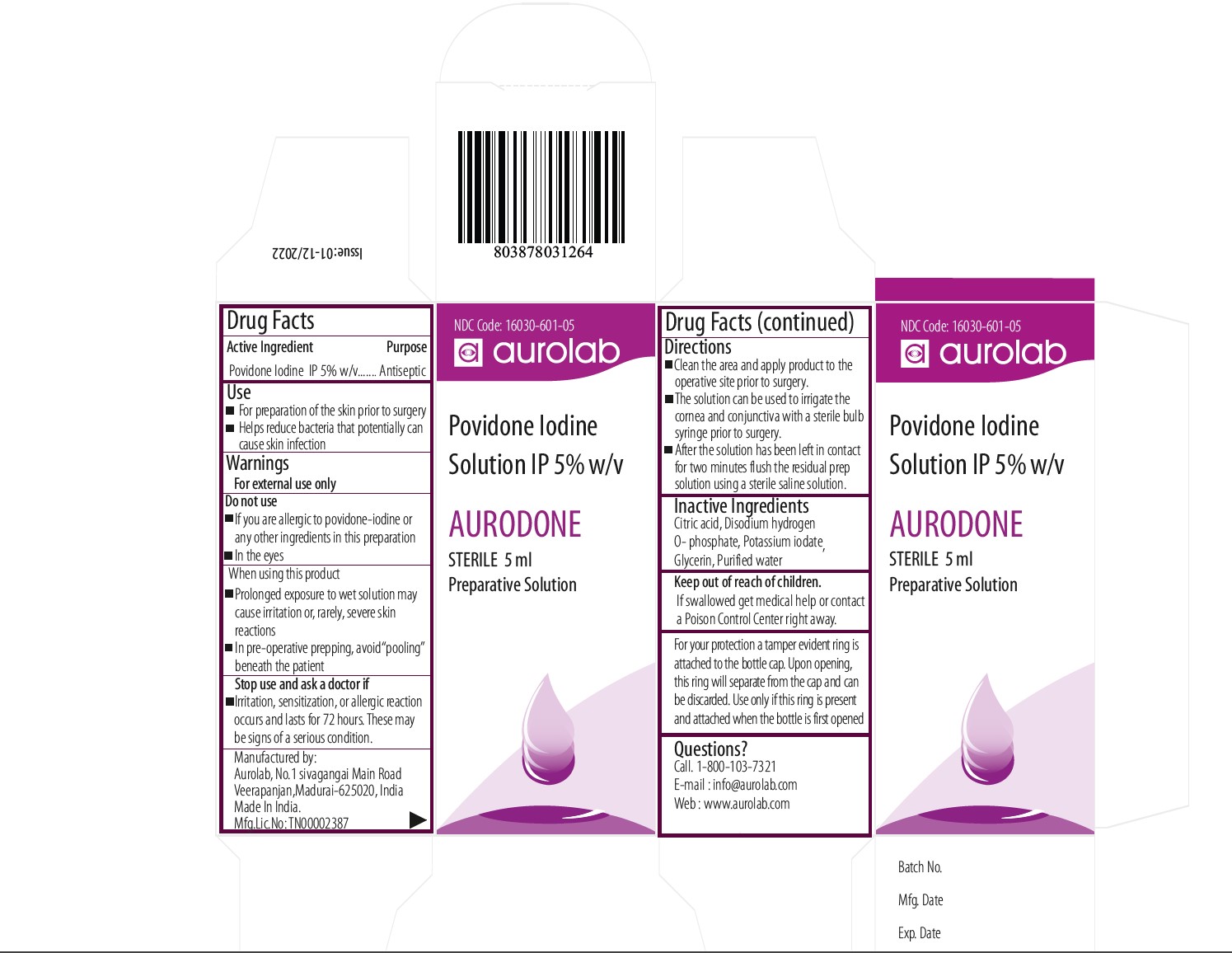
Label: AURODONE POVIDONE IODINE 5 % OPTHALMIC SOLUTION- povidone iodine 5 % topical solution solution
- NDC Code(s): 16030-601-05
- Packager: Aurolab
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 26, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- USE
- QUESTIONS
- KEEP OUT OF REACH OF CHILDREN
- STOP USE
- DO NOT USE
- WARNINGS
- INDIACATIONS AND USAGE
- Purpose
-
Dose
- Clean the area and apply product to the operative site prior to surgery.
- The solution can be used to irrigate the cornea and conjunctiva with a sterile bulb syringe prior to surgery.
- After the solution has been left in contact for two minutes flush the residual prep solution using a sterile saline solution.
- PACKAGE CARTON
-
INGREDIENTS AND APPEARANCE
AURODONE POVIDONE IODINE 5 % OPTHALMIC SOLUTION
povidone iodine 5 % topical solution solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:16030-601 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) CITRIC ACID ACETATE (UNII: DSO12WL7AU) POTASSIUM IODATE (UNII: I139E44NHL) DISODIUM HYDROGEN CITRATE (UNII: 6FO62KCQ7A) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:16030-601-05 5 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 01/31/2023 Labeler - Aurolab (677319965) Establishment Name Address ID/FEI Business Operations Aurolab 677319965 manufacture(16030-601)