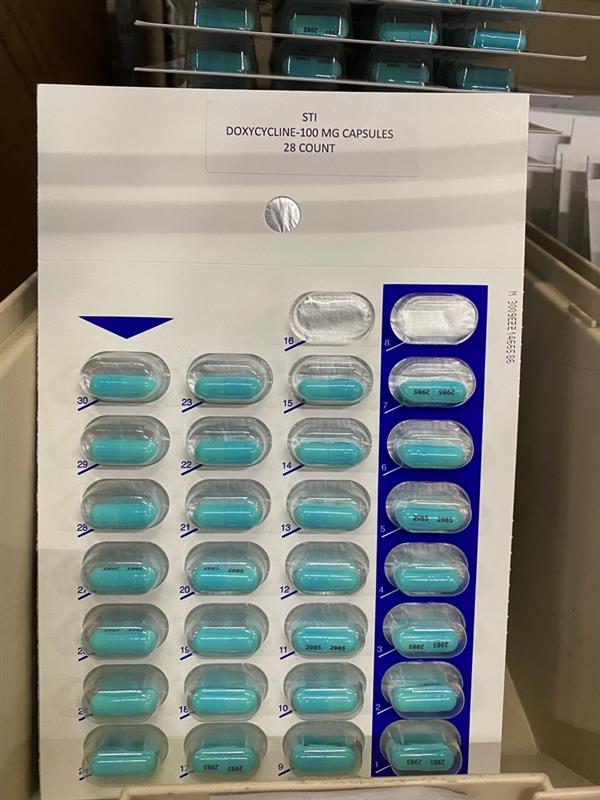
Label: DOXYCYCLINE HYCLATE capsule
- NDC Code(s): 83112-985-05, 83112-985-28, 83112-985-42, 83112-985-56
- Packager: Health Department, Oklahoma State
- This is a repackaged label.
- Source NDC Code(s): 62135-985
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 8, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Doxycycline Hyclate 100 mg Warnings Section
The use of drugs of the tetracycline class during tooth development (last half of pregnancy, infancy and childhood to the age of 8 years) may cause permanent discoloration of the teeth (yellow-gray-brown). This adverse reaction is more common during long-term use of the drugs, but it has been observed following repeated short-term courses. Enamel hypoplasia has also been reported. Use doxycycline in pediatric patients 8 years of age or less only when the potential benefits are expected to outweigh the risks in severe or life-threatening conditions (e.g., anthrax, Rocky Mountain spotted fever), particularly when there are no alternative therapies.
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including doxycycline, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following the use of antibacterial drugs. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing use of antibacterial drugs not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Severe skin reactions, such as exfoliative dermatitis, erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis, and drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported in patients receiving doxycycline. (See ADVERSE REACTIONS.) If severe skin reactions occur, doxycycline should be discontinued immediately and appropriate therapy should be instituted.
Intracranial hypertension (IH, pseudotumor cerebri) has been associated with the use of tetracyclines including doxycycline. Clinical manifestations of IH include headache, blurred vision, diplopia, and vision loss; papilledema can be found on fundoscopy. Women of childbearing age who are overweight or have a history of IH are at greater risk for developing tetracycline associated IH. Concomitant use of isotretinoin and doxycycline should be avoided because isotretinoin is also known to cause pseudotumor cerebri.
Although IH typically resolves after discontinuation of treatment, the possibility for permanent visual loss exists. If visual disturbance occurs during treatment, prompt ophthalmologic evaluation is warranted. Since intracranial pressure can remain elevated for weeks after drug cessation patients should be monitored until they stabilize.
All tetracyclines form a stable calcium complex in any bone-forming tissue. A decrease in fibula growth rate has been observed in prematures given oral tetracycline in doses of 25 mg/kg every 6 hours. This reaction was shown to be reversible when the drug was discontinued.
Results of animal studies indicate that tetracyclines cross the placenta, are found in fetal tissues, and can have toxic effects on the developing fetus (often related to retardation of skeletal development). Evidence of embryotoxicity has also been noted in animals treated early in pregnancy. If any tetracycline is used during pregnancy or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
The antianabolic action of the tetracyclines may cause an increase in BUN. Studies to date indicate that this does not occur with the use of doxycycline in patients with impaired renal function.
Photosensitivity manifested by an exaggerated sunburn reaction has been observed in some individuals taking tetracyclines. Patients apt to be exposed to direct sunlight or ultraviolet light should be advised that this reaction can occur with tetracycline drugs, and treatment should be discontinued at the first evidence of skin erythema.
- Doxycycline Packaging
-
INGREDIENTS AND APPEARANCE
DOXYCYCLINE HYCLATE
doxycycline hyclate capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:83112-985(NDC:62135-985) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOXYCYCLINE HYCLATE (UNII: 19XTS3T51U) (DOXYCYCLINE ANHYDROUS - UNII:334895S862) DOXYCYCLINE ANHYDROUS 100 mg Product Characteristics Color blue (Light Blue) Score no score Shape CAPSULE Size 21mm Flavor Imprint Code 2985 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83112-985-28 28 in 1 PACKAGE; Type 0: Not a Combination Product 02/01/2023 2 NDC:83112-985-42 42 in 1 PACKAGE; Type 0: Not a Combination Product 02/08/2023 3 NDC:83112-985-56 56 in 1 PACKAGE; Type 0: Not a Combination Product 02/08/2023 4 NDC:83112-985-05 1 in 1 PACKET; Type 0: Not a Combination Product 02/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA062500 02/01/2023 Labeler - Health Department, Oklahoma State (143673015) Registrant - Health Department, Oklahoma State (143673015) Establishment Name Address ID/FEI Business Operations Health Department, Oklahoma State 143673015 repack(83112-985)