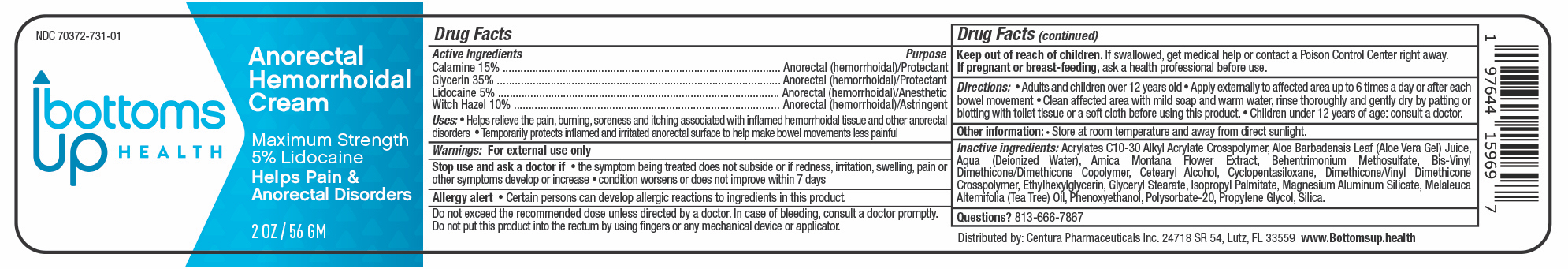
Label: BOTTOMS UP HEALTH- lidocaine,glycerin,calamine,witch hazel cream
- NDC Code(s): 70372-731-01
- Packager: CENTURA PHARMACEUTICALS INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS
- PURPOSE
- USES
- WARNINGS
- STOP USE AND ASK A DOCTOR IF
- ALLERGY ALERT
- OTHER WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- IF PREGNANT OR BREAST FEEDING
-
DIRECTIONS
Adults and children over 12 years old ■ Apply externally to affected area up to 6 times a day or after each bowel movement
■ Clean affected area with mild soap and warm water, rinse thoroughly and gently dry by patting or blotting with toilet tissue or a soft cloth before using this product.
■ Children under 12 years of age: consult a doctor.
- OTHER INFORMATION
-
INACTIVE INGREDIENTS
Acrylates C10-30 Alkyl Acrylate Crosspolymer, Aloe Barbadensis Leaf (Aloe Vera Gel) Juice, Aqua (Deionized Water), Amica Montana Flower Extract, Behentrimonium Methosulfate, Bis-Vinyl Dimethicone/Dimethicone Copolymer, Cetearyl Alcohol, Cyclopentasiloxane, Dimethicone/Vinyl Dimethicone Crosspolymer, Ethylhexylglycerin, Glyceryl Stearate, lsopropyl Palmitate, Magnesium Aluminum Silicate, Melaleuca Alternifolia (Tea Tree) Oil, Phenoxyethanol, Polysorbate-20, Propylene Glycol, Silica.
- QUESTIONS
- PACKAGE LABELING
-
INGREDIENTS AND APPEARANCE
BOTTOMS UP HEALTH
lidocaine,glycerin,calamine,witch hazel creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70372-731 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 5 g in 100 g GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 35 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 15 g in 100 g WITCH HAZEL (UNII: 101I4J0U34) (WITCH HAZEL - UNII:101I4J0U34) WITCH HAZEL 10 g in 100 g Inactive Ingredients Ingredient Name Strength GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) POLYSORBATE 20 (UNII: 7T1F30V5YH) ARNICA MONTANA FLOWER WATER (UNII: U7L2JP51PR) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (SOFT PARTICLE) (UNII: 9E4CO0W6C5) TEA TREE OIL (UNII: VIF565UC2G) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) BEHENTRIMONIUM METHOSULFATE (UNII: 5SHP745C61) PHENOXYETHANOL (UNII: HIE492ZZ3T) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) ALOE VERA LEAF (UNII: ZY81Z83H0X) WATER (UNII: 059QF0KO0R) CARBOMER COPOLYMER TYPE A (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 71DD5V995L) MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70372-731-01 56 g in 1 JAR; Type 0: Not a Combination Product 05/27/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final M015 05/27/2023 Labeler - CENTURA PHARMACEUTICALS INC (084921637) Registrant - CENTURA PHARMACEUTICALS INC (084921637)