Label: ALBOTHYL- policresulen solution liquid
- NDC Code(s): 72988-0035-1
- Packager: Lydia Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated July 10, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
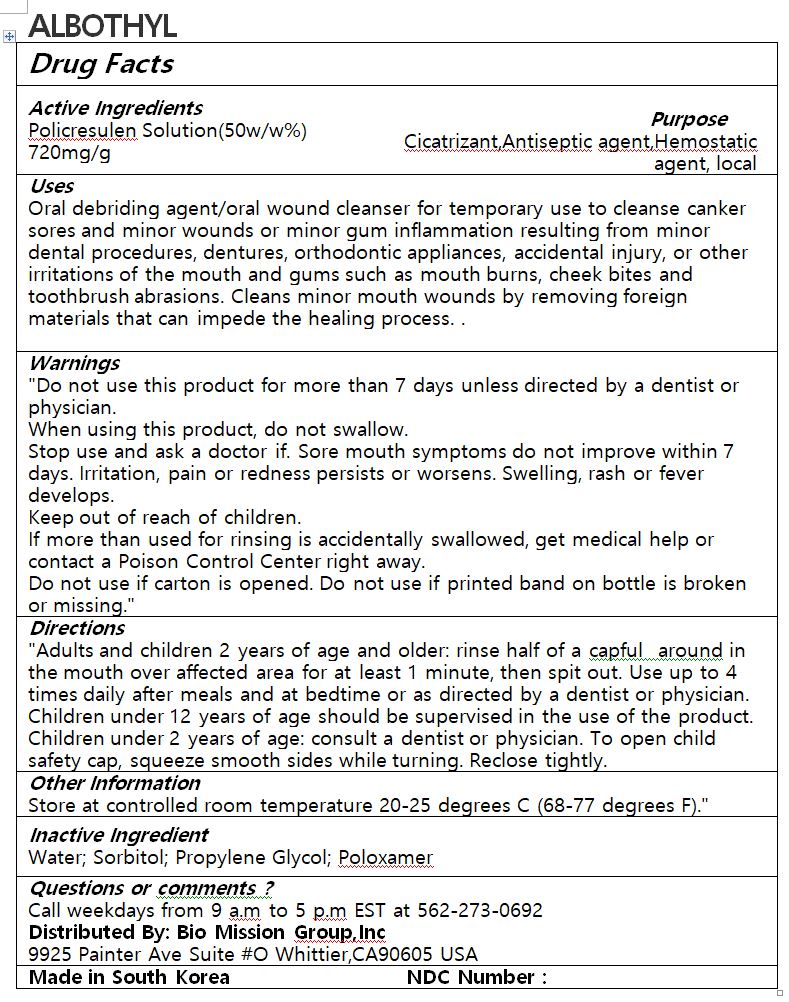
- ACTIVE INGREDIENT
-
PURPOSE
Oral debriding agent/oral wound cleanser for temporary use to cleanse canker sores and minor wounds or minor gum inflammation resulting from minor dental procedures, dentures, orthodontic appliances, accidental injury, or other irritations of the mouth and gums such as mouth burns, cheek bites and toothbrush abrasions. Cleans minor mouth wounds by removing foreign materials that can impede the healing process
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
Adults and children 2 years of age and older: rinse half of a capful around in the mouth over affected area for at least 1 minute, then spit out. Use up to 4 times daily after meals and at bedtime or as directed by a dentist or physician. Children under 12 years of age should be supervised in the use of the product. Children under 2 years of age: consult a dentist or physician. To open child safety cap, squeeze smooth sides while turning. Reclose tightly
-
WARNINGS
Do not use this product for more than 7 days unless directed by a dentist or physician.
When using this product, do not swallow.
Stop use and ask a doctor if. Sore mouth symptoms do not improve within 7 days. Irritation, pain or redness persists or worsens. Swelling, rash or fever develops.
Keep out of reach of children.
If more than used for rinsing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Do not use if carton is opened. Do not use if printed band on bottle is broken or missing
- INACTIVE INGREDIENT
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALBOTHYL
policresulen solution liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72988-0035 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLICRESULEN (UNII: 6I19M5GB0G) (POLICRESULEN - UNII:6I19M5GB0G) POLICRESULEN 720 mg in 1 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72988-0035-1 5 g in 1 BOTTLE; Type 0: Not a Combination Product 01/23/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 01/23/2023 Labeler - Lydia Co., Ltd. (695735569) Registrant - Lydia Co., Ltd. (695735569) Establishment Name Address ID/FEI Business Operations Lydia Co., Ltd. 695735569 manufacture(72988-0035)