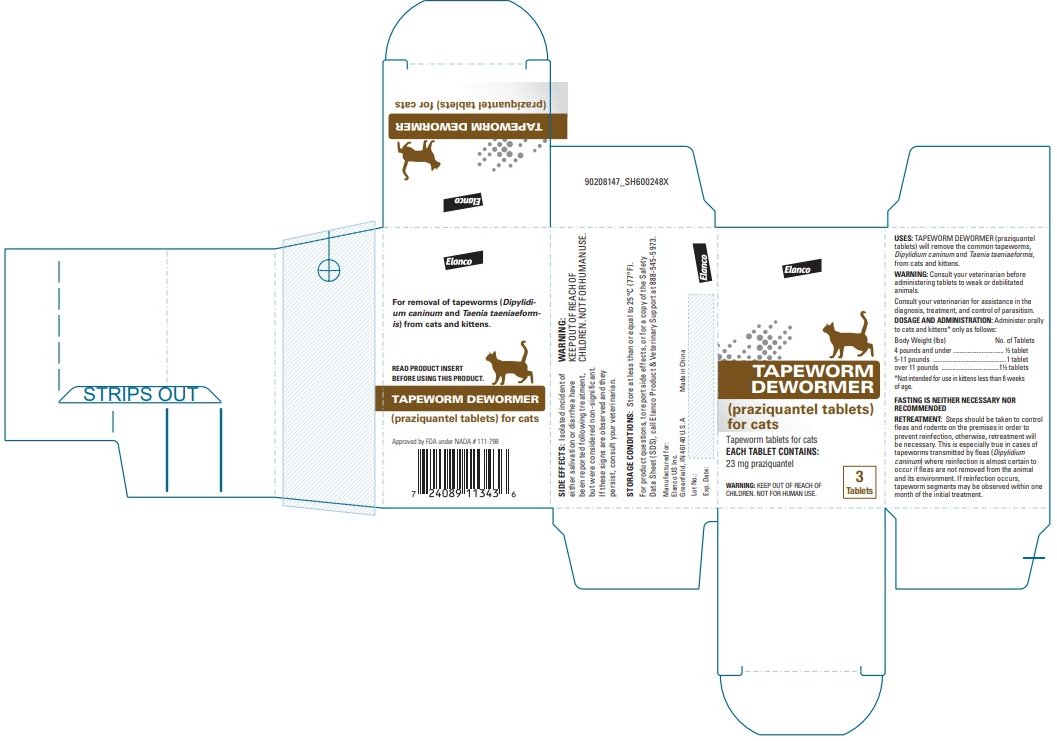
Label: TAPEWORM DEWORMER FOR CATS- praziquantel tablet
- NDC Code(s): 58198-0063-1, 58198-0063-2
- Packager: Elanco US Inc.
- Category: OTC ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated October 24, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- DESCRIPTION:
- USES:
-
DOSAGE AND ADMINISTRATION:
Tapeworm Dewormer (praziquantel tablets) may be given directly in the mouth or crumbled and mixed with the food. Administer to cats and kittens* only as follows:
*Not intended for use in kittens less than 6 weeks of age. Body Weight (lbs)
No. of Tablets
4 pounds and under
½ tablet
5-11 pounds
1 tablet
Over 11 pounds
1 ½ tablets
FASTING IS NEITHER NECESSARY NOR RECOMMENDED.
-
RETREATMENT:
Steps should be taken to control fleas and rodents on the premises in order to prevent reinfection; otherwise, retreatment will be necessary. This is especially true in cases of tapeworms transmitted by fleas (Dipylidium caninum) where reinfection is almost certain to occur if fleas are not removed from the animal and its environment. If reinfection occurs, tapeworm segments may be observed within one month of the initial treatment.
- SIDE EFFECTS:
-
Questions?
For product questions, to report side effects, or for a copy of the Safety Data Sheet (SDS), call Elanco Product & Veterinary Support at 888-545-5973.
For additional information about reporting side effects for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae.
- WARNING:
- HOW SUPPLIED:
-
STORAGE CONDITIONS:
Store at less than or equal to 25°C (77°F).
Elanco™
Manufactured for:
Elanco US Inc.
Greenfield, IN 46140 U.S.A
Approved by FDA under NADA # 111-798Elanco and the diagonal bar logo are trademarks of Elanco or its affiliates.
Made in China Revised: March 2022
90208148_PA600248X
© 2022 Elanco or its affiliates
- Principal Display Panel - 23 mg Carton Label
- Principal Display Panel – 23 mg Bottle Label
-
INGREDIENTS AND APPEARANCE
TAPEWORM DEWORMER FOR CATS
praziquantel tabletProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:58198-0063 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PRAZIQUANTEL (UNII: 6490C9U457) (PRAZIQUANTEL - UNII:6490C9U457) PRAZIQUANTEL 23 mg Product Characteristics Color white (white) Score 2 pieces Shape ROUND (ROUND) Size 10mm Flavor Imprint Code 23 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58198-0063-1 1 in 1 CARTON 1 NDC:58198-0063-2 3 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA111798 05/16/2014 Labeler - Elanco US Inc. (966985624) Establishment Name Address ID/FEI Business Operations TriRx Shawnee LLC 118187894 MANUFACTURE, ANALYSIS, PACK, LABEL Establishment Name Address ID/FEI Business Operations Merck KG auf Aktien 342249299 API MANUFACTURE Establishment Name Address ID/FEI Business Operations Hisun Pharmaceutical (Nantong) Co., Ltd. 421314476 API MANUFACTURE