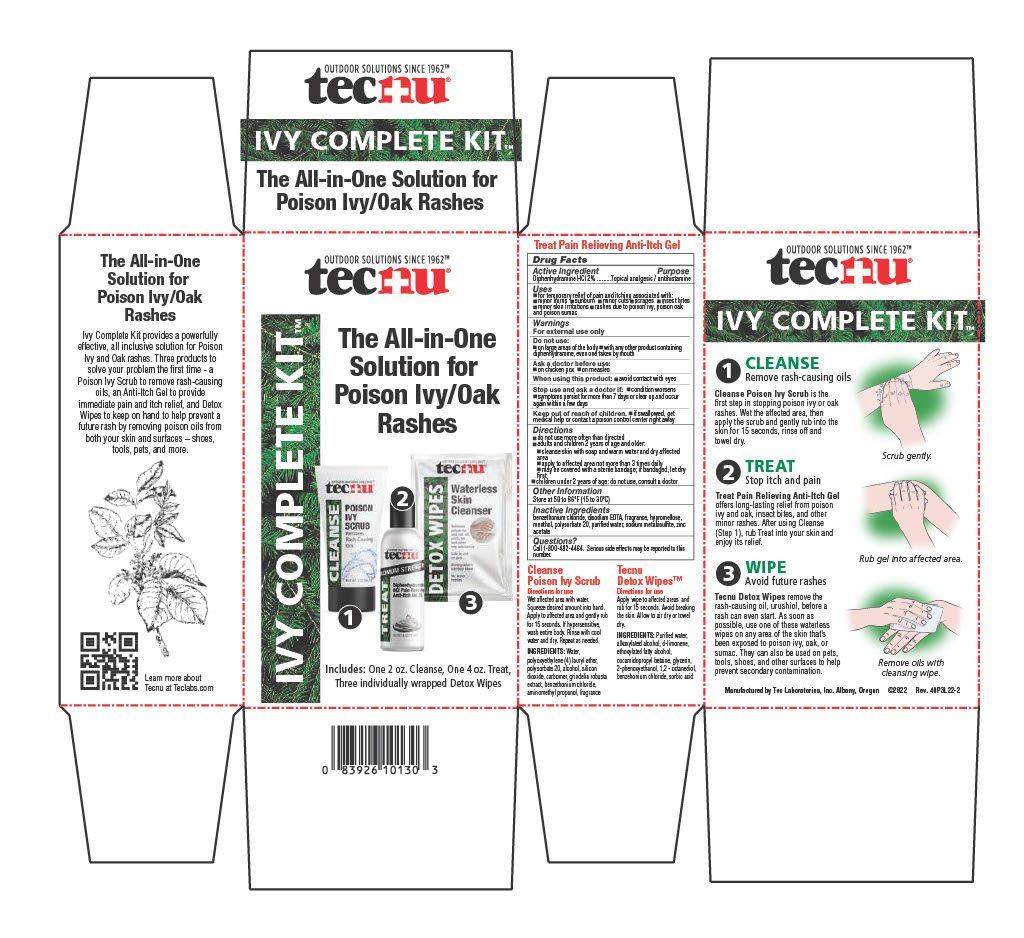
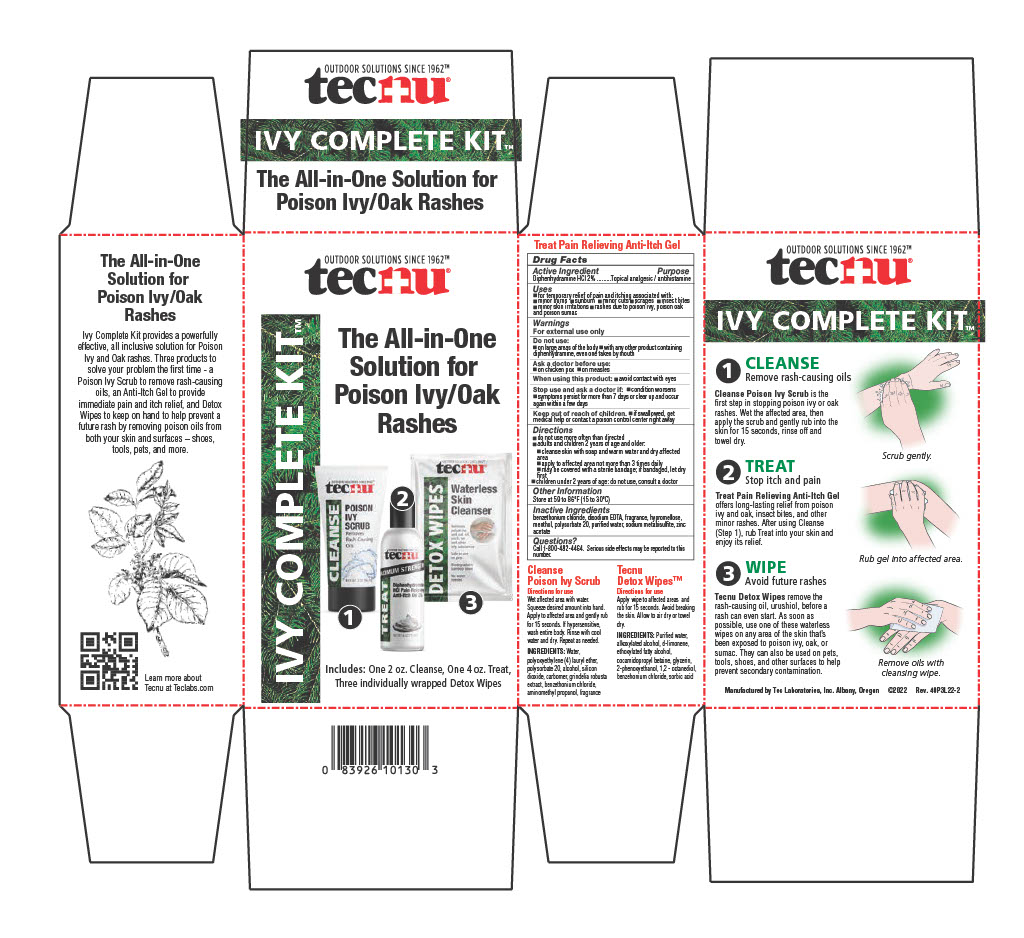
Label: TECNU IVY COMPLETE KIT- ivy treatment kit
- NDC Code(s): 51879-400-04, 51879-410-04
- Packager: Tec Laboratories Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 3, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- ASK DOCTOR
- KEEP OUT OF REACH OF CHILDREN
- WHEN USING
- STOP USE
-
DOSAGE & ADMINISTRATION
Directions
- do not use more than directed
- adults and children 2 years of age and older:
- cleanse skin with soap and warm water and dry affected area
- apply to affected area not more than 3 times daily
- may be covered with a bandage; if bandage let dry first
- children under 2 years of age: do not use, consult a doctor
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TECNU IVY COMPLETE KIT
ivy treatment kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51879-400 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51879-400-04 1 in 1 CARTON; Type 0: Not a Combination Product 01/16/2023 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 113.4 g Part 1 of 1 TECNU TREAT
diphenhydramine hydrochloride gelProduct Information Item Code (Source) NDC:51879-410 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 20 mg in 1 g Inactive Ingredients Ingredient Name Strength ZINC ACETATE (UNII: FM5526K07A) WATER (UNII: 059QF0KO0R) POLYSORBATE 20 (UNII: 7T1F30V5YH) BENZETHONIUM CHLORIDE (UNII: PH41D05744) MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) EDETATE DISODIUM (UNII: 7FLD91C86K) HYPROMELLOSE 2208 (100 MPA.S) (UNII: B1QE5P712K) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51879-410-04 113.4 g in 1 BOTTLE; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 01/16/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 01/16/2023 Labeler - Tec Laboratories Inc. (083647792) Establishment Name Address ID/FEI Business Operations Tec Laboratories, Inc. 083647792 manufacture(51879-400)