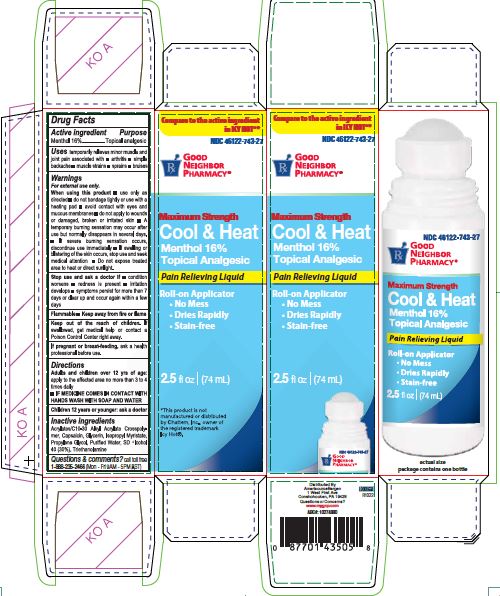
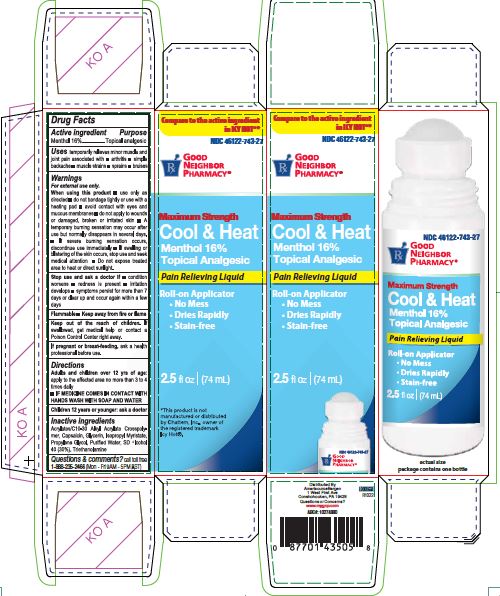
Label: COOL AND HEAT- menthol liquid
- NDC Code(s): 46122-743-27
- Packager: AMERISOURCE BERGEN
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 20, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only.
When using this product ▪ use only as directed ▪ do not bandage tightly or use with a heating pad ▪ avoid contact with eyes and mucous membranes ▪ do not apply to wounds or damaged, broken or irritated skin ▪ A temporary burning sensation may occur after use but normally disappears in several days. ▪ If a severe burning sensation occurs, discontinue use immediately ▪ If swelling or blistering of the skin occurs, stop use and seek and seek medical attention ▪ Do not expose treated area to hear or direct sunlight.
- Stop use and ask a doctor
- SPL UNCLASSIFIED SECTION
- KEEP OUT OF REACH OF CHILDREN
- PREGNANCY OR BREAST FEEDING
- Directions
- Inactive ingredients
- Questions & comments?
-
Carton label
NDC 46122-743-27
GOOD
NEIGHBOR
PHARMACY®Maximum Strength
Cool & Heat
Menthol 16%
Topical Analgesic
Pain Relieving Liquid
Roll-on Applicator
No Mess
Dries Rapidly
Stain-free2.5 fl oz (74mL)
actual size
package contains one bottle
*This product is not
manufactured or distributed
by Chattem Inc., owner of
the registered trademark
Icy Hot®.
Distributed By
AmerisourceBergen
1 West First Ave
Conshohocken, PA 19428
Questions or Concerns?
www.mygnp.com
ABC#: 10274880L0000658
R1022
-
INGREDIENTS AND APPEARANCE
COOL AND HEAT
menthol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:46122-743 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 16 mg in 100 mL Inactive Ingredients Ingredient Name Strength (C10-C30)ALKYL METHACRYLATE ESTER (UNII: XH2FQZ38D8) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) TROLAMINE (UNII: 9O3K93S3TK) ALCOHOL (UNII: 3K9958V90M) CAPSAICIN (UNII: S07O44R1ZM) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:46122-743-27 1 in 1 CARTON 03/15/2023 1 74 mL in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 03/15/2023 Labeler - AMERISOURCE BERGEN (007914906) Registrant - Bionpharma Inc. (079637826) Establishment Name Address ID/FEI Business Operations Pharma Nobis, LLC 118564114 label(46122-743) , pack(46122-743) , analysis(46122-743) , manufacture(46122-743)