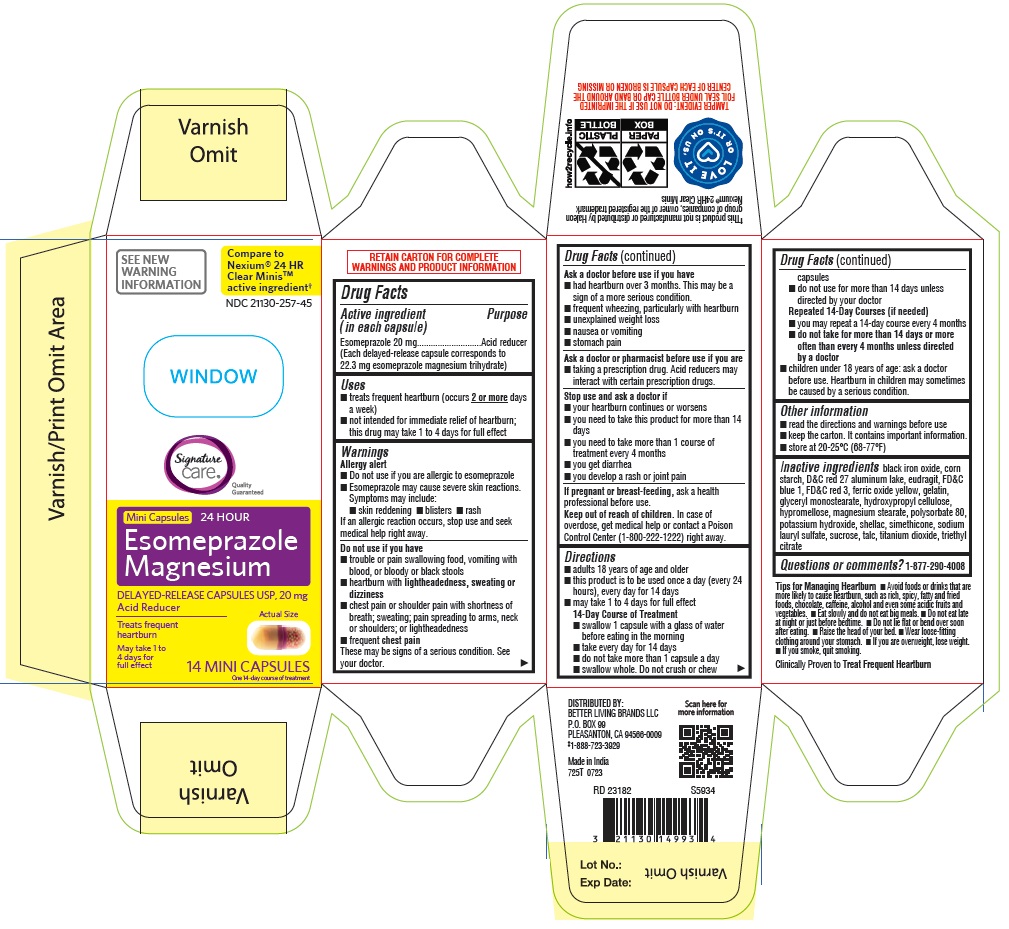
Label: ESOMEPRAZOLE MAGNESIUM capsule, delayed release
- NDC Code(s): 21130-257-41, 21130-257-45
- Packager: BETTER LIVING BRANDS LLC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated April 20, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
Allergy alert
- Do not use if you are allergic to esomeprazole
- Esomeprazole may cause severe skin reactions. Symptoms may include:
skin reddening
blisters
rashIf an allergic reaction occurs, stop use and seek medical help right away.
Do not use if you have
- trouble or pain swallowing food, vomiting with blood, or bloody or black stools
- heartburn with lightheadedness, sweating or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
These may be signs of a serious condition. See your doctor.
Ask a doctor before use if you have
- had heartburn over 3 months. This may be a sign of a more serious condition.
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
Ask a doctor or pharmacist before use if you are
- taking a prescription drug. Acid reducers may interact with certain prescription drugs.
-
DOSAGE & ADMINISTRATION
Directions
-adults 18 years of age and older
-this product is to be used once a day (every 24 hours), every day for 14 days
-may take 1 to 4 days for full effect
14-Day Course of Treatment
- swallow 1 capsule with a glass of water before eating in the morning
- take every day for 14 days
- do not take more than 1 capsule a day
- swallow whole. Do not crush or chew capsules
- do not use for more than 14 days unless directed by your doctor
Repeated 14-Day Courses (if needed)
- you may repeat a 14-day course every 4 months
- do not take for more than 14 days or more often than every 4 months unless directed by a doctor
-children under 18 years of age: ask a doctor before use. Heartburn in children may sometimes be caused by a serious condition.
- STORAGE AND HANDLING
-
INACTIVE INGREDIENT
Inactive ingredients black iron oxide, corn starch, D&C red 27 aluminum lake, eudragit, FD&C blue 1, FD&C red 3, ferric oxide yellow, gelatin, glyceryl monostearate, hydroxypropyl cellulose, hypromellose, magnesium stearate, polysorbate 80, potassium hydroxide, shellac, simethicone, sodium lauryl sulfate, sucrose, talc, titanium dioxide, triethyl citrate
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ESOMEPRAZOLE MAGNESIUM
esomeprazole magnesium capsule, delayed releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21130-257 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ESOMEPRAZOLE MAGNESIUM (UNII: R6DXU4WAY9) (ESOMEPRAZOLE - UNII:N3PA6559FT) ESOMEPRAZOLE 20 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) TALC (UNII: 7SEV7J4R1U) SHELLAC (UNII: 46N107B71O) DIMETHICONE (UNII: 92RU3N3Y1O) SUCROSE (UNII: C151H8M554) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) FERROSOFERRIC OXIDE (UNII: XM0M87F357) METHACRYLIC ACID AND ETHYL ACRYLATE COPOLYMER (UNII: NX76LV5T8J) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) FD&C RED NO. 3 (UNII: PN2ZH5LOQY) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) D&C RED NO. 27 ALUMINUM LAKE (UNII: ZK64F7XSTX) MAGNESIUM STEARATE (UNII: 70097M6I30) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) GELATIN (UNII: 2G86QN327L) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) SODIUM LAURYL SULFATE (UNII: 368GB5141J) POLYSORBATE 80 (UNII: 6OZP39ZG8H) Product Characteristics Color yellow (golden yellow opaque cap, transparent body, light violet to violet color band) Score no score Shape CAPSULE Size 11mm Flavor Imprint Code 112 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21130-257-45 1 in 1 CARTON 04/29/2024 1 14 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:21130-257-41 3 in 1 CARTON 04/29/2024 2 14 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA217264 04/29/2024 Labeler - BETTER LIVING BRANDS LLC. (009137209) Registrant - TIME CAP LABORATORIES, INC. (037052099) Establishment Name Address ID/FEI Business Operations MARKSANS PHARMA LIMITED 925822975 manufacture(21130-257)