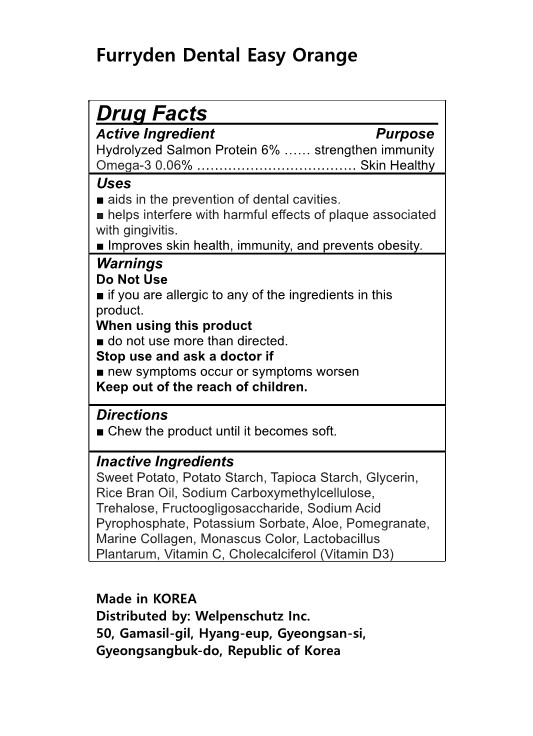
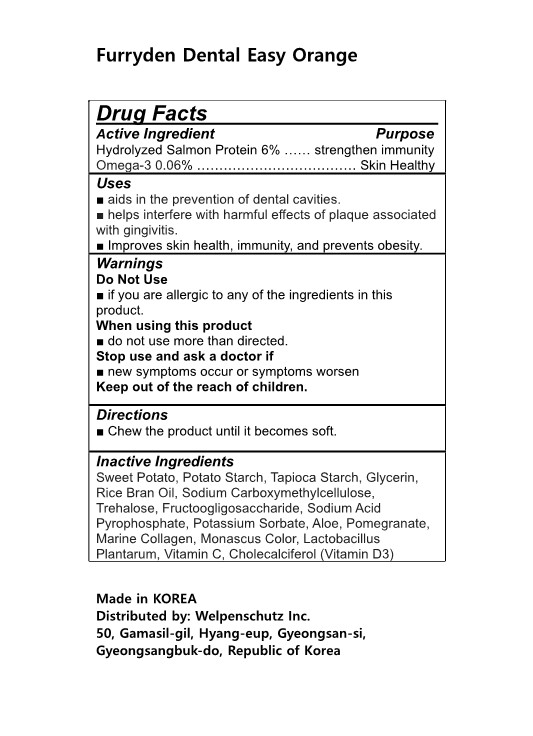
Label: FURRYDEN DENTAL EASY ORANGE- hydrolyzed salmon protein, omega-3 gum, chewing
- NDC Code(s): 83332-301-01
- Packager: Welpenschutz Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated February 9, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purposes
- Uses
- Warnings
- Warnings
- Warnings
- Warnings
- Warnings
- Directions
- Inactive Ingredients
- Label
-
INGREDIENTS AND APPEARANCE
FURRYDEN DENTAL EASY ORANGE
hydrolyzed salmon protein, omega-3 gum, chewingProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83332-301 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALMON, UNSPECIFIED (UNII: 6122W2M0GB) (SALMON, UNSPECIFIED - UNII:6122W2M0GB) SALMON, UNSPECIFIED 6 g in 100 mL OMEGA-3 FATTY ACIDS (UNII: 71M78END5S) (OMEGA-3 FATTY ACIDS - UNII:71M78END5S) OMEGA-3 FATTY ACIDS 0.06 g in 100 mL Inactive Ingredients Ingredient Name Strength RICE BRAN OIL (UNII: LZO6K1506A) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) TREHALOSE (UNII: B8WCK70T7I) SODIUM ACID PYROPHOSPHATE (UNII: H5WVD9LZUD) STARCH, TAPIOCA (UNII: 24SC3U704I) ALOE (UNII: V5VD430YW9) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) STARCH, POTATO (UNII: 8I089SAH3T) SWEET POTATO (UNII: M9WGG9Z9GK) LACTIPLANTIBACILLUS PLANTARUM (UNII: QFC21096ON) CHOLECALCIFEROL (UNII: 1C6V77QF41) POMEGRANATE (UNII: 56687D1Z4D) FRUCTOSE (UNII: 6YSS42VSEV) MONASCUS PURPUREUS (UNII: 1EU532DCW4) GLYCERIN (UNII: PDC6A3C0OX) HYDROLYSED MARINE COLLAGEN (ENZYMATIC; 2000 MW) (UNII: 2WID9OCG7P) ASCORBIC ACID (UNII: PQ6CK8PD0R) Product Characteristics Color orange Score score with uneven pieces Shape ROUND Size 5mm Flavor ORANGE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83332-301-01 200 mL in 1 POUCH; Type 0: Not a Combination Product 02/17/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 02/17/2023 Labeler - Welpenschutz Inc. (695930167) Registrant - Welpenschutz Inc. (695930167) Establishment Name Address ID/FEI Business Operations Welpenschutz Inc. 695930167 manufacture(83332-301)