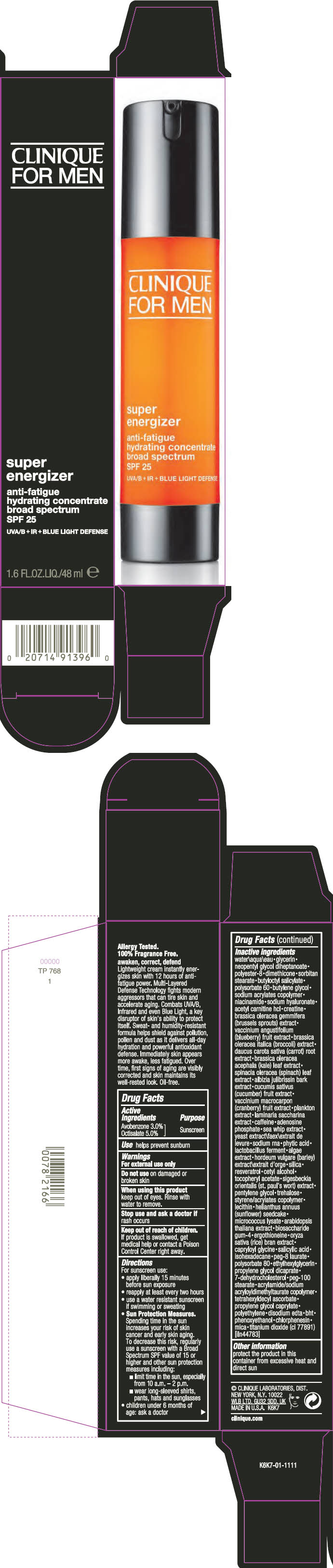
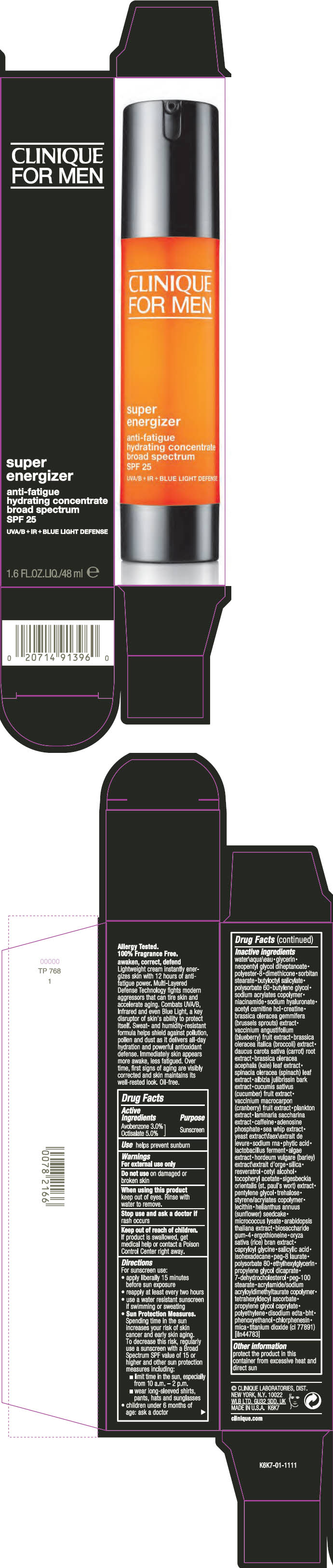
Label: CLINIQUE FOR MEN SUPER ENGERGIZER ANTI -FATIGUE HYDRATING CONCENTRATE BROAD SPECTRUM SPF 25- avobenzone and octisalate cream
- NDC Code(s): 49527-063-01
- Packager: CLINIQUE LABORATORIES LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredients
- Purpose
- Use
- Warnings
-
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply at least every two hours
- use a water resistant sunscreen if swimming or sweating
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: ask a doctor
-
Inactive ingredients
water\aqua\eau • glycerin • neopentyl glycol diheptanoate • polyester-8 • dimethicone • sorbitan stearate • butyloctyl salicylate • polysorbate 60 • butylene glycol • sodium acrylates copolymer • niacinamide • sodium hyaluronate • acetyl carnitine hcl • creatine • brassica oleracea gemmifera (brussels sprouts) extract • vaccinium angustifolium (blueberry) fruit extract • brassica oleracea italica (broccoli) extract • daucus carota sativa (carrot) root extract • brassica oleracea acephala (kale) leaf extract • spinacia oleracea (spinach) leaf extract • albizia julibrissin bark extract • cucumis sativus (cucumber) fruit extract • vaccinium macrocarpon (cranberry) fruit extract • plankton extract • laminaria saccharina extract • caffeine • adenosine phosphate • sea whip extract • yeast extract\faex\extrait de levure • sodium rna • phytic acid • lactobacillus ferment • algae extract • hordeum vulgare (barley) extract\extrait d'orge • silica • resveratrol • cetyl alcohol • tocopheryl acetate • sigesbeckia orientalis (st. paul's wort) extract • pentylene glycol • trehalose • styrene/acrylates copolymer • lecithin • helianthus annuus (sunflower) seedcake • micrococcus lysate • arabidopsis thaliana extract • biosaccharide gum-4 • ergothioneine • oryza sativa (rice) bran extract • capryloyl glycine • salicylic acid • isohexadecane • peg-8 laurate • polysorbate 80 • ethylhexylglycerin • propylene glycol dicaprate • 7-dehydrocholesterol • peg-100 stearate • acrylamide/sodium acryloyldimethyltaurate copolymer • tetrahexyldecyl ascorbate • propylene glycol caprylate • polyethylene • disodium edta • bht • phenoxyethanol • chlorphenesin • mica • titanium dioxide (ci 77891) [iln44783]
- Other information
- PRINCIPAL DISPLAY PANEL - 48 ml Bottle Carton
-
INGREDIENTS AND APPEARANCE
CLINIQUE FOR MEN SUPER ENGERGIZER ANTI -FATIGUE HYDRATING CONCENTRATE BROAD SPECTRUM SPF 25
avobenzone and octisalate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49527-063 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL Inactive Ingredients Ingredient Name Strength CARROT (UNII: L56Z1JK48B) KALE (UNII: 0Y3L4J38H1) SPINACH (UNII: 6WO75C6WVB) CUCUMBER (UNII: YY7C30VXJT) CRANBERRY (UNII: 0MVO31Q3QS) SACCHARINA LATISSIMA (UNII: 68CMP2MB55) CAFFEINE (UNII: 3G6A5W338E) ADENOSINE PHOSPHATE (UNII: 415SHH325A) PSEUDOPTEROGORGIA ELISABETHAE (UNII: UDY3H1OUX5) YEAST, UNSPECIFIED (UNII: 3NY3SM6B8U) FYTIC ACID (UNII: 7IGF0S7R8I) LIMOSILACTOBACILLUS REUTERI (UNII: 9913I24QEE) BARLEY (UNII: 5PWM7YLI7R) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) RESVERATROL (UNII: Q369O8926L) CETYL ALCOHOL (UNII: 936JST6JCN) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) PENTYLENE GLYCOL (UNII: 50C1307PZG) TREHALOSE (UNII: B8WCK70T7I) HELIANTHUS ANNUUS SEEDCAKE (UNII: 482WYF7XLC) ARABIDOPSIS THALIANA WHOLE (UNII: AI3L60HQ81) BIOSACCHARIDE GUM-4 (UNII: 9XRL057X90) ERGOTHIONEINE (UNII: BDZ3DQM98W) WHITE RICE (UNII: A195V20H7A) CAPRYLOYL GLYCINE (UNII: 8TY5YO42NJ) SALICYLIC ACID (UNII: O414PZ4LPZ) ISOHEXADECANE (UNII: 918X1OUF1E) PEG-8 LAURATE (UNII: 762O8IWA10) POLYSORBATE 80 (UNII: 6OZP39ZG8H) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) PROPYLENE GLYCOL DICAPRATE (UNII: U783H9JHWY) 7-DEHYDROCHOLESTEROL (UNII: BK1IU07GKF) PEG-100 STEARATE (UNII: YD01N1999R) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) PROPYLENE GLYCOL MONOCAPRATE (UNII: 19E9J0YN67) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DIHEPTANOATE (UNII: 5LKW3C543X) POLYESTER-8 (1400 MW, CYANODIPHENYLPROPENOYL CAPPED) (UNII: T9296U138P) DIMETHICONE (UNII: 92RU3N3Y1O) SORBITAN MONOSTEARATE (UNII: NVZ4I0H58X) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) POLYSORBATE 60 (UNII: CAL22UVI4M) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) NIACINAMIDE (UNII: 25X51I8RD4) HYALURONATE SODIUM (UNII: YSE9PPT4TH) LEVACECARNINE HYDROCHLORIDE (UNII: NDW10MX58T) CREATINE (UNII: MU72812GK0) BRUSSELS SPROUT (UNII: KHX46H31F8) BLUEBERRY (UNII: 253RUG1X1A) BROCCOLI (UNII: UOI4FT57BZ) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) PHENOXYETHANOL (UNII: HIE492ZZ3T) CHLORPHENESIN (UNII: I670DAL4SZ) MICA (UNII: V8A1AW0880) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49527-063-01 1 in 1 CARTON 10/01/2018 1 48 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 10/01/2018 Labeler - CLINIQUE LABORATORIES LLC (044475127) Registrant - Estee Lauder Companies Inc. (790802086) Establishment Name Address ID/FEI Business Operations The Estee Lauder Inc 802599436 manufacture(49527-063) , pack(49527-063) , label(49527-063)