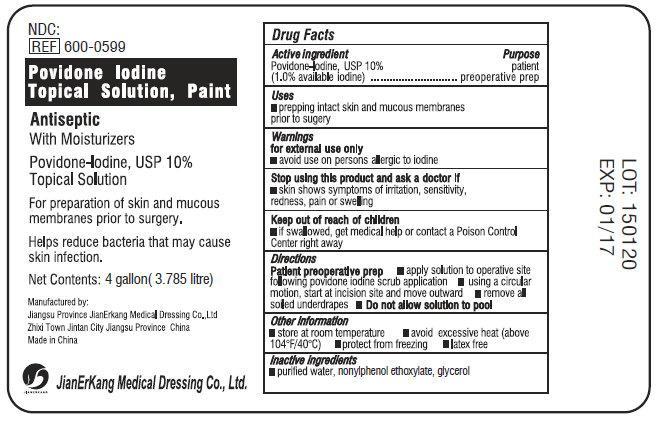
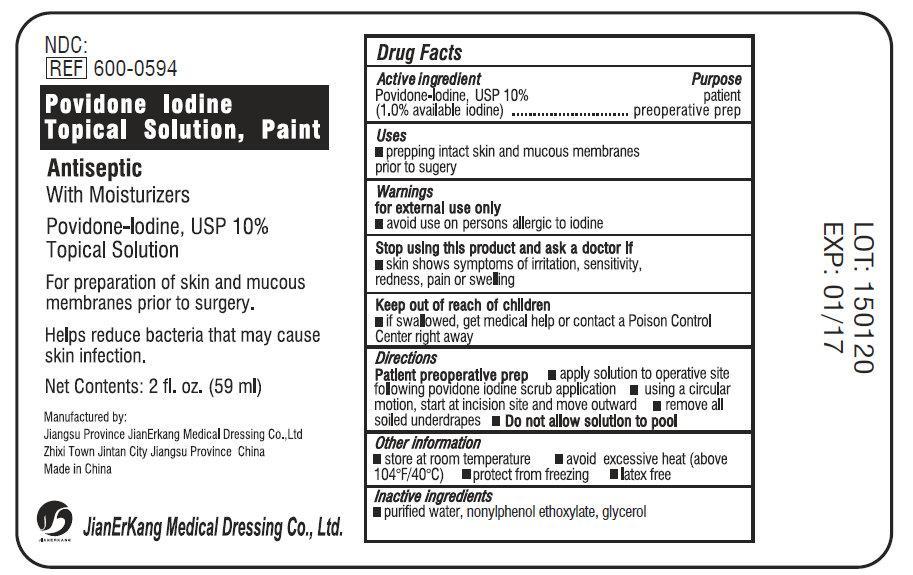
Label: POVIDONE IODINE TOPICAL SOLUTION PAINT- povidone iodine 10% liquid
- NDC Code(s): 34645-1040-1, 34645-1040-2
- Packager: Jianerkang Medical Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 22, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Use
- Warnings
- Stop using this product and ask a doctor if
- Keep out of reach of children.
- Directions
- Other information
- Inactive Ingredients
- Package Labeling
-
INGREDIENTS AND APPEARANCE
POVIDONE IODINE TOPICAL SOLUTION PAINT
povidone iodine 10% liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:34645-1040 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 100 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) NONOXYNOL-9 (UNII: 48Q180SH9T) GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:34645-1040-1 3785 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/31/2015 08/21/2018 2 NDC:34645-1040-2 59 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/31/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 03/31/2015 Labeler - Jianerkang Medical Co., Ltd (530968767) Establishment Name Address ID/FEI Business Operations Jianerkang Medical Co., Ltd 530968767 manufacture(34645-1040)