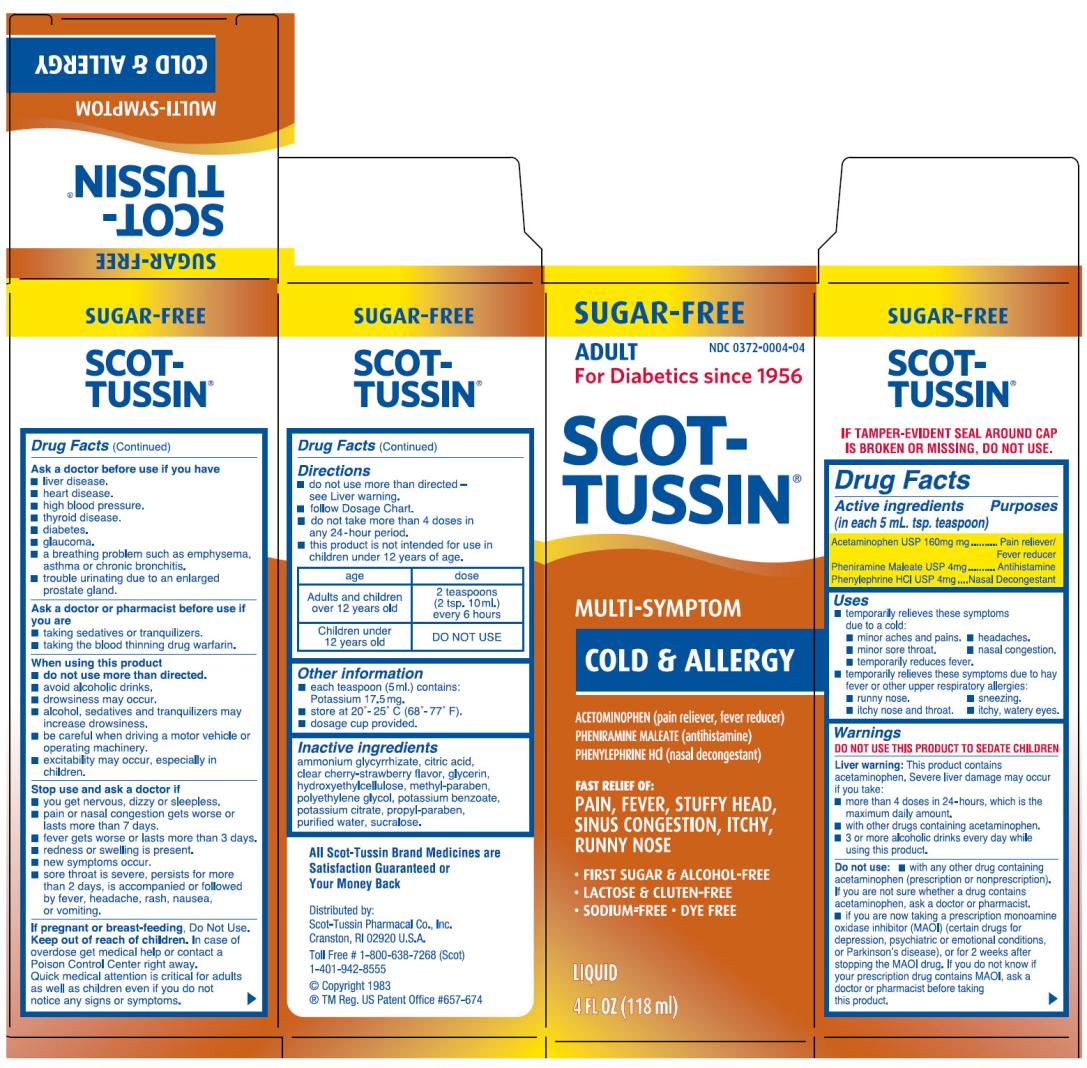
Label: SCOT-TUSSIN ORIGINAL SF MULTI-SYMPTOM- acetominophen, phenylephrine hydrochloride and pheniramine maleate liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 0372-0004-04 - Packager: SCOT-TUSSIN Pharmacal Co., Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 25, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Active Ingredient
- Purpose
- Active Ingredient
- Purpose
-
Uses
- temporarily relieves these symptoms due to a cold:
- minor aches and pains.
- minor sore throat.
- temporarily reduces fever.
- headaches.
- nasal congestion.
- minor aches and pains.
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose.
- itchy nose and throat.
- sneezing.
- itchy, watery eyes.
- runny nose.
- temporarily relieves these symptoms due to a cold:
-
Warnings
DO NOT USE THIS PRODUCT TO SEDATE CHILDREN
Liver warning: This product contains acetaminophne. Severe liver damange may occur if you take:
- more than 4 doses in 24-hours, which is the maximum daily amount.
- with other drugs containing acetaminophen.
- 3 or more alcoholic drinks every day while using this product.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamie oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- liver disease.
- heart disease.
- high blood pressure.
- thyroid disease.
- diabetes.
- glaucoma.
- a breathing problem such as emphysema, asthma or chronic broncititis.
- trouble uritinating due to an enlarged prostate gland.
Ask a doctor or pharmacist before use if you are
- taking sedatives or tranquilizers.
- taking the blood thinning drug warfarin.
When using this product
-
do not use more than directed.
- avoid alcoholic drinks.
- drowsiness may occur.
- alcohol, sedatives and tranquilizers may increase drowsiness.
- be careful when driving a motor vehicle or operating machinery.
- excitability may occur, especially in children.
Stop use and ask a doctor if
- you get nervous, dizzy or sleepless.
- pain or nasal congestion gets worse or lasts more than 7 days.
- fever gets worse or lasts more than 3 days.
- redness or swelling is present.
- new symptoms occur.
- sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting.
- more than 4 doses in 24-hours, which is the maximum daily amount.
-
Directions
- do not use more than directed - see Liver warning.
- follow Dosage Chart.
- do not take more than 4 doses in any 24-hour period.
- this product is not intended for use in children under 12 years of age.
age dose Adults and children
over 12 years old2 teaspoons
(2 tsp. 10ml.)
every 6 hoursChildren under
12 years oldDO NOT USE - do not use more than directed - see Liver warning.
- Other Information
- Inactive Ingredients
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SCOT-TUSSIN ORIGINAL SF MULTI-SYMPTOM
acetominophen, phenylephrine hydrochloride and pheniramine maleate liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0372-0004 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 160 mg in 5 mL PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 4 mg in 5 mL PHENIRAMINE MALEATE (UNII: NYW905655B) (PHENIRAMINE - UNII:134FM9ZZ6M) PHENIRAMINE MALEATE 4 mg in 5 mL Inactive Ingredients Ingredient Name Strength AMMONIUM GLYCYRRHIZATE (UNII: 3VRD35U26C) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) GLYCERIN (UNII: PDC6A3C0OX) HYDROXYETHYL CELLULOSE (2000 MPA.S AT 1%) (UNII: S38J6RZN16) METHYLPARABEN (UNII: A2I8C7HI9T) POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) POTASSIUM BENZOATE (UNII: 763YQN2K7K) POTASSIUM CITRATE (UNII: EE90ONI6FF) PROPYLPARABEN (UNII: Z8IX2SC1OH) SUCRALOSE (UNII: 96K6UQ3ZD4) WATER (UNII: 059QF0KO0R) Product Characteristics Color Score Shape Size Flavor CHERRY, STRAWBERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0372-0004-04 118 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part341 12/01/1956 Labeler - SCOT-TUSSIN Pharmacal Co., Inc. (001203918)