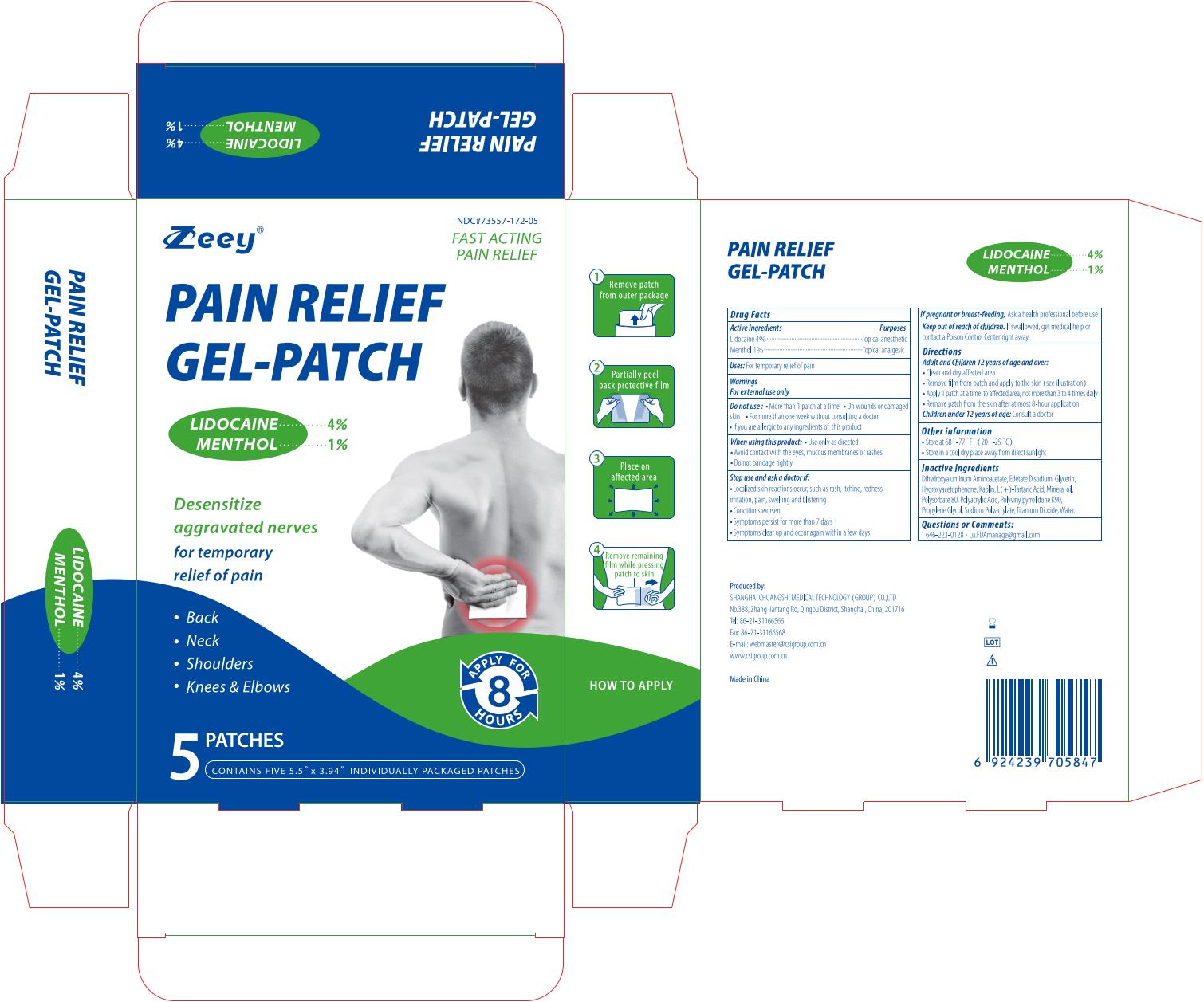
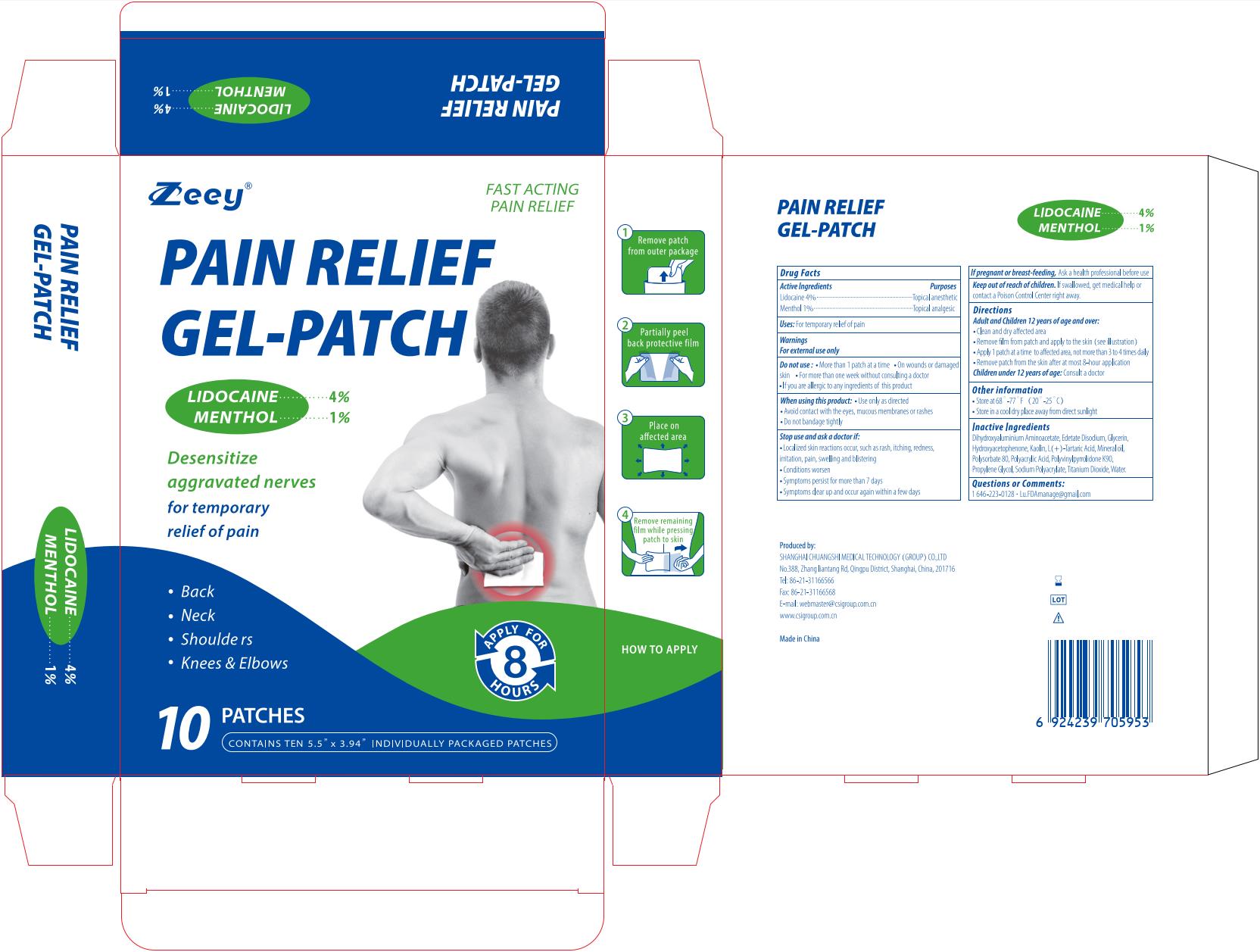
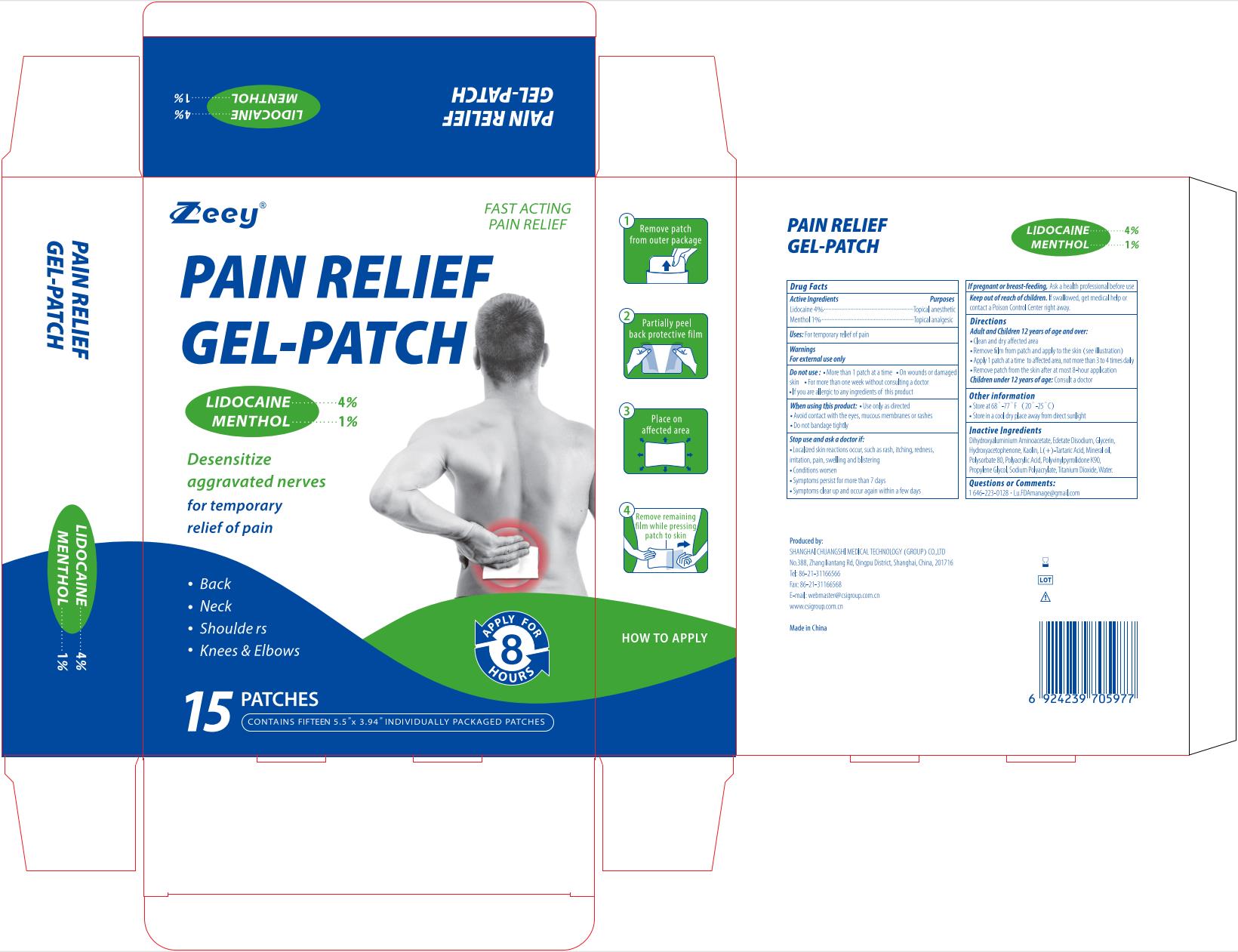
Label: PAIN RELIEF GEL-PATCH- lidocaine and menthol patch
- NDC Code(s): 73557-172-01, 73557-172-05, 73557-172-10, 73557-172-15
- Packager: Shanghai Chuangshi Medical Technology (Group) Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 29, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purposes
- Uses
- Warnings
- When using this product:
- Do not use:
- Stop use and ask a doctor if:
- If pregnant or breast-feeding,
- Keep out of reach of children.
-
Directions
Adult and Children 12 years of age and over:
- Clean and dry affected area
- Remove film from patch and apply to the skin (see illustration)
- Apply 1 patch at a time to affected area, not more than 3 to 4 times daily
- Remove patch from the skin after at most 8-hour application
Children under 12 years of age:Consult a doctor
- Inactive Ingredients
- Questions or Comments
- Other information
- Package label. Principal display panel
-
INGREDIENTS AND APPEARANCE
PAIN RELIEF GEL-PATCH
lidocaine and menthol patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73557-172 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 0.01 g in 1 g LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 0.04 g in 1 g Inactive Ingredients Ingredient Name Strength KAOLIN (UNII: 24H4NWX5CO) TARTARIC ACID (UNII: W4888I119H) WATER (UNII: 059QF0KO0R) DIHYDROXYALUMINUM AMINOACETATE ANHYDROUS (UNII: 1K713C615K) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POLYACRYLIC ACID (250000 MW) (UNII: 9G2MAD7J6W) SODIUM POLYACRYLATE (2500000 MW) (UNII: 05I15JNI2J) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) MINERAL OIL (UNII: T5L8T28FGP) POLYSORBATE 80 (UNII: 6OZP39ZG8H) GLYCERIN (UNII: PDC6A3C0OX) EDETATE DISODIUM (UNII: 7FLD91C86K) POVIDONE K90 (UNII: RDH86HJV5Z) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73557-172-05 5 in 1 BOX 12/30/2022 1 NDC:73557-172-01 12 g in 1 PATCH; Type 0: Not a Combination Product 2 NDC:73557-172-10 10 in 1 BOX 01/09/2024 2 NDC:73557-172-01 12 g in 1 PATCH; Type 0: Not a Combination Product 3 NDC:73557-172-15 15 in 1 BOX 01/09/2024 3 NDC:73557-172-01 12 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 12/30/2022 Labeler - Shanghai Chuangshi Medical Technology (Group) Co., Ltd. (546872672) Registrant - Shanghai Chuangshi Medical Technology (Group) Co., Ltd. (546872672) Establishment Name Address ID/FEI Business Operations Shanghai Chuangshi Medical Technology (Group) Co., Ltd. 546872672 manufacture(73557-172) , label(73557-172)